Abstract
Solitary fibrous tumors (SFTs) are commonly seen in the pleura. SFT involvement of the vulva is rare, and clinical diagnosis is mainly based on histopathological and immunohistochemical analyses. We herein describe the imaging features of a 69-year-old woman with an SFT of the vulva. The SFT was hypointense on T1-weighted images, similar to muscle; however, it showed inhomogeneous hyperintensity predominantly on fat-suppressed T2-weighted images. An area of low signal intensity was evident on T2-weighted images, and the tumor showed progressive enhancement in delayed phases. The tumor also displayed heterogeneous and prolonged, persistent enhancement, and serpentine vessels were present in the peritumoral area as signal voids. Pathological examination confirmed that the lesion was an atypical SFT originating from the vulva, and it was composed of spindle cells and perivascular and stromal hyalinization. This case reveals the characteristic imaging findings of vulvar SFT and their association with the relevant pathological findings, thus contributing to the primary diagnosis and preoperative evaluation of this potentially aggressive tumor.
Keywords
Introduction
Solitary fibrous tumors (SFTs) originate from mesenchymal neoplasms and account for ≤2% of all soft tissue tumors with undefined biological behavior. 1 SFTs usually occur in the pleura. The clinical symptoms of extrapleural SFTs vary according to their anatomic site, such as the urogenital system,2,3 mediastinal space, 4 orbit, 5 thyroid, 6 nasopharyngeal region, 7 pancreas, larynx, or salivary glands. The occurrence of an SFT in the female genital system, especially in the vulva, is uncommon. Pertinent and comprehensive reports and articles pertaining to vulvar SFT during the last two decades were reviewed, showing that no more than 26 cases of vulvar SFT have been reported. To the best of our knowledge, SFT is recognized as a tumor of low malignancy, and most reported cases focused more on its morphological and immunophenotypic features than on its imaging manifestations. However, imaging can offer visual information about its potential for recurrence or metastasis as well as the relationship of nearby organs in the asymptomatic stage of disease. Nevertheless, complete imaging examinations of vulvar SFT are still lacking. 8 We herein describe a patient who received relatively comprehensive imaging examinations and in whom immunohistochemical and pathological analyses confirmed an SFT of the vulva.
We present the following case in accordance with the CARE reporting checklist (available at https://www.equator-network.org/). 9
Case Description
A 69-year-old woman was referred to our hospital with a 1-year history of a palpable mass on the right side of her vulva. The lesion had shown no obvious increase in size during the past year. A suspicious echoic mass arising from the vulva had previously been detected by ultrasound during a routine examination. On admission, the patient’s routine laboratory test results and tumor marker levels were normal. Physical examination of the vulva revealed that the right labium majus adjacent to the urethral opening was hypertrophied. The patient’s family reported no similar symptoms in this site and no malignancies. A cystic lesion measuring about 4 × 4 cm was palpated with normal skin temperature, no tenderness or redness, and no fluctuation. The patient showed no evidence of a paraneoplastic syndrome such as hypertrophic pulmonary osteoarthropathy or hypoglycemia.
Unenhanced computed tomography revealed an approximately 38- × 65-mm well-circumscribed tumor isodense to mildly hypodense to muscle below the groin (26 Hounsfield units) without internal calcification. Neoplastic invasion was not found in adjacent organs, bone, or regional lymph nodes, but enlarged lymph nodes were seen in the bilateral inguinal regions (Figure 1(a)). The patient subsequently underwent magnetic resonance imaging for further diagnosis. This examination revealed overall hypointensity of the mass on T1-weighted fast spin echo imaging (Figure 1(b)) with intratumoral flow voids. Axial fat-suppressed T2-weighted images demonstrated a mass with a predominantly hyperintense signal with a small focal area that displayed low signal intensity (Figure 1(c)). The mass predominantly showed heterogeneous hyperintensity on diffusion-weighted imaging (b value of 700 mm/s2) (Figure 1(d)) and hypointensity on apparent diffusion coefficient mapping (Figure 1(e)). Furthermore, the arterial (Figure 1(f)), venous (Figure 1(g)), and delayed phases (Figure 1(h)) of the tumor showed heterogeneous enhancement and prolonged persistent enhancement. Likewise, the local low-signal intensity within the mass was evident, and hypoenhancement was present in the portal venous and delayed phases. An incomplete capsule-like rim and peripheral tortuous small vessels were seen in the mass after contrast administration. The flow voids on the thick slices of the T1-enhanced sequence were more obvious (Figure 1(i)).
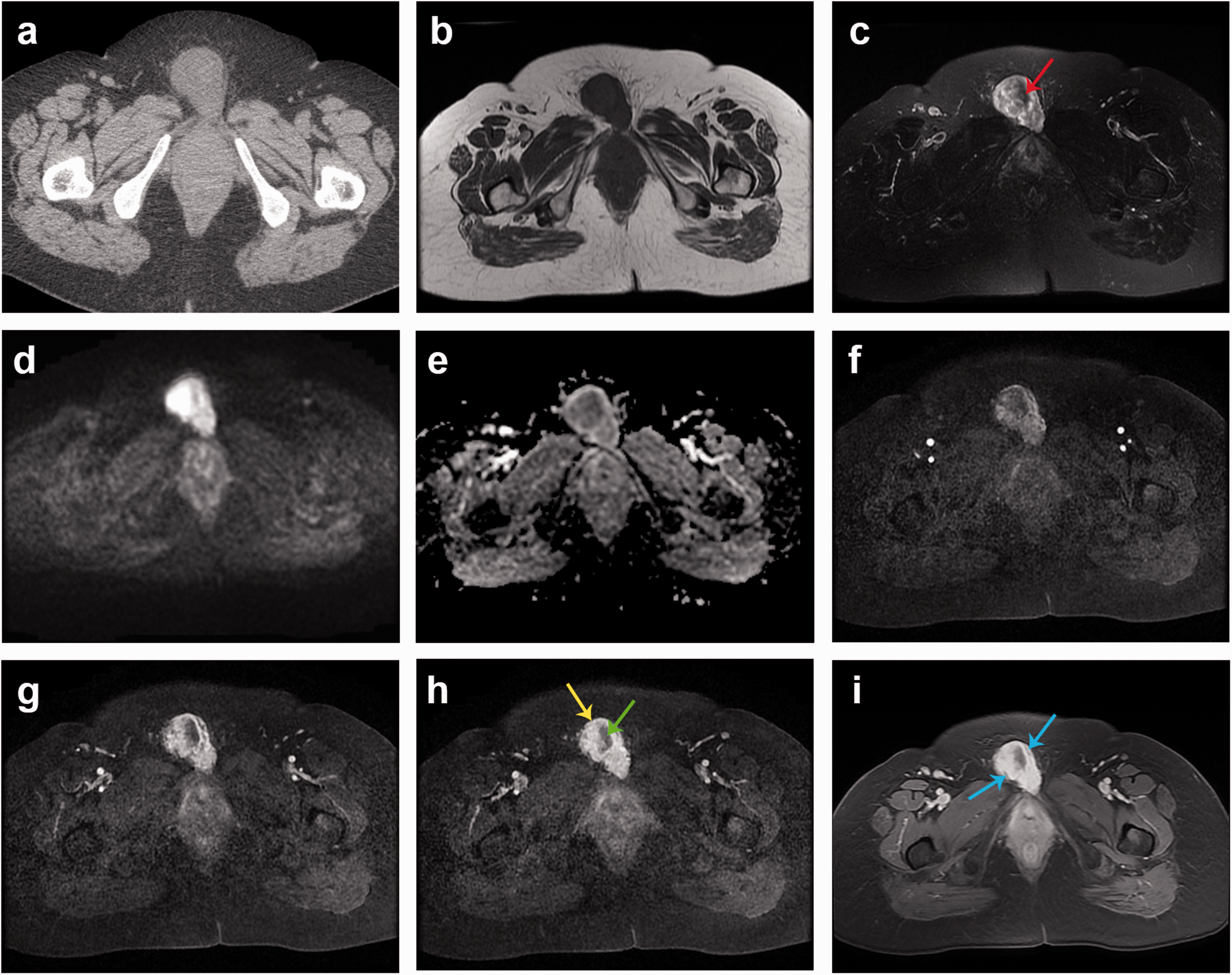
Imaging findings of a 69-year-old woman who presented with an asymptomatic mass arising from her right labium majus. (a) A well-circumscribed isomass was detected by pelvic computed tomography without intratumoral necrosis or calcification. The tumor showed (b) hypointensity on preoperative axial T1-weighted images and (c) heterogeneous hyperintensity on fat-suppressed T2-weighted images. No neighboring tissues or organs were invaded. An intratumoral hypointense region was observed on T2-weighted images (red arrow). The tumor showed (d) heterogeneous high signal intensities on the diffusion-weighted imaging map (b value of 700 mm/s2) and (e) inhomogeneous hyperintensity on the apparent diffusion coefficient map. The mass showed avidly persistent inhomogeneous enhancement in the (f) arterial, (g) portal venous, and (h) delayed phases. (h) The peripheral capsule-like rim showed obvious delayed enhancement (yellow arrow), and the hypointensity was most likely correlated with spindle cells as well as dense collagen, which exhibited prolonged persistent centripetal enhancement in the delayed phase (green arrow) and (i) Consecutive serpentine arteries, depicted as a flow void, were revealed on the thick slice of enhanced T1-weighted images along the peritumoral areas (blue arrow).
The patient underwent complete surgical resection of the tumor. The macroscopic specimen was a gray-white mass with a partially incomplete fibrous capsule (8.0 ×7.0 × 3.5 cm). The cut surface of the excised vulva lesion revealed focal hemorrhage but no necrosis (Figure 2(a), (b)). Further histopathologic examination of the excised tumor revealed patterns consistent with an SFT. The pathological findings suggested that it was a spindle cell tumor; the arrangement was relatively loose and disordered, and irregular vessel dilation as well as a hyalinized matrix were evident in the perivascular space (Figure 2(c)). Mitotic figures were rare (1 mitosis per 10 high-power fields), and few residual tumor cells were present at the periphery of the tissue section. Immunohistochemical analysis revealed that the tumor cells were strongly positive for CD34 (+) (Figure 2(d)), BCL-2 protein (+) (Figure 2(e)), STAT6 (+) (Figure 2(f)), CD99 (+), vimentin (+), and Ki-67 (2%+) but negative for S-100, epithelial membrane antigen, and smooth muscle actin. After surgical excision, no further adjuvant treatment was administered. Neither local recurrence nor progression was found throughout 1 year of regular radiological and clinical surveillance.
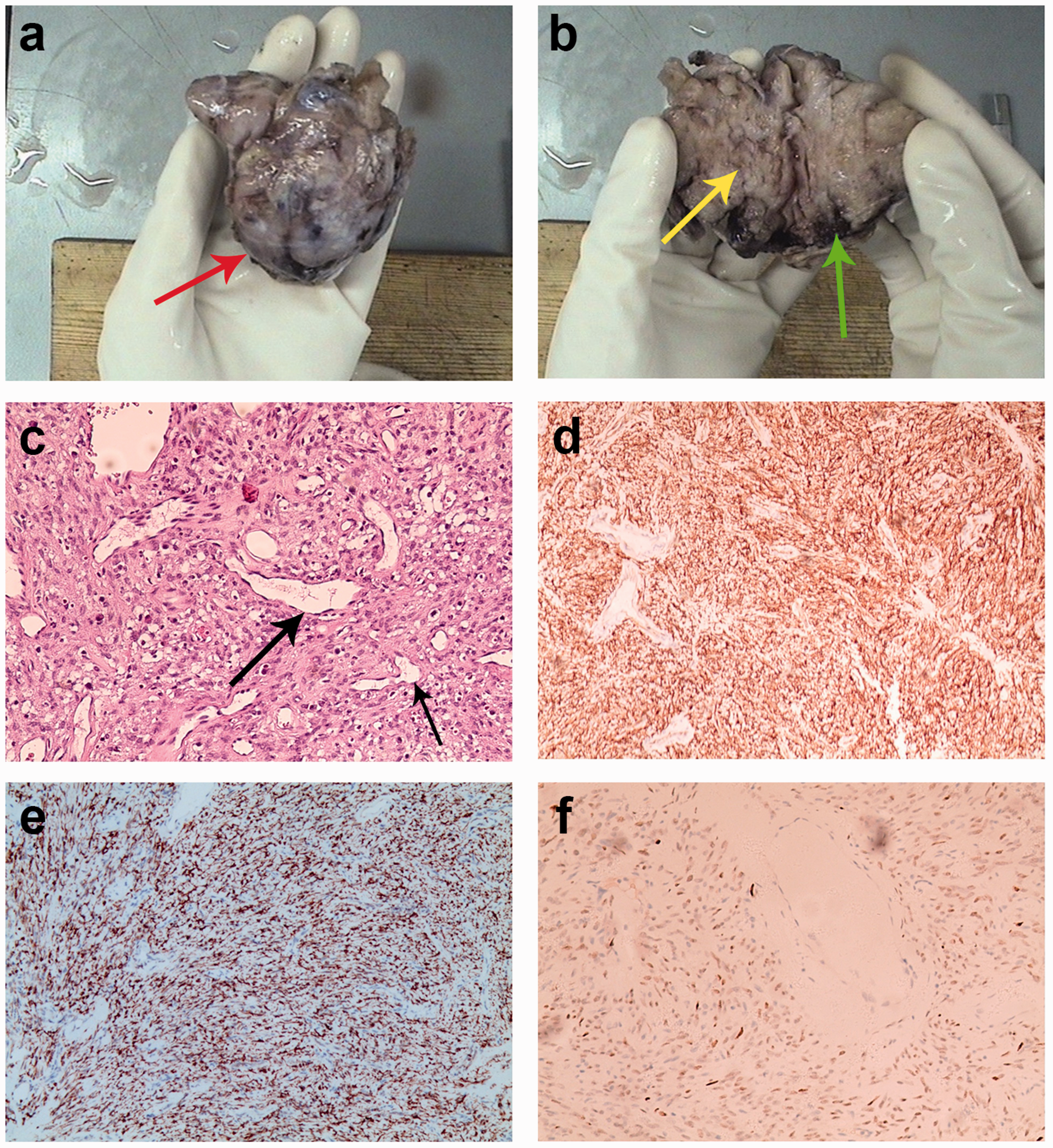
Postoperative examination findings. (a) Photograph of the gross specimen showing a well-circumscribed, firm, 8.0- × 7.0- × 3.5-cm mass with a capsule (red arrow). (b) The cut surface of the excised vulvar tumor was grayish-white and firm, and fibrous compartments were observed (yellow arrow). No obvious necrosis was present, but focal hemorrhage was seen (green arrow). (c) Pathological and immunohistochemical features of the solitary fibrous tumor were examined. Hematoxylin and eosin staining revealed a patternless architecture of spindle cell proliferation with irregular vessel dilation, staghorn-like vessels (black arrows), and subtle peripheral hyaline degeneration (magnification, ×100). Immunohistochemical staining revealed that the tumor was positive for (d) CD34 (magnification, ×40), (e) BCL-2 (magnification, ×40) and (f) STAT6 (magnification, ×40).
Discussion
SFTs were first described by Klemperer and Rabin in 1931, 10 and they mostly originate from pleural or serosal tumors. Although approximately 60% of SFTs may occur in almost any anatomic site, 11 those arising from the vulva as gynecological SFTs are particularly rare. 12 The age of our patient is consistent with the age of patients with SFTs described in previous reports (average age of 14–78 years).13,14 According to the 2020 World Health Organization classification of soft tissue tumors, SFTs are categorized as intermediate (rarely metastasizing) fibroblastic or myofibroblastic tumors, indicating that most exhibit a benign clinical course with no symptoms unless they compress adjacent anatomical structures.15,16 Nevertheless, SFTs may still metastasize. Although the new (2021) FIGO staging system is applicable to most malignancies arising from the vulva, the current literature indicates that primary soft tissue tumors or sarcomas of the vulva should follow the TNM classification of soft tissue tumors.17–19
Although an extrapleural SFT clinically presents as a slowly growing mass with no sex predominance, closer surveillance is still necessary for those exceeding 10 cm in size. Approximately 10% to 30% of patients with SFTs develop hypertrophic pulmonary osteoarthropathy, paraneoplastic syndrome with inflammatory diffuse arthralgia, and Doege–Potter syndrome related to hypoglycemia20,21 because of excessive serum insulin-like growth factor II. The manifestation of Doege–Potter syndrome is associated with approximately 4% of benign and malignant forms of SFTs. Nevertheless, our patient did not develop systemic symptoms. Furthermore, the serum concentration of squamous cell carcinoma antigen is recognized as a potential tumor marker to distinguish vulvar mesenchymal from epithelial lesions. 22
The SFT in our patient predominantly exhibited soft tissue attenuation with well-defined margins on a plain computed tomography scan. According to the published literature, SFTs typically show avidly heterogeneous geographic enhancement features caused by hemorrhage, necrosis, calcification, or cystic degeneration; however, these obvious pathological changes were absent in our case. 11
The signal intensity of the mass in our case was isointense to muscle on T1-weighted images. However, the mass showed inhomogeneous hyperintensity on fat-suppressed T2-weighted images, and the intratumoral area locally presented low signal intensity. This is consistent with a report by Kim et al., 23 who stated that low intensity on T2-weighted images was a characteristic feature of SFTs. Another previous study showed that the signal intensity on T2-weighted images was variable, likely because of the myxoid to hyalinized stromal content. 24 Some authors have also indicated that the presence of areas of low and high signal intensity on T2-weighted images is the characteristic patchy or “ying-yang” appearance of SFT.25,26 Moreover, the region of T2 hypointensity we observed in the present case was considered to be associated with dense collagen, a small number of mobile protons, and fibrosis of the mass. One study directly proved that the strongly hypointense areas on T2-weighted images were hypocellular regions characterized by disorganized spindle cells and thick bands of collagen. 27
The presence of degeneration might also impact the imaging of SFTs. The intratumoral vessels in our case were displayed as the absence of signal (flow voids) on the enhanced T1-weighted axial images, which indicated that vessels had developed along with the mass. Interestingly, our case is consistent with reports by Wang et al. 28 and Seo et al., 29 who stated that intratumoral flow voids on magnetic resonance images are a typical characteristic of SFTs. Most SFTs show serpentine flow voids, which are considered useful adjuncts in the differential diagnosis.28,29 Furthermore, the mass compressed adjacent anatomic structures, and the enhancement was progressively intense and heterogenous. The presence of an intratumoral area of low attenuation is correlated with the paucicellular fibrous components, which accumulate the contrast agent and display a persistent slightly centripetal enhancement pattern on delayed-phase images. We noticed that the capsule was hypointense along the periphery of the tumor on T1- and T2-weighted images. It subsequently displayed intense enhancement in the delayed phase, but this seemed relatively incomplete; this finding was compatible with the microscopic findings of the excised encapsulated vulvar mass.
The recommended criteria for predicting local recurrence or distant metastasis of malignant SFTs are a large tumor size (>10 cm), infiltrative margins, high cellularity, nuclear pleomorphism, regions of necrosis, and an increased mitotic index (>4 mitoses per 10 high-power fields).20,30 However, our patient’s tumor was <10 cm in size and manifested as a slowly growing, asymptomatic mass without infiltration. In addition, the pathology report revealed a patternless distribution of proliferating spindle cells, perivascular stromal hyalinization, and prominent dilated and staghorn-shaped vascular vessels; this was consistent with the typical pathological findings of SFT and the high vascularity of lesions on imaging. However, the behavior of SFTs is unpredictable. Extrapleural SFTs are usually regarded as distinctive lesions that show more aggressive behaviors, whereas the primary vulvar SFT in the present case showed no local infiltration. 31 The risk of local recurrence and metastasis is correlated with the surgical resection margins, and recurrence or metastasis is estimated to occur in 10% to 20% of cases. Pearre et al. 32 reported a case of an aggressive vulvar fibrous tumor that resulted in extensive spinal metastatic disease. Some authors assume that the absence of invasion of regional structures by SFTs probably corresponds to the submesothelial origin of these tumors and their overlying intact mesothelial lining,33,34 but some studies have suggested that the relationship between morphology and outcome is not always explicit. Some morphologically benign tumors behave aggressively. Therefore, long-term follow-up is necessary after complete excision of extrapleural SFTs.
Although complete surgical resection with margins of at least 1 to 2 cm is the primary treatment modality for SFTs, the clinical course of these tumors is unpredictable. 35 The role of adjuvant radiation is not well established. Some research has shown that the combination of surgery and radiation therapy leads to excellent local control. 36 However, the use of radiation is extrapolated from small reports of improved local control in the adjuvant or recurrent settings for both pleural and extrapleural SFTs. Moreover, traditional cytotoxic chemotherapy seems ineffective for the majority of patients with tumors that do metastasize. NAB2-STAT6 oncogenic fusion is reportedly related to the overexpression of vascular antiangiogenic drugs, and the identification of preferentially expressed antigen in melanoma (PRAME) is regarded as a possible new therapeutic target for clinical management. The promising results obtained so far warrant additional investigations.37,38 Notably, the treatment of vulvar SFT still carries a risk of intraoperative blood loss and postoperative complications including surgical site infection and anal dysfunction 39 ; however, our patient did not develop these complications.
Typically, a suspected diagnosis of SFT based on the histopathological pattern is confirmed by the demonstration of CD34, BCL-2, and STAT6 reactivity during immunohistochemical examination. In our case, the immunohistochemical profile was consistent with the above descriptions. Furthermore, STAT6 has been proven to be a sensitive and specific marker in the diagnosis of difficult cases of SFT 40 and is helpful to distinguish similar tumor categories.
In general, the prognosis for patients who have undergone complete surgical excision of an SFT is excellent. Our patient developed no local recurrence throughout 1 year of surveillance, but close long-term follow-up is still needed in case of worsening. The differential diagnoses of SFTs arising in the vulva include any mesenchymal lesions that might occur at this site, such as cellular angiofibroma, superficial or deep angiomyxoma, angiomyofibroblastoma, mammary-type myofibroblastoma, aggressive angiomyxoma, and neurilemmoma. 41 Therefore, based on the preoperative radiological assessment, it is essential to perform complete excision followed by postoperative histopathological and immunohistochemical examinations to establish the correct diagnosis.
Conclusion
A vulvar SFT is an exceedingly rare tumor and usually behaves indolently, but it has the potential for recurrence or metastasis. The imaging features of a vulvar SFT can be generalized as a well-defined mass with a heterogeneous and persistent enhancement pattern, a hypointense region associated with dense fibrous tissue on T2-weighted images, and serpentine vessels corresponding to the hypervascular nature of the tumor. Although the definitive diagnosis primarily depends on immunohistochemical and pathological analyses after complete surgical resection, preoperative imaging evaluation is also necessary for evaluation of resection margins, planning and execution of optimal surgical management, and subsequent patient care.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221112201 - Supplemental material for Magnetic resonance imaging features of a solitary fibrous tumor of the vulva: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221112201 for Magnetic resonance imaging features of a solitary fibrous tumor of the vulva: a case report by Chuxin Lin and Xiangrong Yu in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics
Zhuhai Hospital Affiliated with Jinan University (Zhuhai People’s Hospital) does not require ethical approval for reporting individual cases. Written informed consent was obtained from the patient for publication of this case report and its accompanying images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant no. 82071915).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
