Abstract
Objective
The serum glucose/potassium ratio (GPR) is a potential prognostic predictor for acute brain injury-related diseases. We calculated the serum GPR in patients with acute intracerebral hemorrhage (ICH) and explored its prognostic value for long-term prognoses and ICH severity.
Methods
This retrospective cohort study consecutively included 92 patients with ICH and 92 healthy controls. The National Institutes of Health Stroke Scale (NIHSS) score, Glasgow coma scale (GCS) score, and hematoma volume were used to assess severity. A modified Rankin Scale score > 2 at 90 days post-stroke was defined as a poor outcome.
Results
The serum GPR was significantly higher in patients than controls. The serum GPR was weakly correlated with the NIHSS score, GCS score, and hematoma volume. The serum GPR, GCS score, and hematoma volume were independently associated with poor outcomes. In the receiver operating characteristic curve analysis, the serum GPR remarkably discriminated patients at risk of poor outcomes at 90 days. The serum GPR significantly improved the prognostic predictive capability of hematoma volume and tended to increase that of the GCS score.
Conclusion
Serum GPR is an easily obtained clinical variable for predicting clinical outcomes after ICH.
Introduction
Spontaneous intracerebral hemorrhage (ICH) represents one of the leading causes of death and long-term disability worldwide. 1 In recent decades, researchers have given increasing attention to recognition of prognostic biomarkers because early prognosis prediction can aid in the modification of therapeutic strategies and further improve ICH outcomes. Inflammatory biomarkers (such as circulating matrix metalloproteinases and monomeric C-reactive protein levels),2,3 immune parameters (for instance, the serum neutrophil-to-lymphocyte and lymphocyte-to-monocyte ratios),4,5 and metabolic or homeostatic indices (e.g., serum magnesium and serum calcium levels)6,7 have been extensively studied to assess their ability to predict hematoma growth after ICH. However, those indices have not been applied clinically because they are typically unavailable or because of their insignificant clinical value for prognostic prediction. Serum glucose and potassium are two routine laboratory parameters. High serum glucose levels and low serum potassium levels are frequently found in some critical illnesses, including sepsis, acute myocardial infarction, and hemorrhagic shock. 8 Additionally, these changes have been found in some diseases involving acute brain injury, such as severe traumatic brain injury and aneurysmal subarachnoid hemorrhage.9–11 The serum glucose/potassium ratio (GPR) is calculated as the serum glucose level divided by the serum potassium level. Its elevation has been reported to be highly associated with increasing severity and poor prognosis of certain pathological conditions, e.g., carbon monoxide poisoning. 12 More recently, two reports of patients with severe traumatic brain injury and aneurysmal subarachnoid hemorrhage investigated the relationship between the serum GPR and disease severity as well as the prognosis and found that an increase in this ratio could be used to assess the severity and independently discriminate patients at risk of a poor prognosis. Clearly, elevated serum glucose levels are closely related to a poor functional prognosis of ICH.9–11 Nevertheless, decreased serum potassium levels were not associated with a poor outcome after ICH. 2 However, to the best of our knowledge, it remains unknown whether the serum GPR is related to the prognosis of ICH, independent of the serum glucose and potassium levels. In the current study, we aimed to determine whether the serum GPR could be a potential predictor of the long-term prognosis and severity of ICH.
Materials and methods
Study population
In this retrospective cohort study performed at Hangzhou First People’s Hospital from November 2018 to December 2019, we consecutively recruited all patients with first-ever spontaneous ICH diagnosed via head computed tomography scans. We required that all patients be admitted within 24 hours after symptom onset of ICH. The exclusion criteria included the following: (a) age < 18 years; (b) hemorrhages caused by traumatic brain injury, venous sinus thrombosis, infarction, or tumors; (c) previous stroke; and (d) other diseases such as severe infection within the previous 4 weeks, autoimmune diseases, pregnancy, or known malignancies. Additionally, healthy individuals who underwent a physical examination at our hospital were consecutively recruited as controls from November 2018 to December 2019. We excluded those subjects with previous stroke or other diseases such as severe infection within the previous 4 weeks, autoimmune diseases, pregnancy, or known malignancies. In addition, control subjects were required to have normal routine laboratory test and radiological examination results. This study complied with the ethical guidelines of the Declaration of Helsinki and was approved by the Institutional Review Board at Hangzhou First People’s Hospital on 10 April 2018 (HZSY2018213). Because this was a retrospective study, written informed consent was waived.
Assessments
We collected data including demographics, clinical features, and medical history. Both the National Institutes of Health Stroke Scale (NIHSS) and Glasgow Coma Scale (GCS) were used to evaluate stroke severity at admission.13,14 Hematoma volume was measured in accordance with the ABC/2 method. 15 Briefly, the computed tomography slice with the largest hemorrhage area was identified. The largest diameter (A) of the hemorrhage on this slice was measured. Next, the largest hemorrhage diameter on a line perpendicular to A on the same slice was measured (B). Finally, the number of 5- or 10-mm slices in which the ICH was visible was determined (C). A, B, and C were then multiplied, and the product was divided by 2, which yielded the hemorrhage volume in cubic centimeters. The presence of intraventricular or subarachnoidal extension of hematoma was also recorded. A modified Rankin Scale (mRS) score > 2 points during the 90-day follow-up period after stroke onset was considered an unfavorable outcome. 16 Serum glucose and potassium levels were measured using routine methods at our hospital. Briefly, blood glucose levels were recorded using a Hitachi Modular Analytics System P800 chemistry analyzer (Roche Diagnostics K.K., Tokyo, Japan), and serum potassium was assessed with an indirect ion selective electrode on a Beckman Coulter AU 5800 analyzer (Beckman Coulter Inc., Brea CA, USA). The serum GPR was calculated by dividing the serum glucose level by the serum potassium level.
Statistical analysis
Statistical analyses were conducted with IBM SPSS Statistics for Windows, version 19.0 (IBM Corp., Armonk, NY, USA) and MedCalc 9.6.4.0 (MedCalc Software, Mariakerke, Belgium). Figures were plotted using GraphPad Prism version 5.0 (GraphPad Software Inc., La Jolla, CA, USA). The normality of the data distribution was evaluated using the Kolmogorov–Smirnov test or Shapiro–Wilk test. Except for age, continuous variables were non-normally distributed and therefore are summarized as medians (lower and upper quartiles). Age is presented as the mean ± standard deviation. Categorical variables are reported as counts (percentages). Data were compared among multiple groups using the Kruskal–Wallis H test. Data comparisons between two groups were performed using the χ2 test, Fisher exact test, t test, or Mann–Whitney U test as appropriate. Bivariate correlations were analyzed using Spearman correlation coefficients. To identify predictors independently related to 90-day clinical outcomes, we constructed a binary logistic regression model, which contained the significant variables (P < 0.05) from the univariate analysis. Results are presented as odds ratios and 95% confidence intervals (CI). Receiver operating characteristic (ROC) curve analysis was performed to assess the prognostic predictive accuracy of the serum GPR, and the area under the curve (AUC) was estimated. A value of P < 0.05 was considered statistically significant.
Results
Patient characteristics
During the study period, 125 patients with first-ever spontaneous ICH hospitalized within 24 hours after symptom onset of stroke were analyzed. Afterwards, we excluded 33 patients because of the reasons depicted in Figure 1, and 92 patients with ICH were eventually enrolled for further analysis. The patients included 53 men and 39 women, and their age ranged from 31 to 90 years (mean, 62.2 years; standard deviation, 14.4 years). In addition, 92 controls, including 48 men and 44 women, aged from 30 to 91 years (mean, 61.5 years; standard deviation, 14.1 years) were included. No statistically significant differences were observed in terms of age and sex proportion between patients with ICH and controls (both P > 0.05). Among all participants, 20 were cigarette smokers and 27 had a history of alcohol abuse. A history of illness included hypertension (55 patients) and diabetes mellitus (13 patients). Patients were admitted to the hospital at a median time of 6 hours after symptom onset (range, 1–24 hours; interquartile range, 3–9 hours). Blood was collected at 2 to 28 hours after stroke (median, 8 hours; interquartile range, 6–12 hours). Regarding hemorrhagic severity, the median GCS and NHISS scores were 13 (range, 4–15; interquartile range, 9–15) and 9 (range, 0–38; interquartile range, 2–18), respectively. The radiological parameters included hematoma location (infratentorial hemorrhage, 13 patients), the presence of intraventricular bleeding (25 patients), and hematoma volume (median, 15 mL; interquartile range, 7–34 mL; range, 1–65 mL). According to non-invasive arterial pressure measurements, the median systolic arterial pressure and diastolic arterial pressure values were 155 mmHg (range, 107–234 mmHg; interquartile range, 143–174 mmHg) and 88 mmHg (range, 55–188 mmHg; interquartile range, 79–98 mmHg), respectively.

Flow chart for selecting eligible patients with acute spontaneous intracerebral hemorrhage
Serum GPR and severity
As shown in Figure 2, the median serum GPR was significantly higher in patients with ICH than in controls (median, 1.71 versus 1.33, P < 0.001). Alternatively, weak correlations were observed between the serum GPR and the GCS score (r = −0.429, P < 0.001), hematoma volume (r = 0.242, P = 0.020), and NIHSS score (r = 0.439, P < 0.001). Nevertheless, the serum GPR was not significantly correlated with the onset to blood sampling time (r = −0.181).

Differences in the serum glucose/potassium ratio between healthy controls and patients with intracerebral hemorrhage.
Serum GPR and clinical outcomes
According to the mRS scores at 90 days post-stroke, 56 patients (60.9%) had a poor outcome (mRS score > 2). Death occurred in 13 cases (14.1%). As displayed in Figure 3, the serum GPR was remarkably increased with increasing mRS scores (P < 0.001). Furthermore, as shown in Table 1, significant associations were observed between poor outcome and serum glucose concentration (P < 0.01), serum potassium concentration (P < 0.05), serum GPR (P < 0.01), GCS score (P < 0.01), NIHSS score (P < 0.01), and hematoma volume (P < 0.01). The preceding significant variables were entered into the multivariate logistic regression model, which showed that the factors independently associated with a poor 90-day outcome in patients with ICH included the serum GPR (odds ratio, 2.945; 95% CI, 1.099–7.889, P = 0.032), GCS score (odds ratio, 0.670; 95% CI, 0.478–0.940, P = 0.020), and hematoma volume (odds ratio, 1.089; 95% CI, 1.016–1.168, P = 0.017, Table 2).

Association of mRS scores with serum glucose/potassium ratio after acute intracerebral hemorrhage.
Results of statistical analysis of the risk factors for a poor outcome after acute intracerebral hemorrhage.

Quantitative data are reported as medians (lower and upper quartiles) or the mean ± standard deviation as appropriate. Qualitative variables are reported as counts (percentages). Intergroup comparisons were performed using the χ2 test, Fisher’s exact test, t tests, or the Mann-Whitney U test as appropriate. A modified Rankin Scale score > 2 points during the 90-day follow-up period after stroke onset was considered a poor outcome.
NS, not significant.
Multivariate logistic regression analysis for risk factors of poor outcome of acute intracerebral hemorrhage.

Results are presented as odds ratios (95% confidence interval) according to a binary logistic regression analysis.
Analysis of the predictive capability of the serum GPR
According to the ROC curve analysis, the serum GPR remarkably predicted poor outcomes at 90 days post-stroke among this group of patients with ICH (AUC: 0.720; 95% CI, 0.614–0.826). Moreover, using the Youden index method, an optimal value of the serum GPR was determined (0.408), which generated medium-high sensitivity and specificity values (0.7143, 95% CI, 0.5779–0.8270 and 0.6944, 95% CI, 0.5189–0.8362; Figure 4). Furthermore, the AUCs of the two most common prognostic indicators for ICH (i.e., GCS score and hematoma volume) were 0.879 (95% CI, 0.803–0.956) and 0.804 (95% CI, 0.713–0.894), respectively, for differentiating a 90-day unfavorable outcome. The combination of the serum GPR and hematoma volume on admission showed a significantly higher predictive accuracy (AUC: 0.870, 95% CI: 0.795–0.944) than the serum GPR or hematoma volume alone (P < 0.01 and P = 0.041, respectively, Figure 5). Alternatively, the combination of the serum GPR and GCS score tended to increase the predictive capability (AUC: 0.891, 95% CI: 0.819–0.962) compared with the GCS score alone (Figure 6) and significantly improved the prognostic predictive value of the serum GPR (P < 0.01, Figure 6).
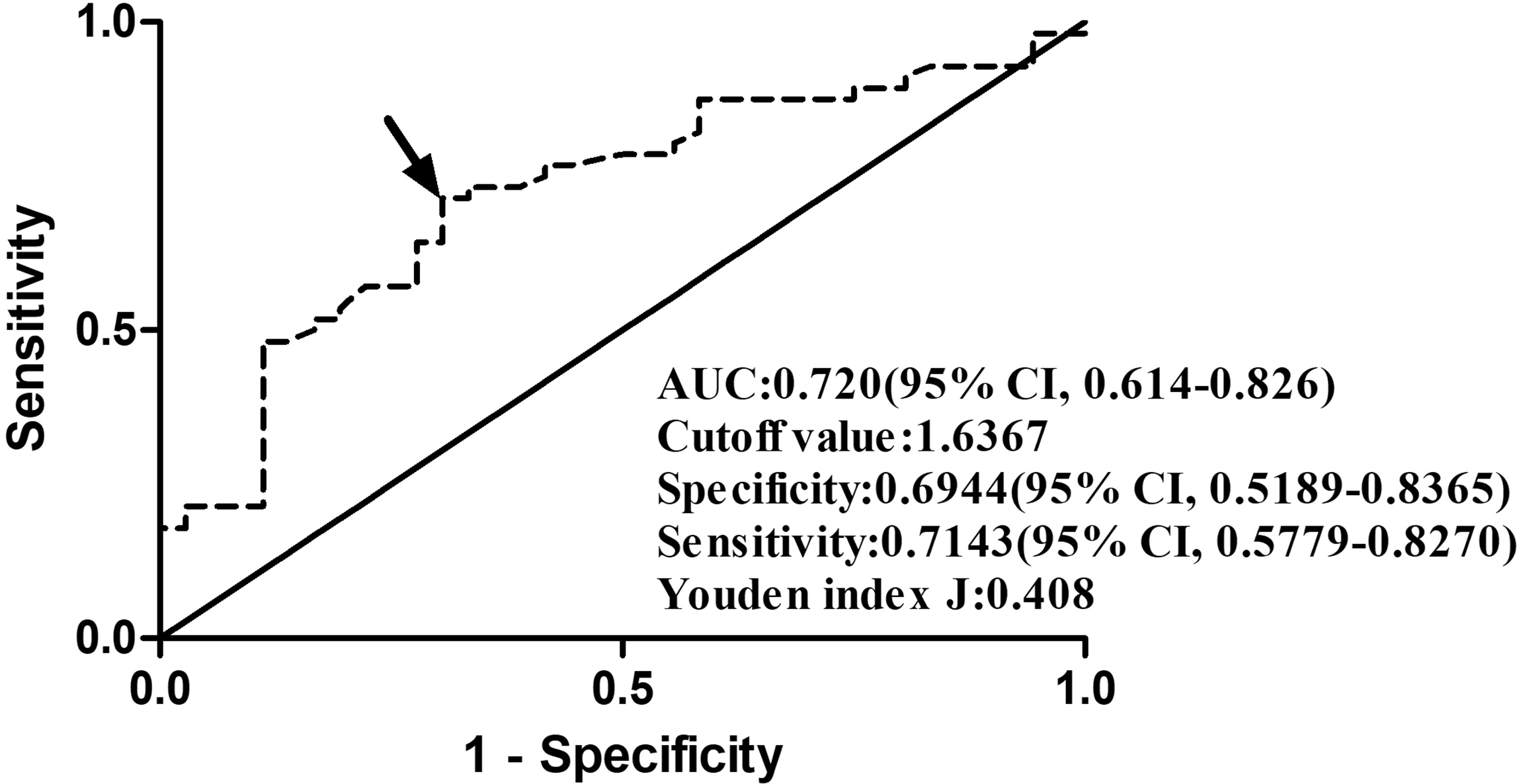
Graph displaying receiver-operating characteristic curve using the serum glucose/potassium ratio to investigate poor outcomes at 90 days after spontaneous acute intracerebral hemorrhage. The arrow indicates the optimal cut-off point of the serum glucose/potassium ratio according to the Youden method

Receiver operating characteristic curve analysis used to determine whether the serum glucose/potassium ratio improved the prognostic predictive capability of hematoma volume after acute intracerebral hemorrhage.

Receiver operating characteristic curve analysis was used to determine whether the serum glucose/potassium ratio improved the prognostic predictive capability of the GCS score after acute intracerebral hemorrhage.
Discussion
This is the first study to examine the correlation of the serum GPR with hemorrhagic severity and long-term outcomes in patients after ICH. The results of this study indicated that the serum GPR is weakly linked to hemorrhagic severity and is independently associated with unfavorable 90-day outcomes in patients with ICH. Moreover, this ratio significantly enhanced the prognostic predictive ability of hematoma volume. Thus, the serum GPR may be used as a prognostic factor for the long-term prognosis after ICH.
According to a previous study, the incidence rate of hypokalemia after ICH is comparatively high. 16 The reported incidence rate of hypokalemia after craniocerebral disease is 43.7% to 65.5%, whereas the overall incidence rate of hyperkalemia is 1.3% to 5.7%.17,18 Furthermore, hypokalemia was shown to have an adverse impact on early death caused by ICH in both male and female patients in a clinical study. 19 However, hypokalemia was not associated with a poor functional outcome after ICH. 2
Post-stroke hyperglycemia has been confirmed to be related to hematoma expansion, presence, and severity of intraventricular extension, perihematomal edema, and poor clinical outcome in patients with acute ICH.20–24 The exact pathophysiological mechanism underlying the relationship between post-stroke hyperglycemia and severe neurological deficit or poor outcome in patients with ICH remains unclear. Some studies have suggested that post-stroke hyperglycemia may arise from a stress reaction, and the major glucose-regulatory hormones involved in the hyperglycemic response include catecholamines, glucagon, and corticosteroids.25–27 Catecholamines are of paramount importance following injury and stress because they increase glucagon secretion, thereby elevating blood glucose concentrations. 28
Potassium, which is stored mostly within cells, is transported by active cellular uptake through the cell membrane and by the sodium/potassium adenosine triphosphatase pump (Na+/K+-ATPase). Na+/K+-ATPase is controlled by catecholamine, B2 adrenergic hormones, and insulin, which can lead to a decline in circulating blood potassium concentrations. 29 Furthermore, ICH results in an elevated serum glucose concentration because of excessive catecholamine secretion as mentioned above; therefore, under this circumstance, insulin secretion intensifies and serum potassium enters the cells. 30 However, in our study, the serum glucose concentration was associated with a poor outcome at 90 days only in the univariate analysis, not in the multivariate model. Furthermore, the close relationship between the serum potassium concentration and prognosis of patients with ICH was only found in the univariate analysis in our study. Instead, the serum GPR, GCS score, and hematoma volume were independent predictors for poor outcome at 90 days. Therefore, the serum GPR better reflects excessive catecholamine levels after ICH than the serum glucose concentration or serum potassium concentration alone.
Our study found no significant relationship between the serum GPR and the time from onset to blood sampling after ICH. Although the serum glucose level is time-dependent, serum glucose levels of patients with ICH are greatly affected by ICH severity. 31 Therefore, the serum GPR may not be related to the time from onset to blood sampling. In addition, the current study revealed that the serum GPR was weakly correlated with the GCS score, NIHSS score, and hematoma volume (all r values less than 0.5). Such weak correlations cannot represent close associations with disease severity. However, other studies have shown that the serum GPR was closely correlated with the severity of acute brain injury. 13 Hence, it is possible that such a weak correlation results from the small sample size (92 patients).
Our study revealed that an increase in the serum GPR, but not in the serum glucose and serum potassium levels alone, was obviously related to a poor clinical prognosis, i.e., 90-day outcome post-stroke, after adjusting for GCS scores, NIHSS scores, and hematoma volume. More interestingly, according to the ROC curve analysis, the serum GPR significantly improved the prognostic predictive ability of admission hematoma volume and tended to increase that of baseline GCS scores in patients with ICH. Thus, the serum GPR, as an easily detectable laboratory index, could be of great value for increasing the predictive ability of the hematoma volume and GCS score for the prognosis of patients with ICH.
This study had several limitations. Initially, we did not collect data on serum hormone levels such as catecholamines, glucagon, and corticosteroids; as a result, the actual effects of the serum GPR in patients with severe ICH are not clear. Second, our analysis was conducted at a single center, resulting in a small sample size. A prospective and multi-center study is needed in the future to support our conclusions. Finally, absolute blood pressure levels and their fluctuations over time have been shown to predict outcomes of patients with cerebral hemorrhage and are targets of intervention. 32 Therefore, the lack of blood pressure variability data is also a limitation of the study.
Conclusions
In this study, for the first time, we discuss and demonstrate the utility of the serum GPR for prediction of the long-term prognosis and assessment of ICH severity. An elevated serum GPR was weakly correlated with ICH severity and was remarkably associated with a poor clinical prognosis. Moreover, this ratio improved the prognostic predictive ability of the GCS score and hematoma volume. Thus, the serum GPR may be useful for predicting the prognosis of ICH.
Footnotes
Acknowledgements
The authors thank all staff members in the Department of Neurosurgery, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine (Hangzhou, China) for their technical support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was financially supported by the Key Plan of Science and Technology Department of Zhejiang Province (Grant No. 2020C03071).
