Abstract
Objective
This study was performed to evaluate the clinical efficacy of subcostal thoracoscopy and median sternotomy as surgical approaches for thymoma resection and lymph node dissection. The feasibility, safety, and clinical outcomes of subcostal thoracoscopy were compared with those of median sternotomy.
Methods
The clinical data of 335 patients with thymoma were retrospectively analyzed. The patients were divided into the subcostal thoracoscopy group and the median sternotomy group. Propensity score matching was performed to obtain comparable subsets of 50 patients in each group. A comparative analysis was conducted on various parameters.
Results
All surgeries were successful, and no conversions to open thoracotomy were required in the subcostal thoracoscopy group. Significant differences in the operative time, intraoperative blood loss, chest tube drainage duration, postoperative hospital stay, patient satisfaction scores, pain assessment, and postoperative complications were observed between the two groups. However, there was no significant difference in the number of lymph nodes or lymph node stations dissected intraoperatively between the two groups.
Conclusion
Subcostal thoracoscopy is not inferior to median sternotomy as a surgical approach for thymoma resection and lymph node dissection. Our research provides important new comparative data on minimally invasive thymoma resection.
Keywords
Introduction
Anterior mediastinal lesions, particularly thymic tumors, are relatively common in thoracic surgery, and approximately 50% of anterior mediastinal tumors are thymomas. The incidence rate of thymomas is approximately 1.5 per million population overall, but it is even higher at 3.74 per million in Asian populations. 1 Because thymoma is a rare disease, many of its aspects remain unclear in clinical practice. Moreover, thymomas present with diverse clinical manifestations. In addition to symptoms of local tumor compression and invasion, a significant proportion of patients also exhibit systemic disorders such as myasthenia gravis, pure red cell aplasia, and hypogammaglobulinemia, further complicating the diagnosis and treatment of thymomas. Importantly, all thymomas should be considered potentially malignant tumors, necessitating a multidisciplinary approach involving thoracic surgery, neurology, oncology, radiology, and radiation therapy. The treatment of thymic tumors primarily relies on surgical intervention, aiming to both relieve tumor-induced compression and symptoms as well as establish an accurate diagnosis. The clinical guideline developed by Ontario Health’s Program recommended surgery as the first-line treatment for thymomas. 2 Median sternotomy is considered the standard surgical approach. 3 However, this approach has been criticized by thoracic surgeons because of its invasiveness. After median sternotomy, patients often experience significant postoperative pain and discomfort, and the recovery time is slow. Because the incision is relatively large, it is more susceptible to infection. Although the advent of thoracoscopic surgery has introduced various minimally invasive approaches, there is still a lack of extensive clinical data regarding the safety and reliability of these minimally invasive techniques.
Through a propensity score-matching analysis, we retrospectively analyzed clinical data of patients with thymoma who underwent surgical treatment at the Department of Thoracic Surgery, Tangdu Hospital, Air Force Military Medical University from January 2011 to December 2018.
Methods
Clinical data and grouping
We retrospectively analyzed the clinical data of patients with thymoma who underwent surgical treatment at the Department of Thoracic Surgery, Tangdu Hospital, Air Force Military Medical University, from January 2011 to December 2018. Patients with concomitant myasthenia gravis were preoperatively evaluated by the same medical team in the neurology department. The patients were in stable condition and underwent liver, gallbladder, pancreas, spleen, and kidney ultrasonography; cardiac ultrasound; electrocardiography; cranial computed tomography; contrast-enhanced chest computed tomography (or conventional chest computed tomography if the use of contrast was contraindicated); chest magnetic resonance imaging; blood gas analysis; pulmonary ventilation and diffusion function tests; routine blood tests; and other examinations to exclude surgical contraindications. The main inclusion criteria were satisfactory cardiopulmonary function for surgery, absence of distant metastasis, and complete tumor resection (R0 resection). Based on the above criteria, 335 patients were finally included in the study. They were divided into two groups according to the surgical approach: the subcostal thoracoscopy group, which comprised 285 patients who underwent thymoma resection and lymph node dissection via the subcostal approach, and the median sternotomy group, which comprised 50 patients who underwent thymoma resection and lymph node dissection via the median sternotomy approach. The baseline characteristics of the patients in the subcostal thoracoscopy group were matched with those of the patients in the median sternotomy group using propensity score matching, resulting in a final sample of 50 patients in each group. The subcostal thoracoscopy group consisted of 28 men and 22 women with a mean age of 49.2 ± 11.2 years (range, 28–69 years), while the median sternotomy group consisted of 31 men and 19 women with a mean age of 50.5 ± 10.8 years (range, 23–71 years). The clinical baseline characteristics of the patients in the two groups after propensity score matching are presented in Table 1. Comparative analysis was performed on various parameters including the surgical time, intraoperative blood loss, chest drainage duration, number of lymph node stations dissected, number of lymph nodes dissected, postoperative hospital stay, patient satisfaction, pain status, and complications (including incision infection, pulmonary infection, and myasthenic crisis).
Basic clinical data after propensity score matching.

Data are presented as mean ± standard deviation or number of patients.
BMI, body mass index; pO2, oxygen partial pressure.
Surgical positioning and techniques
All patients underwent surgery under general anesthesia with a single-lumen endotracheal tube. All surgeries were performed by the same surgeon and first assistant. In the median sternotomy group, the patients were placed in the supine position with the operating surgeon positioned on the right side of the patient and the first assistant on the left side. The midline of the sternum was identified, and the sternum was split using an electric saw. Retractors were used to spread the sternum apart, providing a clear view for resection of the thymoma. In the thoracoscopic group, the patients were placed in the supine position with the legs apart, forming a “T” shape. A 20- to 40-mm longitudinal incision was made below the sternum as an observation port, and a puncture guide was inserted into the anterior mediastinum through the sternum. Carbon dioxide at 8 cmH2O pressure (1 cmH2O = 0.098 kPa) was insufflated, and using the index finger as a guide, 5-mm puncture guides were inserted below the left and right rib margins as operative ports to establish the surgical system.4–7
Both groups of patients underwent thymoma resection and lymph node dissection, which included resection of the thymoma and surrounding fat, lymph node dissection, and resection of invaded organs or tissues if necessary. In the subcostal thoracoscopy group, the surgery was performed using only a pair of grasping forceps and an ultrasonic scalpel. 8 The surgical scope was consistent in both groups, extending upward to the junction of the inferior pole of the thyroid gland and the superior pole of the thymus, downward to the anterior surface of the pericardium, and laterally to 2.0 cm medial to the phrenic nerve on both sides. Complete resection of the thymoma and surrounding fat was ensured. If the tumor invaded the pericardium, partial pericardial resection was performed. If the left brachiocephalic vein was invaded, resection of the brachiocephalic vein was performed. If the tumor invaded the lung lobe, wedge resection of the lung tissue was performed. If the phrenic nerve was invaded, phrenic nerve dissection was performed. After resection of the tumor and thymus, simultaneous lymph node dissection was performed. For lymph node dissection, the lymph nodes were divided into nine regions based on the recommendations of the International Thymic Malignancy Interest Group (ITMIG) and the International Association for the Study of Lung Cancer (IASLC). 9 These regions are shown in Figure 1 and were defined as follows. Zone 1 extended from the right side of the midline of the trachea to the inner side of the right brachiocephalic/internal jugular vein, superiorly to the lower pole of the thyroid gland, and inferiorly to the upper margin of the left brachiocephalic vein. This zone corresponded to the ITMIG deep right paratracheal and IASLC 2R lymph node groups. Zone 2 extended from the left side of the midline of the trachea to the inner side of the left brachiocephalic/internal jugular vein and the inner side of the left common carotid artery, superiorly to the lower pole of the thyroid gland, and inferiorly to the upper border of the aortic arch. This zone corresponded to the ITMIG deep left paratracheal and IASLC 2L lymph node groups. Zone 3 extended from the mediastinal pleura on the right and sides of the phrenic nerve to the inner margin of the superior vena cava, extending outward, and Zone 6 extended from the mediastinal pleura on the left and inner sides of the phrenic nerve. Zones 3 and 6 extended superiorly to the upper pole of the pleural cavity, anteriorly to the sternum, inferiorly to the xiphoid process, and posteriorly to the anterior aspect of the superior vena cava. These zones corresponded to the ITMIG prevascular anterior, para-aortic, ascending aorta, and IASLC 3a lymph node groups. Zone 4 coursed between the superior vena cava and the aorta, extending superiorly to the lower margin of the left brachiocephalic vein and inferiorly to the level of the pericardial reflection at the root of the superior vena cava and the beginning of the ascending aorta. This zone corresponded to the ITMIG lower deep paratracheal and IASLC 4 lymph node groups. Zone 5 extended along the lower margin of the brachiocephalic vein and near the aortic arch, superiorly to the lower margin of the left brachiocephalic vein, and inferiorly to the level of the pulmonary artery trunk. This zone corresponded to the ITMIG deep paratracheal subaortic/subaortic pulmonary window and IASLC 5 + 6 lymph node groups. Zone 7 was the right para-cardiac region, and Zone 8 was the pericardial region. Finally, Zone 9 was the left para-cardiac diaphragmatic region, corresponding to the ITMIG thymic periphery, diaphragmatic, lower phrenic nerve, and pericardial lymph node groups.
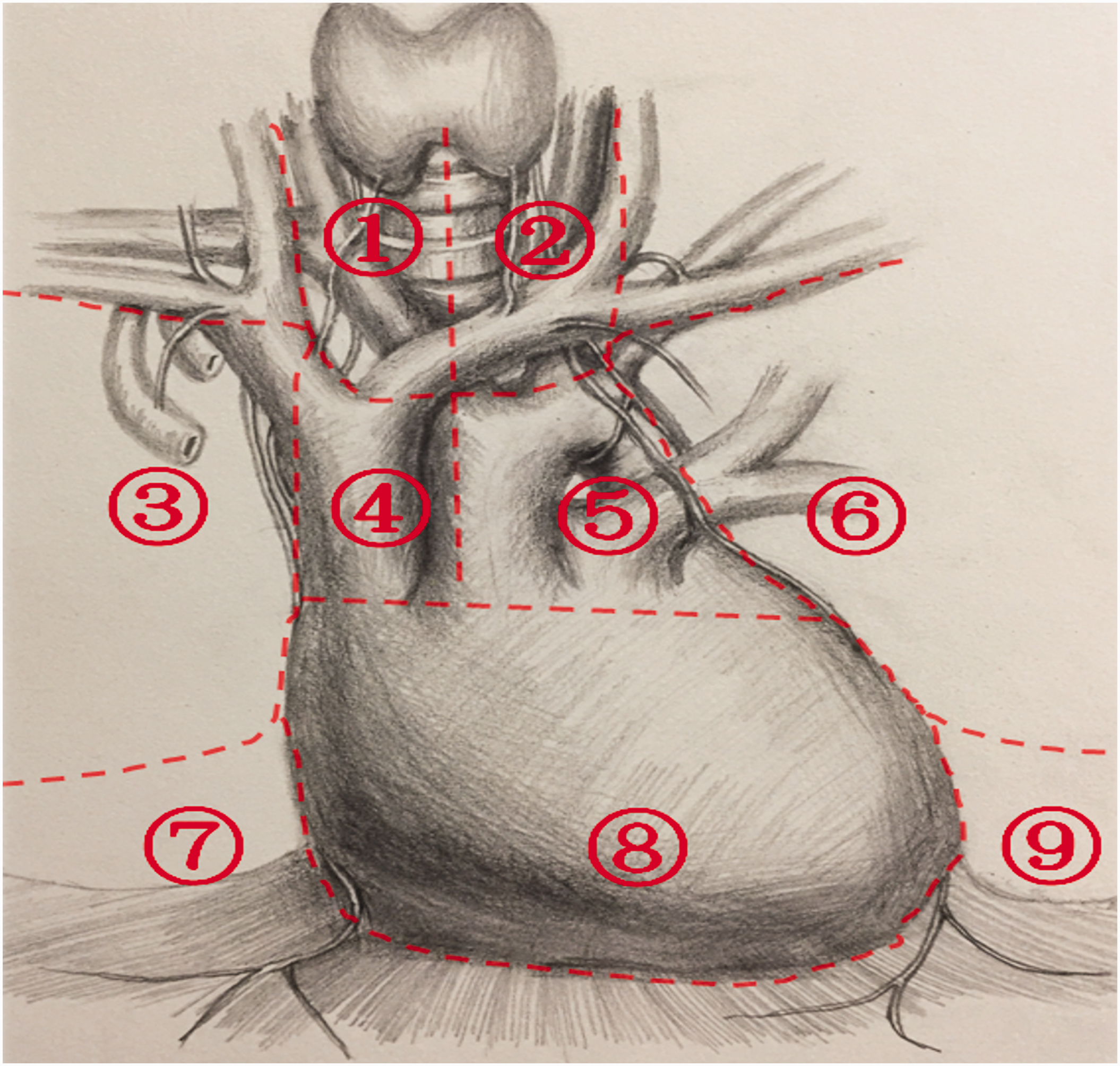
Nine lymph node regions based on the recommendations of the International Thymic Malignancy Interest Group and the International Association for the Study of Lung Cancer.
In patients whose tumor was relatively small and myasthenia gravis symptoms were mild, surgical intervention was the primary treatment; myasthenia gravis-specific therapies were then administered. In patients whose tumor was large but myasthenia gravis symptoms were relatively mild, a combination of radiotherapy, chemotherapy, and other adjuvant treatments was utilized to reduce the tumor size. For patients whose myasthenia gravis symptoms were severe but the tumor was not large, aggressive internal medicine management was essential. Patients with a complex and severe presentation of both myasthenia gravis symptoms and tumors were excluded from the study. After surgery, the patients with myasthenia gravis were admitted to the intensive care unit and closely monitored for development of myasthenic crisis. Medications were resumed after surgery.
The primary reason for the lack of significant postoperative symptom improvement in patients with myasthenia gravis is the presence of ectopic thymic tissue. Ectopic thymic tissue is commonly found in the surrounding adipose tissue of the anterior mediastinum, making the conventional approach of basic thymic range resection (T2a) less commonly used. In the present study, we performed extended thymectomy (T2b) when the patient had myasthenia gravis.
Data collection and standards
Data on the surgical time, intraoperative blood loss, chest tube drainage time, number of lymph node stations dissected, number of lymph nodes dissected, postoperative hospital stay, patient satisfaction, pain levels, and complications were collected and recorded for both groups. The surgical time included the time required for chest opening, thymoma resection, and lymph node dissection, and chest closing. The blood loss was recorded by measuring the volume of fluid in the suction device and the amount of blood on the gauze before chest irrigation. The chest tube drainage time was determined based on the time of chest tube removal, with a drainage volume of <50 mL/day used as the criterion for tube removal. The numbers of dissected lymph nodes and stations were based on the postoperative pathological report. For patients with concomitant myasthenia gravis, improvement in myasthenia gravis symptoms and/or a reduction in the medication dosage were used as evaluation criteria. Patient satisfaction scores were assessed during follow-up visits at 30 to 60 days after surgery, with a score of 10 indicating high satisfaction and 0 indicating dissatisfaction. Pain scores were measured using a visual analogue scale, with 0 indicating no pain and 10 indicating severe pain. Pain scores were recorded on postoperative days 1, 2, 3, and 30. Figure 2 shows a flow diagram of the study.

Flow diagram of the study.
Statistical analysis
Propensity score matching and data analysis were performed using SPSS 20.0 (IBM Corp., Armonk, NY, USA). Normally distributed continuous data are expressed as mean ± standard deviation, while non-normally distributed continuous data are expressed as median. Prior to the data analysis, normality and homogeneity of variance tests were conducted. Normally distributed and homogeneous continuous data were compared using the t-test, while non-normally distributed or heterogeneous continuous data were compared using the Mann–Whitney U test. Categorical data were analyzed using the chi-square test or Fisher’s exact test. A P value of <0.05 was considered statistically significant.
Ethics review
This study was approved by the Ethics Committee of Tangdu Hospital, Air Force Military Medical University (Ethics Approval No. K202104-09). Each patient provided written informed consent prior to undergoing surgery. We have de-identified all patients’ details. The reporting of this study conforms to the STROBE guidelines. 10
Results
All surgeries were successfully completed, and R0 resection was achieved in all patients in the subcostal thoracoscopy group without the need for sternotomy. A comparison of the perioperative indicators between the two groups is detailed in Table 2. Compared with the median sternotomy group, the subcostal thoracoscopy group had a significantly shorter operation time (46.2 ± 19.5 vs. 63.4 ± 23.5 min), less intraoperative blood loss (38.2 ± 15.2 vs. 102.8 ± 27.5 mL), no placement of a chest drainage tube (0.0 vs. 3.4 ± 1.2 days), a shorter postoperative hospital stay (2.9 ± 1.9 vs. 4.6 ± 1.7 days), higher patient satisfaction scores (7.9 ± 2.1 vs. 6.7 ± 1.2), lower pain scores, and a lower incidence of complications (10% vs. 26%) (P < 0.05 for all). However, there were no statistically significant differences between the two groups in the number of lymph node stations dissected (7.9 ± 0.6 vs. 8.0 ± 0.7), number of lymph nodes dissected (8.9 ± 2.3 vs. 9.2 ± 2.7), or resolution of myasthenia gravis symptoms.
Comparison of perioperative results.

Data are presented as mean ± standard deviation or number of patients.
Discussion
Because of the extremely low incidence of thymomas, the diagnostic and therapeutic progress for thymomas has been slow. Unlike other tumors, the malignancy of thymomas cannot be determined solely based on histology; it also requires the evaluation of capsular invasion, surrounding organ invasion, and distant metastasis. All thymomas are considered potentially malignant, and surgical resection is the standard treatment for thymic tumors. Traditional surgical approaches include median sternotomy, transverse sternotomy, and unilateral thoracotomy (left or right), and thoracoscopy has evolved to include unilateral (left or right) or bilateral thoracoscopic thymectomy. Since the introduction of the da Vinci robot, many scholars have adopted robotic-assisted surgery for thymoma treatment. 11 With the continuous advancements in medical technology, the goals of thymoma surgery are to achieve complete tumor resection, alleviate or resolve clinical symptoms, minimize surgical complications, and promote minimally invasive and painless procedures. However, because thymic tumors are relatively indolent, long-term survival can be achieved even in patients with disease progression or recurrence, making it difficult to conduct large-scale prospective randomized studies. Therefore, there is a lack of long-term follow-up evidence for minimally invasive approaches. Expert consensus suggests that minimally invasive surgery can be considered as long as the treatment goal of complete tumor resection is met, but careful preoperative evaluation of conditions such as myasthenia gravis is necessary. 12 Different centers adopt different minimally invasive approaches, and there are variations in reported efficacy. Professor Yongan Zhou from the Department of Thoracic Surgery, Tangdu Hospital has been performing thoracoscopic thymectomy through the xiphoid approach since December 2013. With several years of development and improvement, the procedure has become safer and more effective. It combines the advantages of various surgical techniques and solves the technical challenges of thoracic thymoma surgery in terms of maximizing aesthetics, using a minimally invasive approach, and ensuring thoroughness.
The surgery time is significantly shorter when using the subcostal approach below the xiphoid process than when using the median sternotomy approach. Two main factors may contribute to this difference. First, the minimally invasive subcostal approach requires only a grasping forceps and an extended ultrasonic scalpel, eliminating the need for additional instruments. 13 This surgical procedure has become increasingly streamlined and proficient over time. Second, the median sternotomy procedure may take a longer time to complete because of the steps involved in opening and closing the chest as well as the placement of thoracic drainage tubes. By contrast, the subcostal approach does not require the placement of thoracic drainage tubes, and the incision length is only 20 to 40 mm, resulting in shorter suturing time than that required for open chest procedures. Notably, however, our study was limited by the lack of a separate analysis of the time spent opening and closing the chest, resecting the tumor and surrounding thymic tissue, and clearing the lymph nodes. Our experience suggests that there is no significant difference in the tumor resection time between the two procedures. However, lymph node clearance may be slightly faster when performing median sternotomy than when using the subcostal approach. Furthermore, the median sternotomy group had significantly greater intraoperative blood loss in the present study. This can be attributed to the fact that the median sternotomy approach often requires the use of gauze packing for hemostasis during chest opening and surgical procedures, leading to a higher amount of blood loss. Additionally, thoracoscopy itself provides magnified vision, and to maintain a clear visual field and prevent contamination, even minimal bleeding during the minimally invasive procedure is promptly controlled, with the use of gauze being infrequent.
Our statistical analysis revealed several additional advantages of the subcostal approach over median sternotomy. First, the patients in the subcostal approach group did not require postoperative placement of thoracic drainage tubes, allowing for early mobilization. The safety of not placing thoracic drainage tubes has been validated. 14 By contrast, the patients in the median sternotomy group routinely underwent placement of two thoracic drainage tubes for a duration of 2 to 3 days, increasing the psychological and physical burden on the patients. Second, the median length of hospital stay was shorter for patients in the subcostal approach group than median sternotomy group. The patients in the subcostal approach group were typically discharged within 2 to 3 days after surgery. Since 2021, our center has implemented the subcostal approach as a day surgery procedure, further reducing the average length of hospital stay. By contrast, the patients undergoing routine median sternotomy in this study had an average hospital stay of approximately 5 days. Third, the postoperative pain in patients who have undergone thoracic surgery primarily originates from compression and injury of intercostal nerves by surgical instruments and stimulation of chest wall nerves by thoracic drainage tubes. 15 The subcoastal approach avoids direct entry into the thoracic cavity through the intercostal spaces and does not require the placement of drainage tubes, thus significantly reducing the stimulation of intercostal and chest wall nerves. Therefore, compared with the median sternotomy group, the patients in the subcoastal approach group experienced less severe postoperative pain during both the acute pain period (1–3 days postoperatively) and the chronic pain period (30 days postoperatively). Fourth, the subcoastal approach utilizes a smaller surgical incision than the approximately 20-cm incision used for median sternotomy. The 2- to 4-cm incision in the subcoastal approach group healed more rapidly and provided better cosmetic outcomes in this study. Furthermore, the subcoastal approach is not inferior to median sternotomy in terms of the completeness of tumor resection and the diagnosis and treatment of diseases. It is particularly effective in relieving symptoms in patients with severe myasthenia gravis complicated by thymoma. Additionally, the incidence of complications in the subcoastal approach group was lower than that in the median sternotomy group. Finally, based on the aforementioned points, the subcostal approach combines the numerous advantages of median sternotomy. Consequently, the patient satisfaction rating in the subcoastal approach group was significantly higher than that in the median sternotomy group.
Cosmetic incisions, a shorter hospital stay, reduced postoperative pain, and faster recovery all contribute to an improved surgical experience for patients. The results of our study align with previous research in the field of thymoma surgery. The higher clinical efficacy and patient satisfaction associated with the subcostal thoracoscopy approach in our study is consistent with the findings of other studies, thus adding evidence regarding the advantages of this surgery.16,17
Notably, the subcoastal approach is similar to median sternotomy in terms of lymph node dissection. Lymph node metastasis has received increasing attention in patients with thymoma, a potentially malignant tumor. According to the United States Surveillance, Epidemiology, and End Results database, 18 the lymph node metastasis rate in 442 patients with thymoma was 13.3%, whereas it was 33.5% in 176 patients with thymic carcinoma and as high as 62.3% in 53 patients with neuroendocrine tumors. Patients with lymph node metastasis have a significantly worse prognosis. According to the Japanese Association of Chest Surgery database, 19 the lymph node metastasis rates in 1320 patients with thymic tumors were as follows: 1.8% for thymoma, 27.0% for thymic carcinoma, and as high as 28.0% for thymic carcinoid. Based on the Chinese Alliance for Research in Thymomas database, 20 a prospective multicenter clinical study showed that among 275 patients with thymic tumors, the rates of lymph node metastasis were 2.1% for thymoma, 25.0% for thymic carcinoma, and 50.0% for neuroendocrine tumors. One study even indicated that because of dissection of the anterior mediastinal lymph nodes and sampling of the deep mediastinal lymph nodes, approximately 80% of patients with thymic tumors experienced an increase in clinical staging, with 23.2% of patients with preoperative stage I or II tumors progressing to stage IV and 57.1% of patients with preoperative stage III tumors progressing to stage IV. 21 In summary, lymph node metastasis is a clinically significant issue, and it is associated with histological type and local disease progression. The correlation between lymph node metastasis and tumor size is weak. Moreover, increasing numbers of studies are supporting the better prognosis of lymph node-negative patients.22,23 The fundamental goal of surgery is to prolong the patient’s survival time. When surgical outcomes are not compromised, the subcostal approach can achieve lymph node dissection of the same quality as median sternotomy.
Our study had several limitations. Selection bias may have been present in the choice of the surgical approach for each patient. Our study population comprised patients from a single center, and the decision regarding the surgical approach was based on various clinical factors, which may have introduced bias. We would like to include a broader population in future studies with longer follow-up times to validate our data. Conducting multicenter studies involving diverse patient populations and surgical teams would also enhance the generalizability and external validity of our findings. Collaborative efforts across institutions could provide a broader perspective on the comparative outcomes.
Conclusion
The subcostal approach in thoracoscopic surgery for thymoma is not inferior to the conventional median sternotomy approach. Moreover, this surgical technique offers smaller incisions, faster patient recovery, and lower economic costs. Therefore, it is a surgical approach that deserves to be promoted and widely adopted.
Footnotes
Acknowledgements
The authors thank all patients for their participation.
Authors’ contributions
Sha Xue and Shaoyi Chent designed the study. Yongan Zhou and Zhengwei Zhao collected the data. Yize Guo and Zheng Feng performed the data analysis. Xunliang Yin and Dongliang Yuan performed the data analysis and wrote the manuscript.
Data availability statement
The data used in this research can be obtained upon request.
Declaration of competing interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Key Research and Development Programs of Shanxi Province (2017ZDXM-SF-052).
