Abstract
Objective
To evaluate clinical factors influencing the postoperative pulmonary sarcomatoid carcinoma (PSCs) prognosis.
Methods
We retrospectively evaluated patients with PSCs treated from October 2012 to October 2017. Kaplan–Meier survival curves were calculated using univariable analysis (log-rank test). Univariable/multivariable Cox regression analysis was also performed.
Results
Mixed PSCs were most common (64.10%). Pure PSCs occurred more often with large tumors compared with mixed PSCs. Patients with vs without pleural retraction, respectively, had significantly worse overall survival (OS; 16 vs 23 months) and disease-free survival (DFS; 11 vs 20 months), and patients with airway dissemination had significantly shorter OS (14 vs 21 months) and DFS (11 vs 20 months). Patients with PSC with an adenocarcinoma component had favorable OS. Airway dissemination, pleural retraction, metastatic mediastinal lymph node (LN) number, and pathological tumor-node-metastasis (pTNM) stage were risk factors for short OS. Neither adjuvant chemotherapy nor adjuvant radiotherapy provided a survival advantage. Airway dissemination was an independent prognostic factor (odds ratio, 1.87; 95% confidence interval, 1.04–3.36).
Conclusion
Pure PSCs were more likely with large tumors compared with mixed PSCs. Airway dissemination, pleural retraction, and metastatic mediastinal LN number were associated with OS. Airway dissemination was an independent prognostic factor.
Keywords
Introduction
Pulmonary sarcomatoid carcinoma (PSC) is a rare malignant neoplasm, accounting for less than 1% of all lung cancers.1,2 PSCs are aggressive tumors associated with an extremely poor prognosis compared with other stage-matched non-small cell lung cancers (NSCLC). The World Health Organization (WHO) classification system recognizes five subtypes of PSCs: pure spindle cell, pure giant cell, carcinosarcoma, pulmonary blastoma, and pleomorphic carcinoma. Standard effective treatment is very difficult to determine because of the low morbidity, high mortality, and heterogeneity associated with PSCs. Additionally, PSCs are typically resistant to chemotherapy and radiotherapy. 3 Currently, surgery in early-stage operable PSC has proven to provide the greatest overall survival (OS) benefit and remains the standard of care among eligible patients. 4 However, even after surgery, the 5-year survival rate reported for PSC patients varies between 11% and 24.5%,5–7 which is substantially worse than that for other subtypes of NSCLC, such as adenocarcinoma (ADC) or squamous cell cancer (SCC). Favorable OS rate stratification can be achieved in accordance with the tumor-node-metastasis (TNM) stage and surgical completeness. 3 Our aim was to identify additional high-risk factors that affect survival. In the present study, we performed a survival analysis using retrospective data to determine the survival effect of the combination of airway dissemination and pleural invasion in PSC.
Method
Data collection
Consecutive patients with PSCs who were pathologically diagnosed at Tianjin Cancer Hospital between 1 October 2012 and 1 October 2017 were included in this study. Patients who underwent surgical treatment (lobectomy or pneumonectomy) were included.
All patients’ clinical data were analyzed retrospectively. The follow-up data were collected by contacting patients or their relatives by telephone or from the hospital records. Our study was approved by the Ethics Committee of Tianjin Cancer Hospital (approval No. bc2022081, on 17 March 2022). The reporting of this study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
Diagnosis
All tumors were staged in accordance with the current eighth edition of the TNM system. 9 OS was defined as the time from beginning the first-line treatment to either the date of death or the date of the last follow-up visit. Disease-free survival (DFS) was defined as the time from surgery to progression or to death from any cause or the date of the last follow-up visit, whichever occurred first. Patients without tumor progression or death at the data cut-off date for the analysis or the date of additional anticancer therapy were censored as of their last date of tumor evaluation.
Statistical analysis
Statistical analysis was performed using the program “survival” in R software (version 4.1; www.r-project.org). The patients’ clinical characteristics were compared on the basis of pathological type using the Mann–Whitney U test for continuous data and the chi-square test or Fisher’s exact test for nominal data. Survival curves were estimated using the Kaplan–Meier (KM) method and curves were compared using the log-rank test. The KM survival curves were created using the “survminer” package. Univariable and multivariable Cox regression was used to explore the patients’ risk factors, and p < 0.05 was considered statistically significant.
Results
Baseline data
A total of 187 patients were diagnosed as having PSC during the study period. Male patients had a much higher susceptibility to developing PSC compared with female patients (male to female ratio, 123:64). The mean age at diagnosis was 63.3 ± 9.8 years. Of the included patients, 78 underwent surgery. Lobectomy was performed in 74 (94.87%) patients; 46/78 received adjuvant chemotherapy, and 28/78 patients received adjuvant radiotherapy. Sixty-three (80.8%) patients died, and 15 (19.2%) patients were still alive at the last follow-up. The median follow-up was 20 months (range, 5–60 months) for the surgical patients.
The patients’ clinical parameters are shown in Table 1. Most PSCs developed with other pathological components; 50 (64.10%) and 28 (35.90%) were mixed PSCs and pure PSCs, respectively. Patients smoking more than 20 pack-years (PY) accounted for the highest number of patients with PSCs (n = 52, 66.67%). Statistically significant differences were observed for pathological tumor (pT) stage (p = 0.028) and tumor diameter (p = 0.048) (Table 1), which suggested that pure PSCs were more likely with large tumors compared with mixed PSCs (4.50 cm vs 4.00 cm, respectively; Supplementary Fig. 1).
Patients’ baseline characteristics by pathological type.
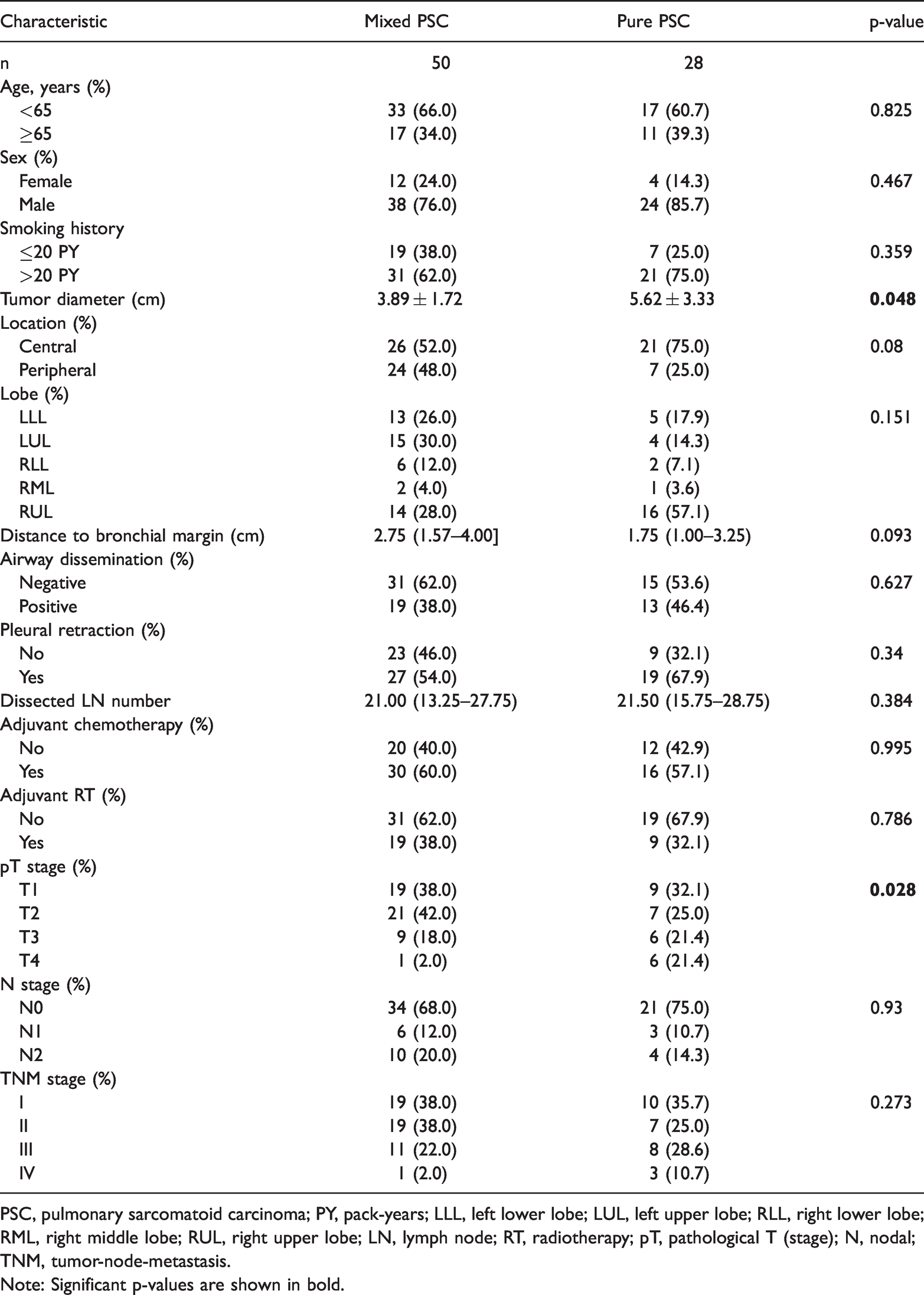
PSC, pulmonary sarcomatoid carcinoma; PY, pack-years; LLL, left lower lobe; LUL, left upper lobe; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe; LN, lymph node; RT, radiotherapy; pT, pathological T (stage); N, nodal; TNM, tumor-node-metastasis.
Note: Significant p-values are shown in bold.
Patient survival according to the risk factors
At the completion of the study, 25 (89.28%) patients with pure PSCs and 38 (76.00%) patients with PSCs with a mixed pathological component died owing to relapse or metastasis. The median OS was 16 months in the pure PSC group and 21 months in the mixed PSC group; however, the log-rank test showed no significant difference (Supplementary Fig. 2). The 5-year survival rate was 3.85%; only 3 patients survived >5 years, and 15.38% survived to 3 years.
Of the 78 patients who underwent surgery, 32 (41.03%) had airway dissemination, which indicated spread through air space (STAS). Patients with vs without airway dissemination, respectively, had significantly shorter OS (14 vs 21 months; p < 0.01; Supplementary Fig. 3a) and DFS. (11 vs 20 months; p < 0.01; Supplementary Fig. 3b). On the basis of computed tomography (CT) imaging, visceral pleural retraction occurred in 58.97% (46/78) of the PSC patients. Patients with pleural retraction had significantly worse OS (16 vs 23 months; p = 0.034) and DFS (11 vs 20 months; p = 0.022) compared with patients without pleural retraction (Supplementary Fig. 3c and d). Neither adjuvant chemotherapy nor adjuvant radiotherapy provided a significant survival advantage (Supplementary Fig. 3e–h), even after the entire cohort was stratified by disease stage.
Risk factor analysis for PSC
Univariable analysis indicated that patients with PSC with an adenocarcinoma component tended to have favorable OS (p = 0.04). Furthermore, the OS of PSC was significantly correlated with airway dissemination (p = 0.004), pleural retraction on CT imaging (p = 0.035), metastatic mediastinal lymph node (LN) number (p = 0.017), and pathological TNM (pTNM) stage. Age, sex, smoking history, tumor diameter, pure vs mixed PSC, central vs peripheral location, distance to the bronchial margin, and dissected LN number were not significantly correlated with OS in patients with PSC in the univariable analysis (Table 2). These statistically significant risk factors were further tested with multivariable Cox regression, which revealed that airway dissemination was an independent prognostic factor (odds ratio (OR), 1.87; 95% confidence interval (CI), 1.04–3.36; p = 0.036; Table 3).
Univariable analysis of OS.
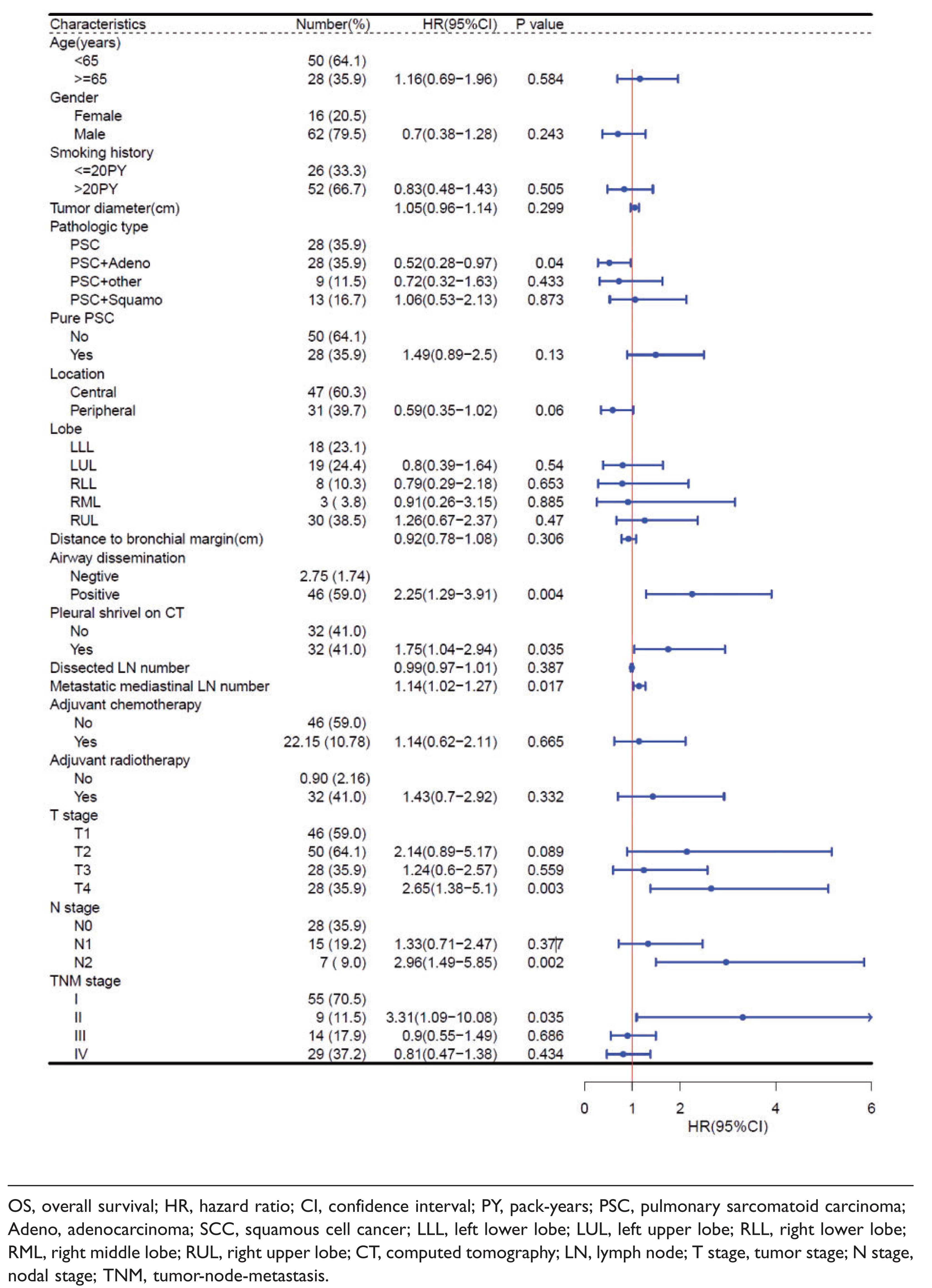
OS, overall survival; HR, hazard ratio; CI, confidence interval; PY, pack-years; PSC, pulmonary sarcomatoid carcinoma; Adeno, adenocarcinoma; SCC, squamous cell cancer; LLL, left lower lobe; LUL, left upper lobe; RLL, right lower lobe; RML, right middle lobe; RUL, right upper lobe; CT, computed tomography; LN, lymph node; T stage, tumor stage; N stage, nodal stage; TNM, tumor-node-metastasis.
Multivariable analysis of OS and DFS

OS, overall survival; DFS, disease-free survival; HR, hazard ratio; CI, confidence interval; PSC, pulmonary sarcomatoid carcinoma; Adeno, adenocarcinoma; SCC, squamous cell cancer; CT, computed tomography; LN, lymph node; TNM, tumor-node-metastasis.
Discussion
PSC is rare tumor with high aggressive and metastatic potential, an extremely poor prognosis, and resistance to cytotoxic chemotherapy or radiotherapy even after early-stage radical resection. 10 In this study, we analyzed the influence of different PSC clinical parameters on patients’ survival after surgery. Lococo et al 10 reported a 5-year survival rate for 148 curative resection PSC patients that ranged between 11% and 24.5%. In our study, the 5-year survival rate was 3.85%; only 3 patients survived >5 years, and 15.38% survived to 3 years. Surgery is still the first choice for early preoperative clinical patients with PSC; however, we consider that there must be patient-related factors that seriously affect the outcome and prognosis after surgery.
According to the 2015 WHO classification, STAS is defined as “micropapillary clusters, solid nests, or single cells spreading within air spaces beyond the edge of the main tumor”. 11 In addition to the existing criteria for invasion (histological subtype other than a lepidic pattern; myofibroblastic stroma associated with invasive tumor cells; vascular or pleural invasion), STAS was established as a fourth category that defines invasion for ADC. 12 A previous study also reported that STAS was associated with aggressive clinical pathological features in lung SCCs, and that STAS was an independent predictor of lung SCC. 13 The present study showed that 41.03% of the patients with PSCs had airway dissemination, which had a strong correlation with unfavorable OS. Dai et al 14 demonstrated that the presence of STAS in ADCs measuring >2 to 3 cm was associated with an increased risk of recurrence and a decreased survival rate. Additionally, Eguchi et al 15 revealed that the 5-year cumulative incidence of recurrence (CIR) and lung cancer-specific death (CID) in patients with ADCs measuring ≤2 cm was stratified by STAS. These results suggest that STAS could be considered a factor in upgrading the T stage, which contributes to the accuracy of predicting the prognosis of early-stage ADCs after resection). Vaghjiani et al 16 reported similar findings, stating that STAS was more common in patients with occult LN metastases (ONM) than in those without ONM (67% versus 39%, respectively; p < 0.001).
Huang et al 17 analyzed 32 studies of early-stage NSCLC and reported that visceral pleural invasion was associated with death. We found similar results in our study; patients with pleural retraction had significantly worse OS (16 vs 23 months; p = 0.034) and DFS (11 vs 20 months; p = 0.022) compared with patients without pleural retraction, respectively. The visceral pleura on the surface of the lung is rich in lymphatic networks and capillaries, and tumor invasion to this pleura can lead to distant metastasis through the lymph and blood vessels. This could explain why patients with pleural invasion have shorter survival times.
In this study, there was no significant difference in survival between patients with central vs peripheral tumors. However, the survival curves of the patients in the two location groups in this study showed a trend toward significance. Additionally, most of the patients in the central group died shortly after surgery due to in situ recurrence. We believe that the location of the tumor has an impact on patient survival in PSC, and that central tumors are more likely to spread and infiltrate the bronchi compared with peripheral tumors, leading to a higher rate of recurrence with central tumors.
In NSCLC, pathological stage II and IIIa patients receive platinum-based chemotherapy after complete surgical resection in accordance with the National Comprehensive Cancer Network (NCCN) guidelines. Several trials18–20 comparing cisplatin-based regimes versus observation have been published and proved the efficacy of adjuvant chemotherapy in these patients. In the present study, we aimed to evaluate the impact of postoperative chemotherapy on DFS in PSC patients with stage-matched disease. The median survival was not statistically different between the patients treated with surgery alone versus with surgery plus adjuvant chemotherapy. Maneenil et al 21 evaluated a large series of patients with PSC from the Mayo Clinic in 2017, and reported similar results, stating that PSCs did not benefit from adjuvant chemotherapy. Additionally, Karim et al 22 reviewed the outcomes of treatment in PSC patients at the University of Cincinnati Medical Center. The study showed that surgery remains the best option in early-stage disease (median survival, 713.5 days), and that patients who underwent surgery and adjuvant chemotherapy showed a trend toward improved survival (median, 457.6 days) over systemic chemotherapy; however, the results were inferior to those with surgery alone.
Immune checkpoint inhibitor immunotherapy has revolutionized the approach to metastatic NSCLC. For patients with unresectable stage III NSCLC, the use of durvalumab, a checkpoint inhibitor, after concurrent chemoradiotherapy has resulted in major improvement in 2-year progression-free survival and OS, which holds promise for an improved cure rate. 23 Recently Jin et al. 24 reported a case in which they administered nivolumab combined with anlotinib synchronously to a PSC patient with high programmed death-ligand 1 (PD-L1) expression who rapidly recurred during postoperative adjuvant chemotherapy. The patient’s clinical symptoms were gradually relieved, and response evaluation on imaging revealed a partial response after 8 weeks.
PSCs are rare subtypes of NSCLC with a poor prognosis, as suggested by the short OS. Adjuvant chemotherapy or adjuvant radiotherapy does not provide a significant survival advantage. Advanced pathological TNM stages and the presence of airway dissemination are independent poor prognostic factors. Considering the frequent presence of airway dissemination, surgeons should take care in recommending surgical resection for patients with PSCs, even for early-stage disease. As the administration of a checkpoint inhibitor appeared to help some patients, we await new, more effective systemic therapies in the future.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221128092 - Supplemental material for Clinical analysis of 78 pulmonary sarcomatoid carcinomas with surgical treatment
Supplemental material, sj-pdf-1-imr-10.1177_03000605221128092 for Clinical analysis of 78 pulmonary sarcomatoid carcinomas with surgical treatment by Ting Gong, Bin Jia, Chen Chen, Zhenfa Zhang, Changli Wang in Journal of International Medical Research
Footnotes
Author contributions
Study conception and design: Bin Jia, Zhenfa Zhang, Changli Wang
Collection and organizing the data: Ting Gong, Bin Jia, Chen Chen
Data analysis and interpretation: All authors
Manuscript writing: Ting Gong, Bin Jia
Final approval of the manuscript: All authors
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Foundation of Tianjin Municipal Education Commission [grant number 2020KJ156].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

