Abstract
Objective
To retrospectively analyze the clinical data and treatment procedures of angiographic embolization (AE) and extraperitoneal pelvic packing (EPP) for traumatic pelvic fractures in our center for the purpose of providing recommendations on the selection of treatment protocols.
Methods
We analyzed 110 patients with traumatic pelvic fractures treated with AE and EPP from January 2015 to May 2023. The patients were divided into the AE group (69 men, 41 women) and the EPP group (20 men, 12 women). The primary outcomes were the mortality rate and incidence of complications.
Results
The mortality rate was slightly lower in the AE than EPP group (7.3% vs. 9.4%). The overall blood transfusion volume was lower and the length of hospital stay was shorter in the AE than EPP group (7.79 ± 12.04 vs. 9.14 ± 14.21 units and 20.48 ± 11.32 vs. 22.14 ± 10.47 days).
Conclusions
Both AE and EPP have good treatment effects. AE is preferred for patients in stable condition with severe hemorrhage. This study suggests that EPP should be the primary treatment and that AE should serve as a complementary treatment for critical patients.
Keywords
Introduction
Traumatic pelvic fractures have a high mortality rate ranging from 5% to 15%. 1 This high mortality rate is mainly associated with direct or indirect injury to the venous plexus or iliac vessels (e.g., lacerations, compressions, and puncture by fracture fragments), resulting in bleeding and subsequent shock, organ failure, and death.2–4 Statistical data indicate that approximately 80% to 90% of pelvic bleeding originates from veins whereas 10% to 20% originates from arteries. 5 Angiographic embolization (AE) and extraperitoneal pelvic packing (EPP) are two main methods of controlling hemorrhage. 6 However, the treatment effects of using AE or EPP to control hemorrhage associated with traumatic pelvic fractures are controversial. 7 Recent studies have explored this controversy. A retrospective cohort study utilizing the American College of Surgeons Trauma Quality Improvement Program database from 2016 to 2018 showed no significant difference in in-hospital survival between the AE and preperitoneal packing groups, although preperitoneal packing was more frequently chosen for severe pelvic fractures necessitating laparotomy. 8 Moreover, a systematic review and meta-analysis underscored the difficulty in comparing these two modalities because of biases, heterogeneity, and inadequate reporting of physiological data, emphasizing the need for standardized reporting in this high-risk group of trauma patients. 9
Since the first report of AE by Margolies et al. 10 in 1972, this technique has undergone several decades of development and improvement and is now widely applied in the treatment of traumatic pelvic fractures. Nevertheless, AE has some limitations. Pelvic fractures are associated with venous hemorrhage, which is difficult to manage by AE. This is primarily because AE is more adept at targeting and controlling active arterial than venous hemorrhage. Venous hemorrhage tends to involve a broader area than arterial hemorrhage, making precise embolization more challenging. In addition, AE requires radiological equipment and specialist radiologists in the operating room. In medical centers with poor medical facilities and resuscitation systems, preoperative preparation requires a long time, especially early in the morning or on weekends; this may drastically lower the success rate of resuscitation. Research has shown that EPP is a relatively simple and effective protocol for controlling pelvic hemorrhage in resource-limited scenarios. 7 Most pelvic hemorrhage originates from venous bleeding, 11 and compression packing is an effective method to stop such bleeding. As a prerequisite for EPP, the pelvic ring must remain stable to provide sufficient support for gauze packing.11,12 However, EPP is an invasive procedure that is associated with a high risk of infection and requires a second laparotomy to remove the packing material.13,14 One study showed that more than 25% of patients initially treated with EPP required additional AE to achieve effective hemorrhage control. 15
Possibly because of publication bias, heterogeneity, and under-reporting of physiological data, scholars and researchers have rarely compared the application of AE and EPP in treating traumatic pelvic fractures and have failed to reach a consensus and provide evidence. 11 The use of AE or EPP for treating traumatic pelvic fractures varies among countries and regions worldwide. For example, AE is the first choice in the United States, whereas EPP is the first choice in Europe. 6 Although two comparative studies on EPP and AE were published in 2016, they were small-cohort and descriptive studies.16,17 In the subsequent 7 years since those studies, no scholars or researchers have published any comparisons of the effects or prognosis of AE and EPP. Therefore, in an effort to fill this knowledge gap, we performed the present study to compare the effects, complications, and influencing factors of AE and EPP in the treatment of traumatic pelvic fractures.
Experimental design
This retrospective cohort analysis was conducted using the clinical data of patients with traumatic pelvic fractures treated at our hospital from January 2015 to May 2023. The data were collected from the self-designed trauma registry system of our hospital. This system detailed the patients’ basic information (age, sex, type of injury, initial vital signs, and trauma score) as well as their treatment process and outcome (treatment method, treatment effect, and complications).
The inclusion criteria for this study were an age of 18 to 75 years, a diagnosis of traumatic pelvic fractures and treatment with AE or EPP, and the availability of complete clinical records. The exclusion criterion was cardiac arrest upon hospital admission. The patients were divided into the AE group and the EPP group according to the expertise, clinical experience, and surgical proficiency of the surgeon.
All patients remained under the strict supervision of our trauma medical team and received an initial assessment and resuscitation according to the Advanced Trauma Life Support guidelines. The main indications for treatment with AE and EPP were exosmosis of contrast on computed tomography, a poor response to resuscitation after the exclusion of other bleeding sources (such as intrathoracic or intra-abdominal bleeding), and pelvic hematomas. After the operation, whether to return the patient to the ward or transfer the patient to the intensive care unit for further observation was determined according to the patient’s condition and the clinical judgment of the surgeon and anesthetist.
Data collection
We collected the patients’ basic information (including age, sex, and type of injury), pelvic fracture type (Tile classification), concomitant injuries (such as head, neck, thoracic, abdominal, extremity, and pelvic injuries), Injury Severity Score (ISS), and initial vital signs from the emergency department (such as systolic blood pressure, diastolic blood pressure, and heart rate). Furthermore, we recorded each patient’s shock index (SI), performance of blood product transfusion, number of units transfused, medication given in the emergency department, surgical interventions, angiographic site, type of intervention (such as embolization or stenting), method of embolization, materials used, and number of days in the intensive care unit.
The technical standards for successful AE were the disappearance of vascular abnormalities (such as contrast extravasation) after the operation and no need for further endovascular or surgical intervention. The technical standards for unsuccessful AE were failure of arterial cannulation caused by pathological or anatomical variants, failure of embolic injection, and new bleeding points in the same vessel.
Multiple computed tomography scans were performed to monitor the patients’ conditions. We evaluated the clinical success according to radiological and clinical evidence of hemorrhage control.
Rigorous data processing and statistical analysis were adopted to guarantee accurate and reliable research results. First, the patients’ medical records were checked to ensure that the patients met the inclusion criteria. Next, data were collected according to a unified process to ensure data completeness and consistency. Finally, the results were analyzed using appropriate statistical methods and software.
All patient data have been deidentified. The reporting of this study conforms to the STROBE guidelines. 18
Protocols
AE
The AE procedures were performed by the trauma surgeons at our hospital. Under the guidance of digital subtraction angiography, a modified Seldinger method was applied to achieve percutaneous femoral artery puncture and intubation. Under X-ray fluoroscopy, the surgeon slowly inserted a catheter into the abdominal aortic bifurcation and spotted the orifice of the lateral common iliac artery. Non-ionic iodine contrast was injected for bilateral iliac artery angiographic examination, and the hemorrhagic conditions and points were identified according to the contrast extravasation. After locating the hemorrhagic points, the surgeon inserted the catheter into the bleeding artery and used gelatin sponge pellets to achieve initial embolization. After successfully embolizing the hemorrhagic points, the surgeon repeated the angiographic procedure and withdrew the catheter after the hemorrhagic signs had disappeared. After identifying the bleeding artery, the surgeon selected an appropriate spring coil according to the internal diameter of the bleeding artery and used the coil for embolization. Fifteen minutes after embolization, the surgeon performed another angiogram, withdrew the catheter after obvious bleeding had stopped, and treated the puncture point with pressure dressing (see anatomical illustration in Figure 1).

Anatomical illustration. Schematic diagram of vascular embolization (left). Following the direction of the guidewire, the thickest main branches are, respectively, the femoral artery, external iliac artery, internal iliac artery, and inferior gluteal artery. The remaining branches are the (1) lumbar iliac artery, (2) superior gluteal artery, (3) lateral sacral artery, (4) obturator artery, (5) umbilical artery, (6) inferior vesical artery, (7) inferior rectal artery, and (8) internal pudendal artery.
EPP
In the EPP protocol, the surgeon first created a 7- to 8-cm vertical skin incision from the pubic symphysis, removed the anterior sheath of the rectus abdominis muscle, and separated the muscle. With medial displacement of the peritoneum, the surgeon performed blunt separation along the lateral edge of the sacroiliac joint in the preperitoneal space and then used circular forceps to tightly pack three pieces of sterile gauze from the proximal side of the sacroiliac joint; this process was repeated on the contralateral side to achieve complete hemostasis. Next, the surgeon closed the site with continuous sutures. After effectively correcting coagulation dysfunction, metabolic acidosis, and hypothermia, the surgeon performed a second observation and removed the sterile gauze packing within 48 hours according to the observation results or pronged the packing for another 12 to 24 hours if the patient’s condition was unstable. Secondary packing was usually avoided to prevent infectious complications.
Statistical analysis
Data are presented as frequency (proportion) or mean ± standard deviation. Differences in categorical variables were analyzed using the χ2 test or Fisher’s exact test, and differences in continuous variables were analyzed using the t-test or Mann–Whitney U-test, as appropriate. All data were analyzed using R version 4.2.3 (R Foundation for Statistical Computing, Vienna, Austria). A P value of <0.05 was considered statistically significant.
Results
Clinical characteristics of patients
During the 8-year research period, 142 patients with hemodynamically unstable traumatic pelvic fractures were admitted to and treated in our hospital. Of these 142 patients, 110 underwent AE and 32 underwent EPP. There was no significant difference in the mean follow-up time between the AE and EPP groups (43.5 ± 24.1 and 48.5 ± 30.16 days, respectively). The 142 patients comprised 89 (62.7%) men and 53 (37.2%) women with a mean age of 55.9 ± 15.98 years. The most common injury mechanism was a car accident (82 patients, 57.8%). The mean initial systolic blood pressure was 104.71 ± 22.63 mmHg, SI was 1.03 ± 0.359, and ISS was 27.78 ± 11.17. The internal and external iliac artery branches were the most commonly involved vessels (60 patients, 42.3%). The liver, spleen, and kidneys were the most commonly injured organs (60 patients, 42.3%), followed by the brain (12 patients, 8.5%). According to the Tile pelvic fracture classification, 68, 60, and 24 patients had Type A, B, and C pelvic fractures, respectively. In total, 132 (87.3%) patients were discharged from the hospital after treatment, 7 (4.9%) died after treatment failure, and 11 (7.7%) were sent to a higher-level hospital for further treatment because of aggravation of their condition or other reasons. Three (2.1%) patients died during follow-up; the mean ISS of these patients was 44.6 ± 11.68.
Preoperative data
The preoperative data of all patients, including their demographic characteristics, injury mechanisms, clinical characteristics, preoperative blood transfusion, and preoperative preparation time, are shown in Table 1, and the data are compared between the two groups in Table 2. Compared with the EPP group, the AE group had a higher proportion of type C pelvic fracture (the most severe type). However, the EPP group had a higher mean age (P = 0.005), ISS, and SI and a lower initial SBP upon hospital admission. The differences in the injury mechanism and preoperative preparation time between the two groups were statistically significant (P < 0.001 for both). Using an ISS of 25 as the cut-off point, the EPP group had a higher proportion of patients with an ISS of >25 (P = 0.029).
Demographic characteristics, injury mechanisms, and clinical characteristics of patients with traumatic pelvic fractures.

Data are presented as mean ± standard deviation or n (%).
ISS, Injury Severity Score; SBP, systolic blood pressure; SI, shock index.
Preoperative clinical characteristics and relevant data in AE and EPP groups.

Data are presented as mean ± standard deviation or n (%).
AE, angiographic embolization; EPP, extraperitoneal pelvic packing; ISS, Injury Severity Score; SBP, systolic blood pressure.
Intraoperative data
The intraoperative data are compared between the two groups in Table 3. The intraoperative time of the AE group was longer than that of the EPP group, but the difference was not statistically significant. In the EPP group, 28 patients underwent gauze removal 2 days after the operation, and 4 patients underwent gauze removal 5 days after the operation because of continuous bleeding. In the AE group, two patients required EPP because of failure to identify the bleeding source. The preoperative and intraoperative blood transfusion volumes were lower in the AE group than in the EPP group, and the difference in the intraoperative volume was statistically significant (P = 0.008).
Intraoperative data.

Data are presented as mean ± standard deviation or n (%).
AE, angiographic embolization; EPP, extraperitoneal pelvic packing.
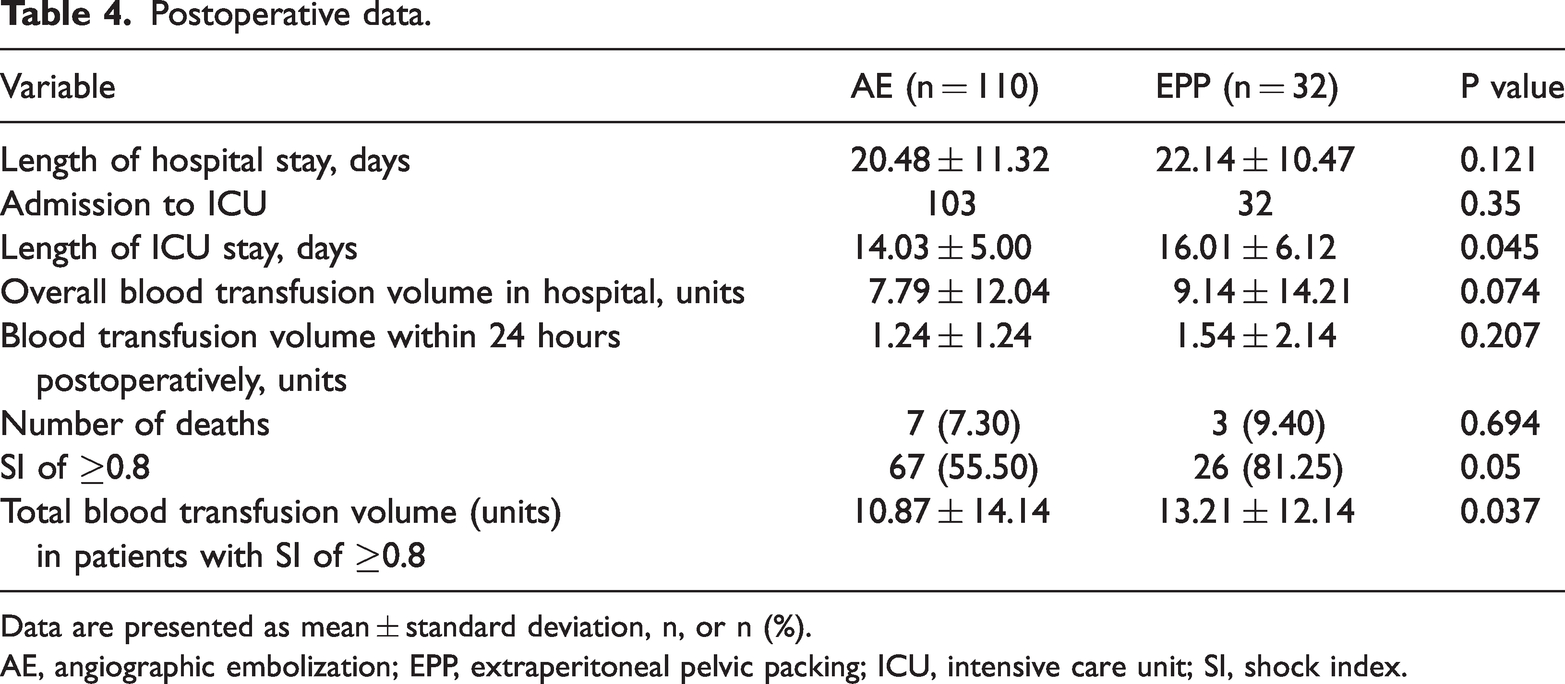
Postoperative data
The postoperative data are compared in Table 4. In the AE group, 17 (15.5%) patients experienced dull pain postoperatively, which was the most common complication in the AE group. Among the 17 patients, 4 developed numbness and local abnormalities in the lower limbs that were relieved within 3 months, and they developed no further complications such as infection or rebleeding. In the EPP group, four patients developed an internal pelvic infection 5 to 7 days postoperatively. These patients underwent treatment by surgical debridement, lavage, drainage tube placement, and upgraded antibiotics. However, the postoperative complications were not comparable between the two groups.
Postoperative data.
Data are presented as mean ± standard deviation, n, or n (%).
AE, angiographic embolization; EPP, extraperitoneal pelvic packing; ICU, intensive care unit; SI, shock index.
In the AE group, seven patients died during their hospital stay (mortality rate of 7.3%). In the EPP group, three patients died (mortality rate of 9.4%). There was no significant difference in the mortality rate (Table 4). In the AE group, one patient died of brain herniation on the first postoperative day, one died of rebleeding after embolization, two died of multiorgan failure, one died of cardiac arrest, and two died of hemorrhage from other organs. In the EPP group, one patient died of multiorgan failure, one died of infectious shock, and one died of uncontrolled pelvic hemorrhage.
The total blood transfusion volume among patients with an SI of ≥0.8 was significantly higher in the EPP than AE group (P = 0.037). However, the two groups showed no statistically significant difference in the blood transfusion volume 24 hours after the operation (Table 4).
Discussion
The present study showed that despite the extended duration of preoperative preparation and longer intraoperative time in the AE group, there was no significant increase in mortality. This finding slightly differs from that of previous studies.12,19 This pivotal discovery challenges the established belief that time is a decisive factor in the mortality rates of patients with hemodynamically unstable traumatic pelvic fractures, and it emphasizes the critical importance of early intervention and hemorrhage control in saving patients’ lives. 20 Notably, one study highlighted that a 3-minute delay in the emergency or resuscitation room could escalate the mortality rate by 1% during the initial 90 minutes of resuscitation. 21
With the ongoing advancements in medical instruments, materials, and AE techniques, increasing numbers of trauma centers are adopting AE as the preferred treatment method. 8 Conversely, EPP is a more invasive procedure that requires a follow-up surgery to remove the pelvic packing, thereby significantly elevating the risk of infection.13,14 Consequently, our center predominantly performs AE, which explains the disparity in the sample sizes between the two groups in this study.
Notably, the relatively small sample size of our study might have compromised the accuracy and generalizability of our findings and hindered our ability to conduct comprehensive comparisons and in-depth analyses akin to studies based in larger centers. Interestingly, we found a considerably lower mortality rate than in previous research.16,17,22,23 This disparity might have originated from the research design (patients who were deceased upon hospital admission were excluded from our sample) rather than a difference in treatment protocols.
Despite these limitations, our study demonstrated a higher survival rate associated with AE than EPP in our center. This can be attributed to the proficient surgical skills and extensive clinical experience of the surgical team coupled with the significant advancements in interventional techniques and the enhancement of embolization materials in recent years. Consistent with this, a review by McDonogh et al. 8 of literature published from 2000 to 2020 indicated a gradual, albeit subtle, decline in the overall mortality rate among patients who underwent AE, a trend not observed in the EPP group.
This research offers fresh perspectives, suggesting that AE might be a more favorable option, particularly when considering its potential for higher survival rates and reduced risk of complications.
AE can effectively seal the damaged vessels through an intervention that does not require the pelvis to be opened. However, AE cannot stop venous bleeding. Blood flowing into the peritoneal cavity due to retroperitoneal perforation would be catastrophic. By contrast, EPP can directly manage proximal retroperitoneal arterial bleeding. In patients with distal iliac arterial bleeding, the surgeon should advance more deeply into the pelvic cavity to separate more tissue structures; however, this may cause more complications. Fortunately, no such complications occurred in the present study. Infection remains a serious complication of EPP because of foreign body packing and massive surgical trauma. The primary cause of complications in AE is inappropriate embolization, which may cause problems such as renal failure or ischemic necrosis at non-targeted embolization sites. According to previous studies, bilateral nonselective vascular embolization bears a high risk of serious complications, including gluteal muscle necrosis, deep bladder necrosis, and deep infection.24,25 Unilateral or selective vascular embolization is the preferred choice to avoid these complications. Vague pain in the embolized muscle groups, numbness in the lower limbs, and sensory abnormalities occurred in the patients in our study, but no patients developed any of the above-described serious complications. This suggests that such sensory abnormalities are related to vascular issues in addition to neurogenic issues.
El-Menyar et al. 26 performed an in-depth study of the SI of trauma patients and found that an SI of ≥0.8 was the cut-off value for severe hemorrhage with resultant shock in these patients. In the present study, we conducted a subgroup analysis using an SI of 0.8 as the cut-off point and found that patients with an SI of ≥0.8 had a much higher total transfusion volume (13.61 ± 14.36 units) than those with an SI of <0.8 (3.52 ± 2.1 U) (Table 1), and all patients who died in this study had an SI of >0.8. This further proves that the SI is an important index for predicting the severity and prognosis of the patient’s condition. Furthermore, the ISS, an important prognostic index, can predict the short-term survival of patients with traumatic pelvic fractures. 27 Some studies have shown that with an ISS of >57, the mortality rate of patients with hematologically unstable traumatic pelvic fractures was as high as 57%. 28
Blood transfusion is a crucial means of maintaining vital signs and critically assessing patients with traumatic pelvic fractures. According to relevant reports, every 1-unit/hour increase in the transfusion demand increases the risk of mortality by 62%. 29 Bize et al. 30 found that the number of transfusions was correlated with the prognosis. In managing traumatic fractures, patients with 10 or more transfusions had a mortality rate of 59%, whereas patients with fewer than 10 transfusions had a mortality rate of 20%. 30 In the present study, patients in the EPP group required more blood transfusions, perhaps because of the higher ISS in this group (a higher ISS suggests more severe trauma and more blood loss). We considered four possible reasons for the higher number of blood transfusions in the EPP than AE group. First, the cutting of tissue during the surgical operation may had led to secondary blood loss. Second, it was impossible to precisely determine the point of bleeding during EPP, which may have led to continuous bleeding. Third, when the hematoma was removed, the pressure of the hematoma on the bleeding point disappeared, potentially resulting in rebleeding at the site of vascular injury. Fourth, it was difficult to stop bleeding from small blood vessels in many cases. Although the AE group had a lower ISS, the total blood transfusion volume was not significantly lower in the AE than EPP group because of the longer preoperative preparation and intraoperative times in the AE group. We found a significant decrease in the transfusion volume in both the AE and EPP groups within 24 hours after the intervention, which is consistent with the findings reported by Li et al. 16 and Tai et al. 19 Osborn et al. 23 found a significant decrease in the transfusion volume within 24 hours after the EPP intervention but no change in the transfusion volume within 24 hours after the AE intervention, presumably because their study was performed in 2019 when AE techniques and hemostatic materials were poor and affected the hemostatic outcome. In the present study, some patients in the EPP group required additional AE to achieve final transfusion control. Our data indicate that AE was superior to EPP in terms of hemostasis. It is essential to point out that the AE protocol requires specialist equipment and radiological staff and is associated with longer preoperative preparation and intraoperative times, making EPP the preferred choice for patients who cannot tolerate a long waiting period because of critical illness. This view was supported by Suzuki et al., 31 who believed that EPP should be the first choice for hemodynamically unstable patients. Totterman et al. 32 suggested that once patients become hemodynamically stable, AE can serve as a supplement to EPP. The complementary combination of AE and EPP is the best choice in the clinical setting according to the recommendations of the World Society of Emergency Surgery guidelines.11,33
Notably, this was a retrospective single-center study with an unbalanced sample size. Additionally, the treatment protocol was not randomly selected but determined according to the subjective judgment and personal preference of the surgeon present. The specific treatment and embolization methods were not standardized, and differences in technique, experience, practice, equipment, and materials may have affected the treatment outcomes. These limitations highlight the descriptive nature of our study and suggest that the conclusions drawn should be interpreted with caution. Additional large-scale, multicenter, randomized experiments should be conducted to deepen research in this field.
In summary, our findings suggest that both AE and EPP can offer therapeutic benefits. Although AE appears to be a favorable option for patients in stable condition with severe bleeding, it is important to note that this preference is based on a limited dataset and specific center experience. Because preoperative preparation usually requires a long time, the treatment protocol should be selected according to the patient’s vital signs and the hospital facilities. This study proposes a nuanced approach in which surgeons might consider EPP as a primary option complemented by AE, particularly for patients in critical condition with severe hemorrhage. However, we emphasize that these conclusions are preliminary and that further substantiated research is necessary to provide a more comprehensive perspective.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231208601 - Supplemental material for Extraperitoneal pelvic packing versus angiographic embolization for hemodynamically unstable pelvic fractures: a retrospective single-center analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605231208601 for Extraperitoneal pelvic packing versus angiographic embolization for hemodynamically unstable pelvic fractures: a retrospective single-center analysis by Jisen Chu, Changyuan Xie, Jiaojiao Fu and Weigen Yao in Journal of International Medical Research
Footnotes
Acknowledgement
We thank Dr. Kevin Li for providing assistance with language revision.
Authors’ contributions
Js C and Cy X drafted the manuscript. Wg Y performed the literature revision. Jj F performed the procedures and was involved in the collection and processing of the data. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
We obtained written informed consent for publication from the patients described in this study.
Declaration of competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Medical Ethics Committee of Yuyao People’s Hospital (Approval No. S2023-103). The clinical data were retrospectively registered.
Funding
This study was supported by the SRIP Research Project Fund of Ningbo University (2023SRIP1921, 2023SRIP1940) and the Research and Innovation Foundation of Ningbo University (IF2023059).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
