Abstract
Objective
The relationship between the worst pattern of invasion (WPOI) and the prognosis of oral squamous cell carcinoma (OSCC) remains controversial. This systematic review and meta-analysis was performed to determine the impact of the WPOI on the prognosis of OSCC.
Methods
Studies from six databases were assessed for quality using the Newcastle–Ottawa Scale, and data were analyzed using Stata software.
Results
Eighteen studies involving 3954 patients showed that patients with a WPOI of 4 to 5 had significantly worse overall survival, disease-specific survival, and disease-free survival than patients with a WPOI of 1 to 3. Significant associations of the WPOI with locoregional recurrence and mortality were found.
Conclusion
A higher WPOI was significantly associated with a worse prognosis of OSCC across various outcomes. Incorporating the WPOI into standard histopathological assessments may guide personalized treatments and improve outcomes.
Keywords
Introduction
Oral cancer ranks among the top 10 most common malignancies worldwide and is associated with high morbidity and mortality rates.1,2 Oral squamous cell carcinoma (OSCC) accounts for more than 90% of oral cancer cases. Despite early diagnosis and treatment, a considerable number of patients with early-stage OSCC die of the disease. 3 The prognosis of OSCC depends on various factors that can influence cancer outcomes, including histomorphological parameters. These parameters can be used to predict patient survival and classify patients as having high-risk or low-risk disease. Patients with high-risk disease face adverse outcomes such as local recurrence, regional metastasis, and reduced survival rates, necessitating more aggressive treatments such as multimodal therapy. By contrast, patients with low-risk disease have increased chances of favorable outcomes. 4
The worst pattern of invasion (WPOI) describes the infiltration of tissue by cancer cells at the tumor/host interface and is considered a critical factor in histological grading systems, particularly for OSCC. The WPOI is typically detected histologically after surgical tumor removal. 5 In 2005, Brandwein-Gensler et al. 6 identified and validated five WPOI categories for OSCC. The low-invasiveness types were type 1, which has broad pushing borders and is cohesive; type 2, which has broad pushing finger-like growths or separate large tumor islands; and type 3, which has invasive tumor islands containing more than 15 cells per island. The high-invasiveness types were type 4, which has invasive tumor islands containing fewer than 15 cells per island that are separated from the main tumor mass, and type 5, which has tumor satellites of any size that are 1 mm or further distant from the main tumor or next closest satellite with intervening normal tissue. Subgroups of these types were based on invasive properties and patient survival rates.7,8 The American Joint Committee on Cancer staging system recently considered the presence of a WPOI of 5 at the advancing tumor edge as a prognostic key in oral cancer. 9
Some authors have suggested that the WPOI is correlated with decreased T-cell infiltration, which contributes to adverse outcomes in patients with head and neck squamous cell carcinoma. 8 Other studies proposed that the depth of invasion (DOI) and WPOI are the most significant pathological predictors for locoregional recurrence (LRR). 3 However, few studies with adequate sample sizes have analyzed the WPOI types in terms of their relationship to the prognosis of OSCC, leaving the issue controversial.5,10–12 This systematic review was performed to determine the impact of the WPOI on the prognosis of OSCC.
Methods
We adhered to the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. 13 This systematic review and meta-analysis was registered at the PROSPERO international prospective register of systematic reviews (registration number: CRD42023423023). The requirement for ethics committee or institutional review board approval was waived because of the nature of the study (systematic review and meta-analysis of published data).
Eligibility criteria
The following inclusion criteria were used for the selection of studies.
Population: Patients with OSCC were included independent of the location, size, thickness, depth, stage, or differentiation of their tumors. Exposure: The study provided information on the WPOI prevalence, type, or impact. Comparison: The study reported on patients with WPOIs of different grades. Outcomes: The study provided information on correlations between the WPOI and overall survival (OS) rate, disease-specific survival (DSS) rate, disease-free survival (DFS) rate, and recurrence rate. Study design: Observational studies (i.e., case-control, cohort, and cross-sectional studies) were included. Language: The article was published in the English language.
Studies were excluded if they were case reports, were not written in English, were abstracts from conferences, involved research on animals, or were inconsistent articles.
Search strategy
In June 2022, we conducted a database search using the terms “(oral OR tongue OR lingual OR buccal OR gingiva OR mouth cancer OR squamous cell carcinoma OR tumor OR neoplasm) AND (prognosis OR predict OR survival OR recurrence OR mortality OR metastasis) AND (depth of invasion OR invasion OR tumor thickness OR tumor length OR tumor budding OR budding OR pattern of invasion OR tumor invasion OR tumor infiltration OR tumor island)” in the databases of Cochrane Central, ProQuest, PubMed, Scopus, Science Direct, and Web of Science. We checked the reference lists of all included citations and imported them into Rayyan, a web application for systematic reviews. 14 We recorded the study ID, publication year, title, abstract, keywords, digital object identifier, and URL. Duplicates were removed.
Selection process
Two independent reviewers (N.B. and Y.M.) conducted the selection process using a two-step screening technique. Step one involved screening the title and abstract of all studies identified during the literature search to determine which studies would proceed to full-text screening. In step two, the reviewers assessed each study’s eligibility by reading the full text.
Data extraction
The same two reviewers (N.B. and Y.M.) independently extracted the following data from the included studies and entered them into an Excel spreadsheet: demographic data of the included patients (age and sex), study characteristics (study groups, study duration, follow-up duration, total sample size, country, and main findings), lesion characteristics (type, size, location, thickness, depth, surgical margins, and WPOI), and outcomes (rates of recurrence, OS, DSS, and DFS).
Risk of bias and quality assessment
Quality assessments were independently performed by the same two reviewers (N.B. and Y.M.). Discrepancies in assessments were resolved through discussion until a consensus was reached. The Newcastle–Ottawa Scale was used to assess the risk of bias in the included studies. 15 The scale contains eight items within three domains, and the maximum score is 9. A study with a score of 7 to 9 is of good quality, that with a score of 4 to 6 is of fair quality, and that with a score of 0 to 3 is of poor quality.
Statistical analyses
A meta-analysis was performed using the DerSimonian and Laird random-effects model. 13 Statistical analyses were carried out using Stata Version 17 (StataCorp, College Station, TX, USA). Pooled estimates of the hazard ratios (HRs) with 95% confidence intervals (CIs) were obtained by a fixed-effects or random-effects meta-analysis according to the heterogeneity using the inverse-variance weighting method based on published CIs for the HRs. Using the I2 statistic, we calculated the percentage of heterogeneity and inconsistency between studies, with values of 25%, 50%, and 75% deemed low, moderate, and high, respectively. The random-effects model was employed if the heterogeneity was considerable and the I2-value was >50%; otherwise, the fixed-effects model was used. Publication bias was not assessed because the generated forest plots included fewer than 10 studies. 16 A p-value of <0.05 was considered statistically significant.
Results
Search results
We performed a comprehensive search across 6 authentic databases, and we found 4892 references that met our inclusion criteria. Using Rayyan, a platform for systematic reviews, we removed the duplicates and arrived at a total of 4849 articles that could be subjected to further screening. During the initial title and abstract screening, we excluded 4795 citations from our study screening sheet. The remaining 54 studies were then subjected to full-text screening. After this thorough screening process, we agreed to include 18 studies involving 3954 patients in our qualitative and quantitative synthesis.1–5,8,10–12,17–25 The study flow diagram of the selection process is shown in Figure 1.

PRISMA flow diagram. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of included studies
The year of publication ranged from 2011 to 2022. Six of the included studies were from India, two were from the United States, and one each was from China, Scotland, Japan, Sweden, Finland, Brazil, and Ireland. The remaining studies were multicentric studies. All included studies were of a cohort design. The average percentage of male patients in the included studies was 61.94%. Tables 1 and 2 summarize the characteristics of the included studies and patients, respectively.
Study and patient characteristics.

WPOI, worst pattern of invasion; OS, overall survival; DFS, disease-free survival; PNI, perineural invasion; MOI, mode of invasion; TB, tumor budding; LHR, lymphocytic host response; OSCC, oral squamous cell carcinoma; DOI, depth of invasion; LRR, locoregional recurrence; GBCSCC, gingivobuccal complex squamous cell carcinoma; LN, lymph node; BPOI, biopsy pattern of invasion; NA, not available.
Histopathological characteristics of included patients.

WPOI, worst pattern of invasion; PNI, perineural invasion; TB, tumor budding; NA, not available; LVI, lymphovascular invasion
Quality assessment of included studies
Based on the Newcastle–Ottawa Scale, one, eight, six, and three studies received a score of 6, 7, 8, and 9, respectively. The quality assessment is summarized in Supplementary File 1.
Overall survival rates
Three studies reported data regarding the association between the WPOI and OS rate.5,10,23 The pooled data showed that patients with a WPOI of 4 to 5 had a worse OS rate than those with a WPOI of 1 to 3 (HR, 2.17; 95% CI, 1.79–2.55, p < 0.001) (Figure 2). The pooled data were homogeneous (I2 = 0.0%, p = 0.45). Individual studies showed that a high WPOI was associated with a worse OS rate. Marinelli et al. 20 demonstrated that patients with a WPOI of 5 had a worse 5-year OS rate than those with a WPOI of 1 to 4 (HR, 321.70; 95% CI, 14.31–7228.42; p < 0.001); however, we did not include this study in the analysis because it was considered an outlier. Chaitra et al. 17 reported that the mortality rate was 70% in patients with a WPOI of 5 compared with 26% in patients with a WPOI of 0 to 4 (p < 0.01).
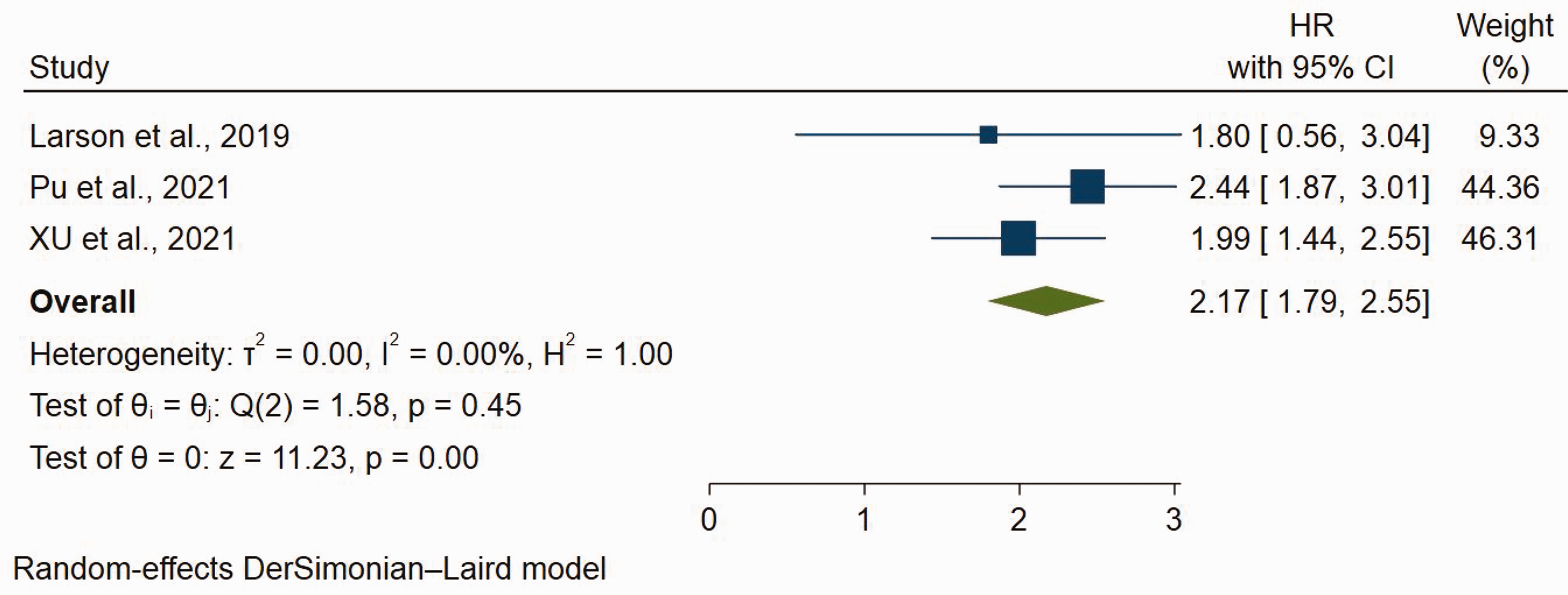
Forest plot of comparison of WPOI 1 to 3 and WPOI 4 to 5 in terms of OS rate in patients with OSCC. WPOI, worst pattern of invasion; OS, overall survival; OSCC, oral squamous cell carcinoma.
Disease-specific survival rates
Two studies reported data regarding the association between the WPOI and DSS rates.10,19 The pooled data showed that patients with a WPOI of 4 to 5 had worse DSS rates than those with a WPOI of 1 to 3 (HR, 4.67; 95% CI, 1.30–8.04; p < 0.001) (Figure 3). The pooled data were highly heterogeneous (I2 = 93.47%, p < 0.001).

Forest plot of comparison of WPOI 1 to 3 and WPOI 4 to 5 in terms of DSS rates in patients with OSCC. WPOI, worst pattern of invasion; DSS, disease-specific survival; OSCC, oral squamous cell carcinoma.
Disease-free survival rates
Four studies reported data regarding the association between the WPOI and the DFS rates.8,22,23,25 The overall effect estimates showed that patients with a high WPOI of 4 to 5 had worse DFS rates than those with a WPOI of 1 to 3 (HR, 1.58; 95% CI, 1.10–2.07; p < 0.001) (Figure 4). The pooled data were moderately heterogeneous (I2 = 68.6%, p = 0.02).

Forest plot of comparison of WPOI 1 to 3 and WPOI 4 to 5 in terms of DFS rates in patients with OSCC. WPOI, worst pattern of invasion; DFS, disease-free survival; OSCC, oral squamous cell carcinoma.
Locoregional recurrence rates
Two studies reported data regarding the association between the WPOI and LRR.10,19 The random-effects model of the pooled data showed a significant association between the WPOI and LRR (HR, 3.37; 95% CI, 2.62–4.12; p < 0.001) (Figure 5). The pooled data were homogeneous (I2 = 8.8%).

Forest plot of comparison of WPOI 1 to 3 and WPOI 4 to 5 in terms of LRR rates in patients with OSCC. WPOI, worst pattern of invasion; LRR, locoregional recurrence; OSCC, oral squamous cell carcinoma.
Locoregional recurrence-free survival rates
Two studies reported data regarding the association between the WPOI and locoregional recurrence-free survival (LRFS).5,18 The random-effects model of the pooled data showed a significant association between the WPOI and LRFS (HR, 1.68; 95% CI, 1.03–2.33; p < 0.001) (Figure 6). The pooled data were mildly heterogeneous (I2 = 46.52%).

Forest plot of comparison of WPOI 1 to 3 and WPOI 4 to 5 in terms of LRFS rates in patients with OSCC. WPOI, worst pattern of invasion; LRFS, locoregional recurrence-free survival; OSCC, oral squamous cell carcinoma.
Mortality rates
Results from two individual studies3,4 showed that a WPOI of 4 to 5 was associated with a higher mortality rate than a WPOI of 1 to 3, with an estimated HR of 3.86 (95% CI, 2.84–4.88) and 2.34 (95% CI, 1.72–2.96), respectively.
Discussion
The DOI is a significant predictor of the prognosis of OSCC, and multiple studies have established its association with lymph node metastasis and local recurrence and its use in guiding treatment plans.26–29 However, measurement of the DOI is limited to the postoperative pathological analysis, and preoperative imaging techniques may yield inaccurate results. By contrast, the WPOI can be observed microscopically at and beyond the invasive margin and serves as another valuable predictor of the prognosis of OSCC, including local recurrence and occult neck metastases.3,4,6,8,30 Recent research has suggested that patients with higher WPOI scores may require wider resection margins and neck dissection, even those with node-negative clinical disease.18,31 The present systematic review and meta-analysis was performed to determine the impact of the WPOI on the prognosis of OSCC by assessing the available literature. Our study included 18 eligible articles representing a total of 3954 patients. Based on the pooled results, patients with a high WPOI (4–5) exhibited worse OS, DSS, DFS, and LRFS rates and higher mortality and LRR rates when compared with patients with a low WPOI (1–3).
Several studies have explored the relationship between the WPOI and OS rates or DFS rates in patients with OSCC. In 2005, Brandwein-Gensler et al. 6 discovered that the WPOI independently predicted OS and DFS rates in a cohort of 103 patients with OSCC, with higher WPOI scores associated with reduced OS and DFS rates. In a multicenter study involving 311 patients with OSCC, the WPOI was found to be an independent prognostic factor for the OS and DFS rates, indicating its potential value in risk stratification and treatment decisions. 3 Furthermore, one study demonstrated that patients with a WPOI of 5 had a worse 5-year OS rate than those with a WPOI of 1 to 4 (HR, 321.70; 95% CI, 14.31–7228.42; p < 0.001), 20 whereas another reported a 70% mortality rate for patients with a WPOI of 5 compared with 26% for those with a WPOI of 1 to 4 (p < 0.01). 17 By contrast, another research group found no significant difference in the OS rate between patients with a WPOI of 1 to 4 and those with a WPOI of 5. 10 However, these researchers adjusted their analytical models for the DOI. When stratified by the DOI, a WPOI of 5 no longer showed a significant association with the LRR and DSS rates, nor did it predict higher rates of occult nodal metastases when analyzed separately from other histological parameters. 10
Our findings regarding the DFS rate were consistent with those of two other studies in which patients with OSCC who had a WPOI of 4 to 5 had worse DFS rates.22,23 However, two research groups found no significant difference in DFS between the two WPOI groups.8,25 One of these research groups reported that the intensity of tumor budding was more effective in predicting the DFS rate than the mode of invasion and WPOI. 25 The discrepancy between these studies concerning the impact of the WPOI on the DFS rate may be attributed to various factors, including differences in patient populations, methodologies, and focuses on specific tumor characteristics. This same research group found that the inclusion of only patients with early-stage OSCC (cT1,2N0M0) might have led to under-representation of patients with a WPOI of 5, skewing the results. 25 The focus on tumor budding intensity as a more effective predictor of DFS might have also contributed to the conflicting findings because this factor can provide additional insights into the tumor’s invasive behavior, which could influence the prognosis. Other possible explanations for the discrepancy could be the heterogeneity in how the WPOI was assessed across different studies as well as variations in the definitions and cut-offs used for categorizing WPOI groups. These inconsistencies might have led to variations in the reported outcomes, making direct comparisons between studies challenging. Moreover, some studies may not have considered potential confounding factors, such as patient demographics, tumor stages, and treatment modalities, which could influence the relationship between the WPOI and DFS rates. Therefore, it is essential to take these factors into account when interpreting the results and comparing different studies.
The ability of the WPOI to predict the LRR and LRFS rates in patients with OSCC has significant implications for clinical practice.5,10,18,19 LRR is a crucial determinant of patient outcomes because it often correlates with increased morbidity, diminished quality of life, and an elevated mortality risk. 6 However, longer LRFS is associated with a better prognosis, decreased morbidity, and improved quality of life. By accurately assessing the LRR and LRFS based on the WPOI, healthcare professionals can devise personalized treatment plans in which surgical interventions, adjuvant therapies, and follow-up schedules are individualized to cater to each patient’s unique needs. This approach may lead to enhanced patient outcomes, decreased recurrence rates, and a more comprehensive understanding of OSCC tumor biology. Our meta-analysis showed that the WPOI was effective in predicting the LRR and LRFS rates in patients with OSCC. These findings were consistent with those of individual studies.10,18,19 Only one study showed no significant difference in the LRFS rate between a WPOI of 4 to 5 and a WPOI of 1 to 3. 5 Integrating the WPOI into the routine evaluation of patients with OSCC may contribute to refinement of the staging system and improvement of the prognostic accuracy.3,24 This enhanced information could help identify high-risk patients who might benefit from more aggressive treatment strategies or closer monitoring.3,6 A deeper understanding of the relationship between the WPOI and prognostic parameters, especially LRR and LRFS, could also inspire new avenues of research, such as investigating the molecular mechanisms that drive OSCC tumor invasiveness.6,26 Ultimately, these insights could lay the groundwork for the development of innovative targeted therapies and preventive measures aimed at addressing the challenges of LRR in patients with OSCC.
This systematic review and meta-analysis had two main limitations that should be acknowledged. First, the heterogeneity observed in some of the pooled analyses may have impacted the precision of the effect estimates. This heterogeneity might have been due to differences in study designs, patient populations, tumor characteristics, and treatments received among the included studies. Second, the number of studies included in some of the analyses was relatively small, and this may limit the generalizability of the findings. In future research, it will be important to include a larger number of studies to strengthen the evidence supporting the impact of the WPOI on the prognosis of OSCC.
In conclusion, this systematic review and meta-analysis demonstrated that a higher WPOI was significantly associated with a worse prognosis in patients with OSCC, including the OS, DSS, DFS, LRR, LRFS, and mortality rates. These findings suggest that the WPOI could be an important prognostic factor in OSCC and may be useful for risk stratification and treatment decision-making. Incorporating the WPOI into the standard histopathological assessment of patients with OSCC may help to guide more personalized and effective treatment strategies, this improving patient outcomes.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231206260 - Supplemental material for Impact of worst pattern of invasion on prognosis of oral squamous cell carcinoma: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605231206260 for Impact of worst pattern of invasion on prognosis of oral squamous cell carcinoma: a systematic review and meta-analysis by Nada O. Binmadi and Yassmin A. Mohamed in Journal of International Medical Research
Footnotes
Acknowledgement
We would like to thank Dr. Bahbah Eshak for his help in the statistical analysis.
Author contributions
N.B. and Y.M. contributed to study design, data collection and analysis, and writing of the manuscript. N.B. supervised the data gathering, statistical analysis, and interpretation of the results. Both authors have read and approved the manuscript.
Data availability statement
The datasets analyzed in this study are available from the corresponding author upon request.
Declaration of conflicting interest
The authors declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
