Abstract
Lymphoepithelioma-like intrahepatic cholangiocarcinoma (LEL-ICC) is a rare distinctive variant of liver cancer with unique epidemiological and pathological characteristics, including dense lymphocyte infiltration. We herein describe a 67-year-old Chinese man with LEL-ICC. The patient had undergone endoscopic extraction of a bile duct stone 1 month prior. Contrast-enhanced abdominal computed tomography (CT) revealed a 2.5- × 2.5- × 1.5-cm low-density mass located in a covert part of the left lateral segment of the liver. Contrast-enhanced magnetic resonance imaging revealed a hyperintense lesion on T2-weighted and diffusion-weighted images of the left lateral liver, with similar size and signal characteristics in the arterial and portal venous phases. The patient subsequently underwent left lateral laparoscopic hepatectomy. The results of postoperative pathology and immunohistochemistry allowed for the definitive diagnosis. In situ hybridization using an Epstein–Barr virus-encoded RNA probe revealed extensive reactivity in the tumor cell nuclei, supporting a diagnosis of LEL-ICC. The patient was recurrence-free at 12 months postoperatively as shown by CT. A literature review indicated that in middle-aged patients with Epstein–Barr virus infection, a liver mass with a well-defined margin and a combination of hypervascularity and delayed intratumoral enhancement on CT and magnetic resonance imaging may suggest a diagnosis of LEL-ICC.
Keywords
Introduction
Lymphoepithelioma-like carcinoma (LELC) is a rare malignant tumor that was first found in the nasopharynx in 1982. 1 It is composed of undifferentiated epithelial cells and marked lymphoid infiltration.1,2 LELC in the liver is unusual and can be divided into two types: lymphoepithelioma-like intrahepatic cholangiocarcinoma (LEL-ICC) and lymphoepithelioma-like hepatocellular carcinoma (LEL-HCC). The occurrence of liver LELC might originate from a distinctive immune response in the context of liver tumors, and in most cases, 3 Epstein–Barr virus (EBV) infection has been regarded as playing an essential role in the carcinogenesis of LELC. Because of the lack of specific manifestations regarding its imaging findings and laboratory test results, LELC is difficult to diagnose without postoperative histopathologic and immunohistochemical examinations. To our knowledge, fewer than 150 cases of liver LELC have been reported to date, including both LEL-ICC and LEL-HCC, and the understanding of LELC is quite limited. Therefore, more reports are needed to elucidate the comprehensive characteristics of liver LELC. We herein describe a 67-year-old man with LEL-ICC complicated by EBV infection who underwent laparoscopic left lateral hepatectomy. We present the following case in accordance with the CARE reporting checklist. 4
Case presentation
A 67-year-old Chinese man presented to our hospital’s outpatient clinic with abdominal pain. The patient was diagnosed with intrahepatic bile duct stones by computed tomography (CT). His medical, family, and psychosocial history were unremarkable. He had undergone biliary stone removal under endoscopic retrograde cholangiopancreatography 1 month prior. Unfortunately, a tumor located in the left liver as shown by CT imaging was missed. However, a small hepatic mass was discovered on magnetic resonance imaging (MRI), and the patient was referred to the department of hepatobiliary surgery for further evaluation. Laboratory investigations revealed a normal total bilirubin concentration (0.6 mg/dL), serum albumin concentration (3.9 g/dL), and international normalized ratio (1.3). No tumor markers were elevated, including carcinoembryonic antigen, cancer antigen 125, carbohydrate antigen 19-9, and α-fetoprotein. Viral serology markers were negative, and liver function test results were normal. According to the Barcelona Clinic Liver Cancer staging system, the patient was determined to have very early stage cancer, and curative surgical resection of the tumor with negative surgical margins was carried out at our center. 5
The CT scan displayed an approximately 2.6- × 1.7-cm slightly low-density mass with a blurred boundary and intratumoral punctate calcification (Figure 1(a)). The tumor protruded beyond the contour of the liver. The mass demonstrated partial signal enhancement in the arterial phase and further enhancement in the portal venous phase (Figure 1(b), (c)). T2-weighted and diffusion-weighted MRI showed high-signal-intensity foci in the left lateral liver (Figure 2(a), (b)). Like CT, MRI also revealed a homologous signal characteristic in the arterial and portal venous phases (Figure 2(c)–(e)). The tumor was more clearly displayed in the coronal image, which showed that it protruded from the liver boundary (Figure 2(f)). The patient underwent laparoscopic left lateral hepatectomy under the assumption of hepatic malignancy. The duration of the operation was 1 hour, and the intraoperative blood loss was about 5 mL. The patient recovered smoothly and was discharged on postoperative day 8. No evidence of recurrence was found during the routine follow-up examinations at 1, 3, and 6 months postoperatively, and the patient’s liver function remained normal.

Appearance of tumor on computed tomography. CT image demonstrated a slightly low-density lesion and punctate calcification within the tumor in the lateral segment of the left liver (a, white triangle). Contrast-enhanced CT revealed a relatively vague margin mass, 2.6 cm in diameter. The tumor showed an enhancement in the arterial phase (b) and progressive enhancement in the late phase (c).
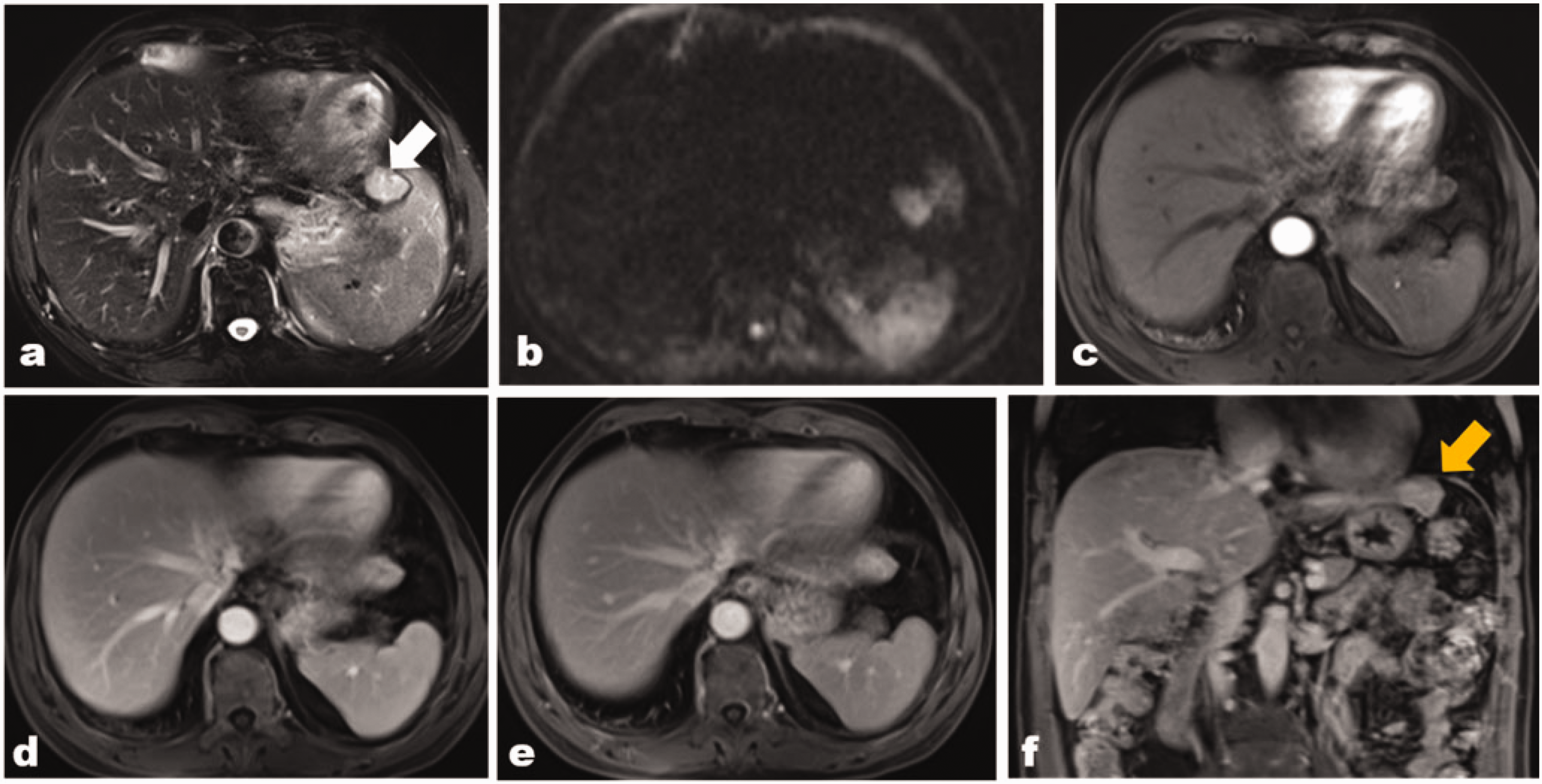
Magnetic resonance imaging demonstrated a 2.7- × 2.1- × 2.0-cm lesion in liver segment II. The tumor appeared hyperintense on (a) the T2-weighted image (white triangle) and (b) the diffusion-weighted image. (c) In the arterial phase after administration of gadopentetate dimeglumine, the tumor showed homogeneous marked enhancement. (d), (e) On contrast-enhanced magnetic resonance imaging obtained in the delayed phase, the tumor showed progressive enhancement and (f) The tumor shape was comprehensively observed in the coronal image (yellow triangle)
Gross examination of the resected specimen showed gray foci measuring 2.5 × 2.5 × 1.5 cm in the resected segment. Microscopically, irregular small bile duct atypical hyperplasia was seen on a background of highly proliferative lymphoid tissue and fibrosis, with flake distribution in part areas (Figure 3(a), (b)). EBV-encoded RNA in situ hybridization was performed, and the tumor tissues were found to be positive (Figure 3(c)). Immunohistochemical examination revealed positivity for Ki-67 (40%), CK (pan), CK19, CD20, CD3, and Pax-5 and negativity for CD10 and p63. The tumor-free surgical margin was at least 10 mm. All features fulfilled the criteria for a diagnosis of LEL-ICC.
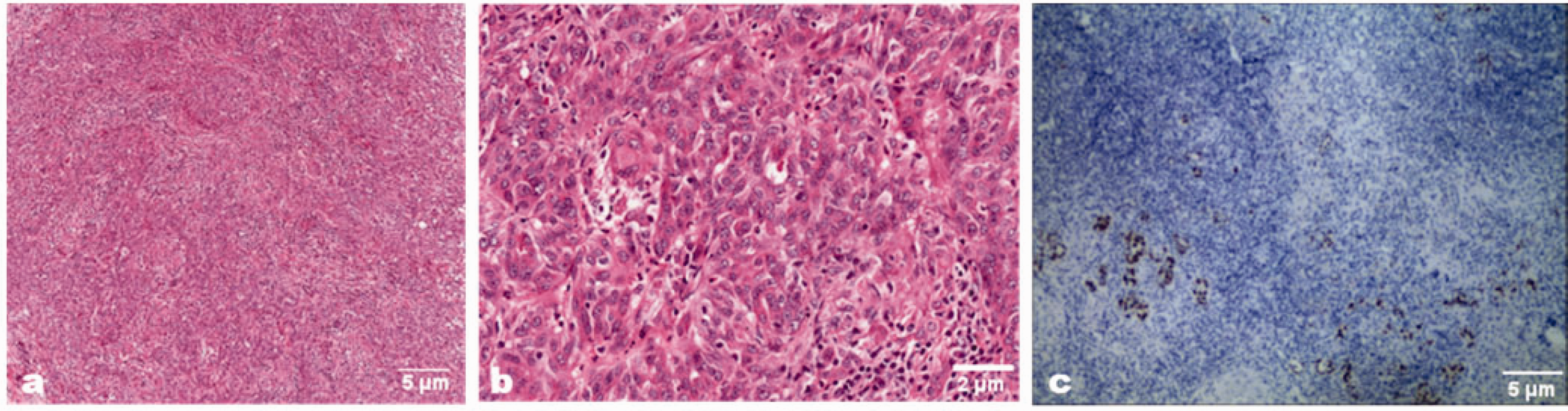
Pathological and Epstein–Barr virus-encoded RNA (EBER) in situ hybridization examinations after surgery. Microscopically, the larger lesion was composed of undifferentiated epithelial cells with vesicular nuclei, prominent nucleoli, and significant lymphocytic infiltration. The tumor tissues were positive for EBER. (a) HE ×40. (b) HE ×100. (c) EBER in situ hybridization, ×40. HE, hematoxylin and eosin
Discussion
We have herein described a rare case of LEL-ICC associated with EBV infection in a Chinese man of advanced age. The radiologist missed the mass in the left liver on the patient's initial CT scan; this oversight occurred because sagittal and coronal image reconstructions of the left liver were not included in the CT scan. However, we fortunately found the tumor by MRI examination as it included coronal scans. The patient underwent laparoscopic left lateral hepatectomy, and histopathologic and immunohistochemical examinations confirmed LEL-ICC.
LELC is morphologically characterized by the proliferation of undifferentiated epithelial cells with intense lymphocytic infiltration. These tumors have been reported to arise in various sites such as the nasopharynx, stomach, salivary glands, lungs, and thymus. 6 The first case of LEL-ICC was reported by Hsu et al. 7 in 1996. The tumor occurred in an Asian woman with a liver mass who underwent left lobectomy, and pathologic examination revealed two different tumor components: well-differentiated adenocarcinoma and LELC. 7 To the best of our knowledge, 24 cases of LEL-ICC with a description of the radiological characteristics have been reported during the last decade, as shown in Table 1.8–19 Most of the case reports came from Asian countries. The mean age of the patients was 49 years (range, 22–77 years), with 70.8% (17/24) being female and 83.3% (20/24) being Asian. The mean size of the LEL-ICCs was 4.8 cm (range, 1.0–32.5 cm).
Reported cases of liver LEL-ICC from 1 January 2012 to 27 April 2022

F, female; M, male; +, positive;
Like nasopharyngeal carcinomas, most LEL-ICCs are speculated to be related to EBV infection. 9 Among the 24 cases reported to date, 21 (87.5%) patients were EBV-positive. This finding indicates that EBV might play a crucial role in the tumorigenesis of LEL-ICC, and this role may involve the immune response caused by EBV infection. Furthermore, involvement of the EBV genome in LELC depends on the origin of the LELC. Recent studies have suggested that EBV-positive LEL-ICC with a significantly increased density and proportion of CD8+ T cells and CD20+ B cells was significantly associated with longer 2-year overall survival and recurrence-free survival than EBV-negative LEL-ICC. Both programmed cell death 1 (PD-1) and programmed cell death ligand 1 (PD-L1) in tumor-infiltrating lymphocytes and PD-L1 in tumor cells were overexpressed in EBV-positive LEL-ICC. 20 However, Adachi et al. 21 found no evidence of histopathologic differences between EBV-negative and EBV-positive LEL-ICC. Therefore, because of the rarity of LEL-ICC, the role of EBV in the tumorigenesis of LELC remains controversial and poorly understood.
Histologically, LELCs either contain two components (LELC and more typical well-differentiated adenocarcinoma) or consist solely of LELC. 11 The LELC component, which is by definition poorly differentiated (grade 3), is composed of neoplastic cells arranged in sheets, cords, or poorly formed glands surrounded and infiltrated by a distinctive dense lymphoid population. 2 The World Health Organization acknowledges LEL-ICC as a variant of ICC, but it does not further define the diagnostic conditions for the density of the lymphocytic infiltrate. About immunohistochemical indexes, tumor cells express epithelial membrane antigen and pan keratins (e.g., AE1/AE3), the bile lineage markers epithelial cell adhesion molecule and carbohydrate antigen 19-9, and bile type keratins K19 and K7. 2 The tumor-infiltrating lymphocytes are predominantly CD3-positive T cells, with focal CD138-positive plasma cells and CD20-positive B cells. 22 More than half of tumors demonstrate mutation-related p53 immunohistochemical patterns. 22
Distinguishing LEL-ICC from LEL-HCC is similar to the differential diagnosis between ICC and HCC. CK19 and CK7 are specific immunohistochemical factors for the diagnosis of cholangiocellular carcinoma, while α-fetoprotein and glypican-3 proteins are specific immunohistochemical factors for the diagnosis of HCC. In most cases, the pathological diagnosis depends on these particular immunohistochemical factors.
Radiologically, our patient showed an ovoid mass with hypervascular arterial enhancement and delayed central enhancement. Yang et al. 17 found that the tumor size tended to be related to the morphologic pattern and texture uniformity: smaller tumors (<5 cm) were round or ovoid with a smooth or slightly lobulated margin, and larger tumors (≥5 cm) showed an irregular shape with a lobulated margin. 17 We also reviewed the imaging features of 24 cases of LEL-ICC reported in the literature (Table 1). Among these 24 cases, 41.6% (10/24) lesions showed marked enhancement in the arterial phase.12,13,17–19 Two tumors of <3 cm were reported in the literature, showing delayed central scarring or patchy enhancement. 8 Central delayed hyperenhancement might be associated with the amount of fibrous stroma, which is more commonly found in ICC. The tumor in our case was nonencapsulated. In the literature, three cases showed similar delayed pseudocapsule enhancement on contrast-enhanced ultrasound and contrast-enhanced CT 10 and enhanced MRI.8,14 The pseudocapsule may be similar to the histologic fibrous capsule in terms of tumor invasiveness; therefore, a fibrous capsule or pseudocapsule in LEL-ICC has been considered a favorable prognostic factor. 23
The prognosis of LEL-ICC is reportedly better than that of classical ICC, with a 5-year survival rate of 100% and 13.2%, respectively. 24 There is no consensus on a standardized treatment strategy for LEL-ICC, and surgical resection remains the treatment of choice. However, chemotherapy, radiotherapy, or targeted therapy have occasionally be adopted. Nogami et al. 18 described a patient with early-stage LEL-ICC who was treated by radiofrequency ablation and achieved radical cure. Lee 22 described a patient with lymph node metastasis who underwent surgery and postoperative radiation and was still alive without recurrence at 54 months postoperatively. Huang et al. 20 suggested that patients with EBV-positive LEL-ICC would benefit from anti-PD-1/PD-L1 therapy. With respect to treatment, surgical resection should be considered first. LEL-CC is a rare variant of ICC characterized by a better outcome than that of classical ICC. For patients with postoperative recurrence and metastasis of LEL-ICC, surgical resection remains an effective approach.
Conclusion
LEL-ICC often occurs in middle-aged Asian patients with EBV infection, and abnormal tumor marker levels are uncommon. Multiplanar reconstruction should be added to the imaging process to reduce the chance of missing such lesions. LEL-ICC is characterized by a well-defined margin, is regularly found in the left liver, and exhibits enhancement characteristics of hypervascularity, washout, and delayed intratumoral or fibrous septum enhancement. These characteristics help to confirm the diagnosis of LEL-ICC. Surgical resection is the most effective therapy for early-stage carcinoma. Nevertheless, preoperative imaging identification of LEL-ICC remains problematic for surgeons and radiologists because of the rarity of this tumor. More case studies are needed to gain a better understanding of LEL-ICC.
Ethics statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). The study was approved by the Academic Ethics Committee of Shaoxing People’s Hospital. Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Reporting checklist
The authors have completed the CARE Checklist for this case report.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Supplemental Material
sj-mp4-1-imr-10.1177_03000605231210174 - Supplemental material for Potential missed opportunities for diagnosis of lymphoepithelioma-like intrahepatic cholangiocarcinoma: report of a rare case
Supplemental material, sj-mp4-1-imr-10.1177_03000605231210174 for Potential missed opportunities for diagnosis of lymphoepithelioma-like intrahepatic cholangiocarcinoma: report of a rare case by Wei Tang, Ya’nan Huang, Shanlu Yu, Ting Wang, Jianfeng Yang, Zhenhua Zhao in Journal of International Medical Research
Footnotes
Consent
The patient provided written informed consent for both treatment and publication of this report.
Acknowledgement
The authors would like to thank the patient for his cooperation and consent to publication of this report.
Author contributions
WT and YH contributed to the data acquisition and analysis and the writing of the first draft. ZZ contributed to the conception of the work and revised the paper. SY provided support with pathological knowledge. TW contributed to collection of the data and communicated with the patient’s family. JY contributed to the search for information regarding the patient’s disease. SY assisted with the pathological examination.
Declaration of conflicting interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This work was supported by the Zhejiang Medical and Health Platform Program (2021KY371), Key Laboratory of Functional Molecular Imaging of Tumor and Interventional Diagnosis and Treatment of Shaoxing City.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
