Abstract
Objective
To determine the relationship between use of nebulized heparin and clinical outcomes in mechanically ventilated patients.
Methods
The Medline, Embase, Web of Science, Cochrane Library, and PubMed databases were searched for relevant randomized controlled trials (RCTs), published between database inception and May 2022. Primary outcomes were intensive care unit (ICU) length of stay and in-hospital mortality; secondary outcomes included duration of mechanical ventilation, ventilator-free days (VFDs) in 28 days, and length of hospitalization. The study protocol was registered on PROSPERO (registration No: CRD42022345533).
Results
A total of eight RCTs (651 patients) were included. Nebulized heparin was associated with reduced ICU length of stay (six studies; mean difference [MD] –1.10, 95% confidence interval [CI] –1.87, –0.33, I2 = 76%), reduced duration of mechanical ventilation (two studies; MD –2.63, 95% CI –3.68, –1.58, I2 = 92%) and increased VFDs in 28 days (two studies; MD 4.22, 95% CI 1.10, 7.35, I2 = 18%), without increased incidence of adverse events, such as bleeding; but was not associated with a reduction in length of hospitalization (three studies; MD –1.00, 95% CI –2.90, –0.90, I2 = 0%) or in-hospital mortality (five studies; odds ratio 1.10, 95% CI 0.69, 1.77, I2 = 0%).
Conclusion
Nebulized heparin reduces ICU length of stay and duration of mechanical ventilation in mechanically ventilated patients, but has no effect on length of hospitalization or mortality.
Keywords
Introduction
Patients on mechanical ventilation often have some form of lung injury, including acute respiratory distress syndrome (ARDS), pneumonia, aspiration trauma, and ventilator-associated lung injury.1,2 Pulmonary coagulation dysfunction is a common feature of all forms of lung injury and activation of pulmonary coagulation is involved in the control of inflammation or infection at the site of injury, possibly as a host protective mechanism. 3 However, these localized coagulation disorders can also be detrimental, with fibrin deposition and hyaline membrane shapes being important early features of diffuse alveolar injury and a hallmark of ARDS. Excessive or persistent fibrin deposition may lead to impaired alveolar surfactant function, 4 pulmonary oedema, and impaired gas exchange, ultimately leading to alveolar collapse due to pulmonary fibrosis.
Pre-clinical studies have shown that nebulized inhalation or systemic use of anticoagulants can prevent lung injury in animals; 5 however, conflicting results have been revealed in clinical studies. A randomized controlled trial (RCT) of mechanically ventilated critically ill patients revealed that nebulized heparin was associated with a reduction in the number of days of mechanical ventilation; 6 however, another study showed that nebulized heparin did not reduce the duration of mechanical ventilation or the incidence of ventilator-associated pneumonia (VAP) and did not improve patient prognosis. 7 Increased concentrations of anticoagulants in the alveoli have been suggested to be required to treat pulmonary disease. 5 Therefore, topical application of anticoagulants to the alveoli may be an effective anticoagulant intervention.
Studies in healthy volunteers have shown that nebulized inhaled heparin reaches the lower airways, is evenly distributed in the lungs, and exerts a local anticoagulant effect. 8 Over the past few decades, nebulized inhaled heparin has been used safely in a variety of lung diseases; 9 it has been shown to reduce pulmonary coagulation dysfunction in patients with acute lung injury and its intrapulmonary mode of administration does not increase the risk of systemic bleeding.10,11 However, data on the feasibility and safety of nebulized inhaled heparin in mechanically ventilated patients are scarce. Whether nebulized inhaled heparin therapy is beneficial for all mechanically ventilated patients in the intensive care unit (ICU) remains unclear. Therefore, the aim of the present study was to conduct a meta-analysis of previously published RCTs investigating the use of nebulized heparin in mechanically ventilated patients in the ICU, to determine the relationship between nebulized inhaled heparin and clinical outcomes in mechanically ventilated patients.
Materials and methods
This systematic evaluation and meta-analysis was performed based on the Preferred Reporting Items for Systematic Evaluation and Meta-Analysis (PRISMA) 2020 statement. 12 The protocol for this study was registered on PROSPERO (registration number: CRD42022345533) and study results were reported using the PRISMA checklist (Supplemental Table 1).
Search strategies by database.

Indexes: SCI-EXPANDED, SSCI, AHCI, CPCI-S, CPCI-SSH, BKCI-S, BKCI-SSH, ESCI, CCR-EXPANDED, IC ARDS, acute respiratory distress syndrome; ALI, acute lung injury; Mesh, medical subject headings; TX, all text; SCI-EXPANDED, science citation index - expanded; SSCI, social sciences citation index; AHCI, arts humanities citation index; CPCI-S, conference proceedings citation index - science; CPCI-SSH, conference proceedings citation index - social science; BKCI-S, book citation index - science; BKCI-SSH, book citation index - social science; ESCI, emerging sources citation index; CCR-EXPANDED, current chemical reactions - expanded; IC, index chemicus.
Search strategy and study selection
Two researchers (YZ and JL) systematically searched the Medline, Embase, Web of Science, Cochrane Library, and PubMed databases for RCTs published from database inception until May 2022. The search strategy included terms such as ARDS, acute lung injury, mechanical ventilation and nebulized heparin (Table 1). The reference lists of the included articles were also hand searched for any eligible trials. The search was conducted for English language articles only.
Once duplicates had been excluded, two investigators (YZ and JL) independently screened the titles and abstracts to determine applicability based on the outcome metrics. The full text of relevant articles was then searched and analysed to clarify their eligibility for inclusion. In case of controversy, a third investigator (ZJQ) was included for discussion to resolve the disagreement.
The following inclusion criteria were applied: (1) RCT design; (2) comparison of nebulized heparin with nebulized placebo or conventional therapy; (3) study population comprised patients who were receiving invasive mechanical ventilation in the ICU; and (4) RCT provided at least one primary or secondary outcome indicator. Case series, case studies, cohort studies, editorials, practice guidelines, and other observational studies were excluded. Articles or conference reports that were available only in abstract form were also excluded. Detailed inclusion and exclusion criteria, including outcome variables, are shown in Table 2.
PICOS criteria for inclusion and exclusion of studies into meta-analysis.
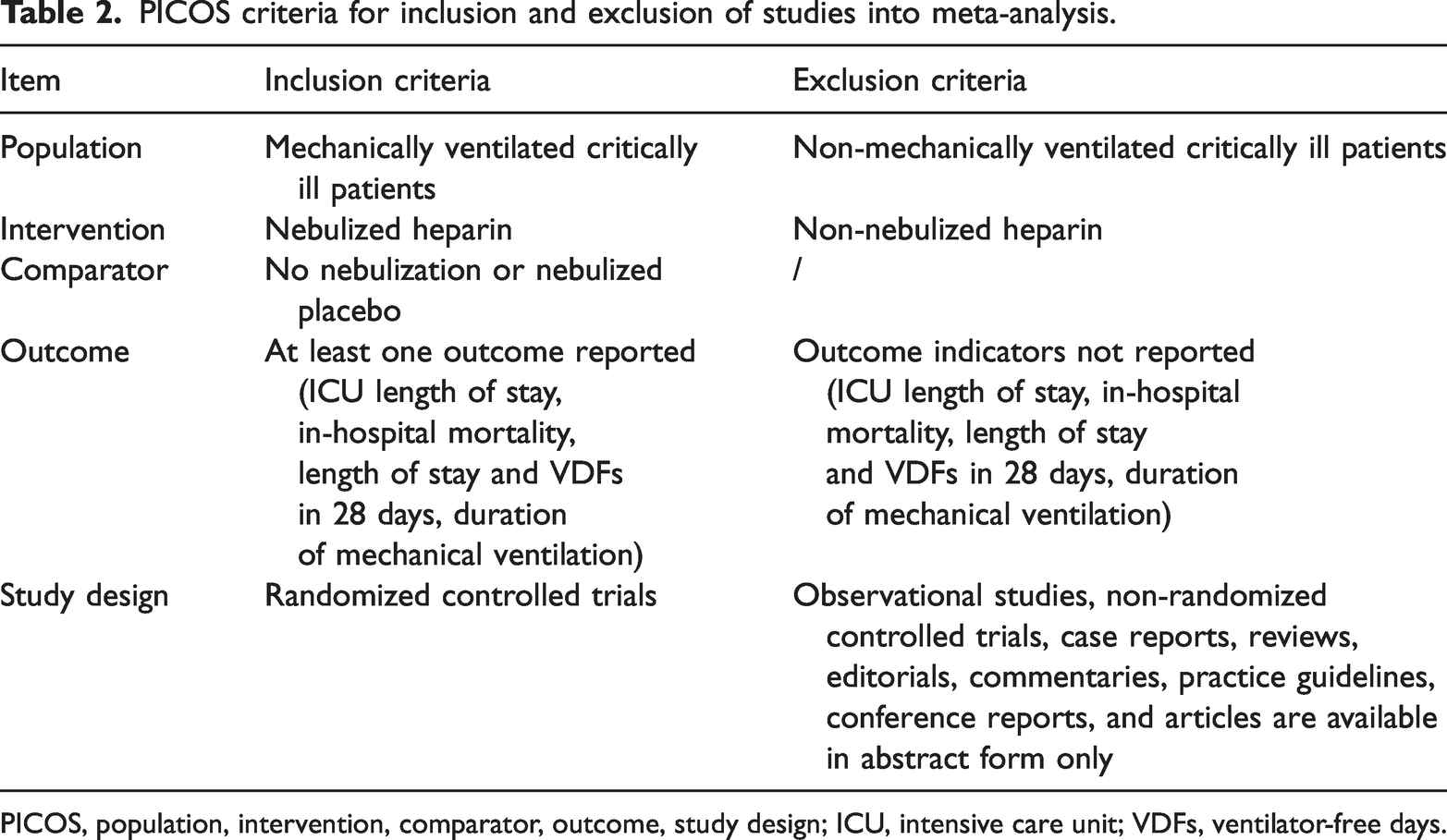
PICOS, population, intervention, comparator, outcome, study design; ICU, intensive care unit; VDFs, ventilator-free days.
Data collection and quality assessment
Two researchers (YZ and JL) collected data independently using a pre-developed data extraction form. The following data were collected: first author; year; study site, type, and population; number of patients; interventions (dose and frequency of nebulized heparin); primary and secondary outcome indicators; and occurrence of adverse events. Disagreements were resolved through discussion and negotiation. Attempts were also made to contact the corresponding authors of the study to request unreported or insufficient data in the main publication.
Risk of bias assessment
Two investigators (YZ and JL) independently used the Cochrane Risk of Bias tool to assess risk of bias in the included RCTs, using seven domains: random sequence generation, allocation method, allocation scheme concealment, blinding, completeness of outcome data, selective reporting of study results, and other sources of bias. Each potential source of bias was marked as high, low, or unclear.
Definitions and outcomes
Primary outcomes were: (1) ICU length of stay – defined as length of stay in the ICU for the first time after patient admission; and (2) in-hospital mortality. Secondary outcomes included: (1) ventilator-free days (VFDs) in 28 days; (2) duration of mechanical ventilation; and (3) length of hospitalization.
Statistical analyses
Meta-analysis was performed using Review Manager 5.4 (RevMan, Cochrane Collaboration, Oxford, UK) and STATA software, version 17.0 (Nordic Cochrane Centre, Cochrane Collaboration, Copenhagen, Denmark). Estimates of the combined continuous and dichotomous values are presented as mean difference (MD) or dominance ratio (odds ratio [OR]), respectively, with 95% confidence intervals (CIs). When continuous values were presented in the RCT results as median and interquartile range (IQR), the mean ± SD values were calculated according to the method of Wan et al. 13 Statistical heterogeneity was assessed using I2, with I2 values > 0%, > 30%, > 50% and > 75% indicating low, medium, high, and considerable heterogeneity, respectively. A P value < 0.05 was considered statistically significant.
Results
Literature search
The study selection process is summarised in Figure 1. The search identified a total of 298 studies, 53 of which were found to be duplicates and removed. After screening titles and abstracts, 203 records were excluded, leaving 42 records for full-text review. A further 34 studies were excluded and eight completed trials were eligible for inclusion in the present meta-analysis. The number of results from each search is detailed in Table 1.

Flow diagram of study selection.
Study characteristics and risk of bias
The characteristics of the included studies are summarized in Table 3.6,7,14–19 All studies were prospective randomized parallel-group controlled trials involving a total of 651 patients on mechanical ventilation (329 [intervention group] and 322 [control group]). A total of four of the eight studies were conducted in Australia and one each in Ireland, Iran, Netherlands, and Egypt. Nebulized heparin was administered at doses of 10 000 IU/4 h (n = 1), 25 000 U/4 h or/6 h (n = 4), 5 000 u/q12 h or/q6 h (n = 2), or 50 000 U administered as a single dose (n = 1). The number of patients in each trial ranged between 13 and 252. The maximum follow-up period among the trials was 90 days.
Characteristics of eight randomized controlled trials included in the systematic review and meta-analysis.
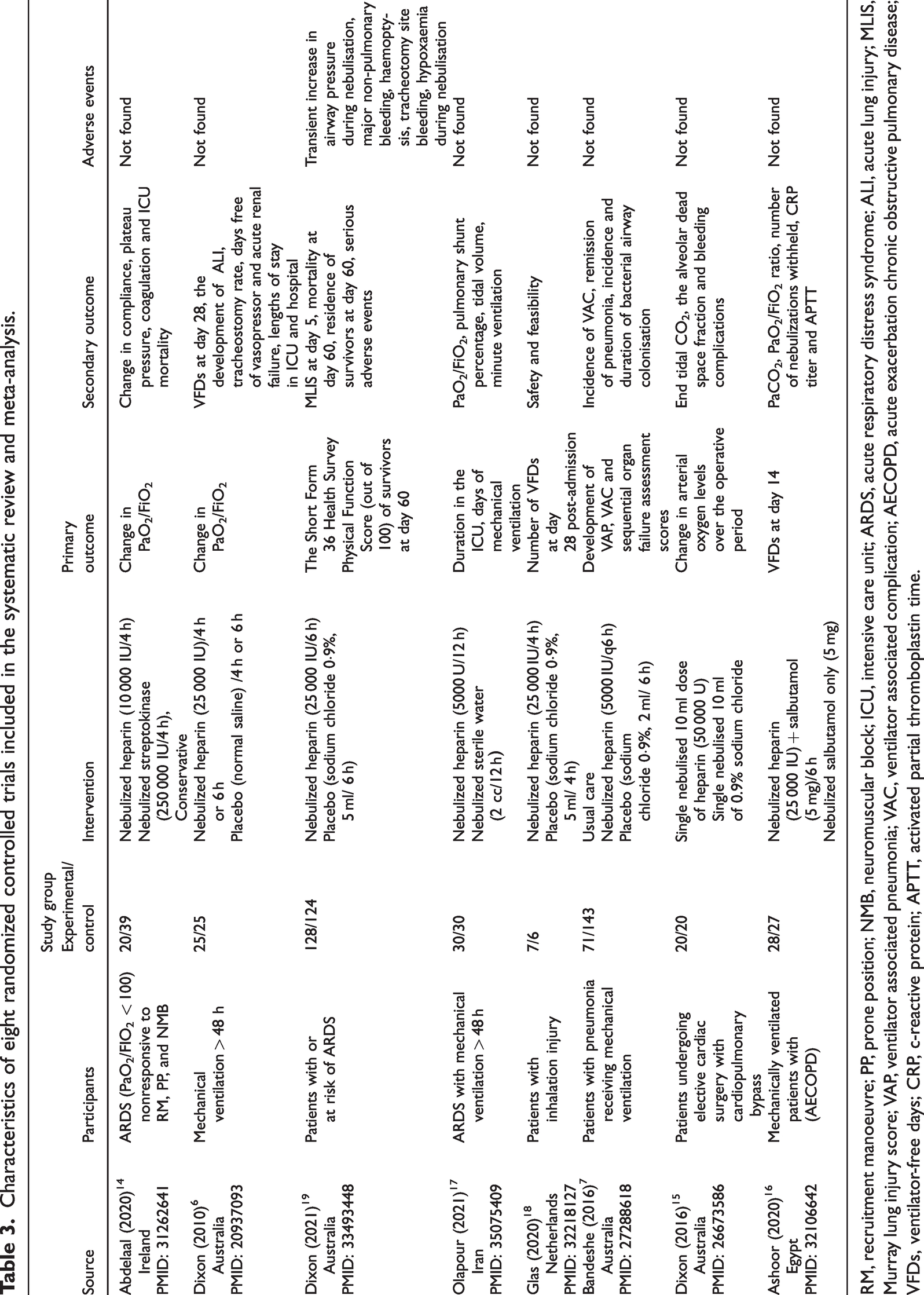
RM, recruitment manoeuvre; PP, prone position; NMB, neuromuscular block; ICU, intensive care unit; ARDS, acute respiratory distress syndrome; ALI, acute lung injury; MLIS, Murray lung injury score; VAP, ventilator associated pneumonia; VAC, ventilator associated complication; AECOPD, acute exacerbation chronic obstructive pulmonary disease; VFDs, ventilator-free days; CRP, c-reactive protein; APTT, activated partial thromboplastin time.
The risk of bias assessment is summarised in Figure 2. Four trials had a low overall risk of bias. Four trials were judged to be at high risk of bias in at least one domain. After an evaluation of outcome indicators, six trials had an unclear risk of bias regarding detection bias. One trial was considered to have an unclear risk of bias in two areas, three trials had a high risk of bias for incomplete outcome information, two trials had a high risk of bias for reporting bias, and three trials had an unclear or high risk of bias in other areas.

Graph showing risk-of-bias in the eight randomised controlled trials included in the meta-analysis (+, low risk of bias; ?, unclear risk of bias; –, high risk of bias).
Primary outcome
ICU length of stay
Seven trials reported ICU length of stay; however, one study reported data on days surviving 90 days and not in the ICU, and was excluded. Six studies were included in the statistical analysis (n = 582 patients), which revealed that patients in the heparin group had a shorter ICU length of stay than controls (six studies, MD –1.10, 95% CI –1.87, –0.33, P = 0.005, I2 = 76%), with statistically significant results but with high heterogeneity (Figure 3a).

Forest plots of research findings: (a) intensive care unit length of stay; (b) in-hospital mortality; (c) ventilator-free days in 28 days; (d) duration of mechanical ventilation; and (e) length of hospital stay. OR, odds ratio; CI, confidence interval; MD, mean difference.
In-hospital mortality
Mortality was reported in five trials (n = 544 patients). Nebulized heparin had no effect on mortality (OR 1.10, 95% CI 0.69, 1.77, P = 0.69, I2 = 0%; Figure 3b). There was no significant publication bias in the funnel plot estimates.
Secondary outcomes
Number of ventilator-free days in 28 days
Two trials reported VDFs in 28 days (n = 63 patients). Pooled analysis showed that the use of nebulized heparin increased the number of VDFs in 28 days in mechanically ventilated patients (MD 4.22, 95% CI 1.10, 7.35, P = 0.008, I2 = 18%; Figure 3c), with low heterogeneity and no significant publication bias in funnel plot estimates.
Duration of mechanical ventilation
Three studies provided data on the duration of mechanical ventilation. One study presented mechanical ventilation time in hours and was excluded. Two studies were included in the statistical analyses (n = 202 patients) and showed that nebulized heparin reduced the duration of mechanical ventilation in patients compared with placebo (MD –2.63, 95% CI –3.68, –1.58, P < 0.05, I2 = 92%; Figure 3d); however, there was considerable heterogeneity in the results. Due to the small number of studies, the source of heterogeneity could not be identified.
Length of hospitalization
Length of hospitalization was reported in three trials (n = 232 patients). Pooled analysis showed no effect of nebulized heparin use on length of hospitalization (MD –1.00, 95% CI –2.90, –0.90, P = 0.30, I2 = 0%; Figure 3e), with low heterogeneity.
Adverse event profile
Adverse events occurred in the trial by Dixon et al. (2021), 19 comprising heparin sodium (25 000 IU/q6 h) in the trial group and 0.9% sodium chloride (5 ml/q6 h) in the control group. Transient increases in airway pressure during nebulization (n = 3 in the heparin group), major non-pulmonary bleeding (n = 2 in each group), haemoptysis (n = 1 in the heparin group), tracheotomy site bleeding (n = 1 in the heparin group), and hypoxaemia during nebulization (n = 1 in the placebo group), were not confirmed to be associated with nebulized heparin in the study. The remaining trials did not identify or report adverse events with nebulized heparin.
Sensitivity analysis and publication bias
Sensitivity analyses were conducted by deleting the data of one article at a time for results that included ≥ 3 studies, and by changing the merging model for results that included < 3 studies (Figure 4). The results showed that the conclusions of the meta-analyses of ICU length of stay, in-hospital mortality, and length of hospital stay were reliable (Figure 4a–c). Analyses of the duration of mechanical ventilation and VDFs in 28 days yielded inconsistent results using fixed-effects and random-effects models, thus, any conclusions should be applied with caution (Figure 4d and e).

Sensitivity analyses of different research findings: (a) intensive care unit length of stay; (b) in-hospital mortality; (c) length of hospital stay; (d) ventilator-free days in 28 days; and (e) duration of mechanical ventilation. OR, odds ratio; CI, confidence interval; MD, mean difference.
Assessment of publication bias revealed largely symmetrical scatter, as shown in the funnel plots of the study results (Figure 5). However, publication bias in literature reviews is inevitable and difficult to overcome, and should be taken into account when interpreting the results.

Funnel plots of publication bias related to different research parameters: (a) intensive care unit length of stay; (b) in-hospital mortality; (c) ventilator-free days in 28 days; (d) duration of mechanical ventilation; and (e) length of hospital stay. SE, standard error; OR, odds ratio; MD, mean difference.
Subgroup analysis of vibrating mesh nebulizer
Four of the included studies applied vibrating mesh nebulization, one study used jet nebulization, one study used a mixture of jet nebulization and vibrating mesh nebulization, and the nebulization modality was unavailable in the other two studies. Subgroup analyses of the different nebulization modalities showed that a vibrating mesh nebulizer did not reduce ICU length of stay, in-hospital mortality, or length of hospitalization (Figure 6).

Forest plots of subgroup analysis: (a) intensive care unit length of stay; (b) in-hospital mortality; (c) length of hospital stay. CI, confidence interval; SD, standard deviation.
Discussion
In this RCT meta-analysis, nebulized inhaled heparin was found to improve most outcome indicators in mechanically ventilated patients. Patients who received nebulized heparin demonstrated shorter duration of mechanical ventilation and ICU length of stay and more VDFs in 28 days, but no effect on patient mortality or hospital length of stay was observed. The study also demonstrated that use of nebulized heparin in mechanically ventilated patients did not increase the incidence of adverse events, such as bleeding.
In the present study, nebulized inhaled heparin was found to reduce ICU length of stay in mechanically ventilated patients. This may be related to the fact that nebulized heparin reduces lung inflammation and decreases the duration of mechanical ventilation. An important inflammatory mechanism of lung injury is fibrin deposition (hyaline membrane formation) in the pulmonary microcirculation and alveolar sacs, which impairs alveolar perfusion and ventilation. Heparin may improve clinical outcomes by reducing fibrin deposition in the lungs, 20 and it also inhibits adhesion of bacteria and viruses on the respiratory surface, thereby, inhibiting their growth in the lungs.17,20 However, the results of some studies showed no significant effect of inhaled heparin on the duration of ICU stay, which may be due to heterogeneity of the studies. The population of these studies mainly consisted of patients with airway burns,18,21 while some were non-RCT studies, and their findings need further validation.
In the present meta-analysis, nebulized heparin was found to reduce the duration of mechanical ventilation in patients and increase the number of VDFs in 28 days, further supporting the outcome that nebulized heparin reduces ICU length of stay. Previous studies have reported mixed conclusions regarding nebulized heparin reducing the duration of mechanical ventilation; the presence of a dose-dependent effect of nebulized heparin has been suggested, with one investigator using a dose of 30 000 U/day for improving outcomes in paediatric patients, 22 but another showing it failed to improve outcomes in adults, 23 while higher doses have shown improved outcomes in adult patients. 21 Administration of a 20 000 U/day dose did not reduce the duration of mechanical ventilation in patients in one of the studies included in the present meta-analysis; 7 similarly, administration of a 15 000 U/day dose did not reduce the duration of mechanical ventilation in patients in another of the included trials. 18 Therefore, we believe that heparin has a dose-dependent effect and that neither insufficient nor excessive doses of heparin are beneficial for patients; different doses and frequencies of heparin need to be selected for patients with different aetiologies, ages, and body surface areas.
The effect of nebulization modality on clinical outcomes in mechanically ventilated patients was assessed in the present study. Subgroup analyses showed that vibrating mesh nebulization did not reduce ICU length of stay, in-hospital mortality, and length of hospital stay. Currently, commonly used nebulization modalities include jet nebulization, ultrasonic nebulization, and vibrating mesh nebulization, each with its own advantages and disadvantages. 24 The therapeutic effect of different nebulization modalities on nebulized heparin is unknown and needs to be confirmed by future RCTs.
In the present study, nebulized heparin had no effect on mortality and length of hospitalization in mechanically ventilated patients. A total of four articles (n = 381 patients) were included in this meta-analysis, of which three studies (n = 342 patients) showed no effect of nebulized heparin on mortality, which may imply that critical illness in patients is often accompanied by severe systemic pathophysiological alterations, such as in the cardiovascular system, and immunosuppression. Local nebulized heparin may only have a better effect on local lung inflammation and coagulation, but not decisively on the pathophysiological alterations of the patient’s system. Therefore, nebulized inhaled heparin may not improve patient prognosis.
Adverse events caused by nebulized heparin, which occurred in the test and control groups in the trial by Dixon et al. (2021), 19 were also analysed. Transient increases in airway pressure during nebulization, non-pulmonary bleeding, haemoptysis, tracheotomy site bleeding, and hypoxemia during nebulization were not associated with the use of nebulized heparin as assessed by the investigators. 19 No adverse events with nebulized heparin were reported in any of the other included studies.6,7,14–18 In a previous systematic review, nebulized inhaled heparin was also not found to be associated with an increased risk of bleeding, 25 and similarly, in a study addressing the safety and potential anticoagulant effects of nebulized inhaled heparin, no increased risk of bleeding associated with nebulized inhaled heparin was found. 11 Hence, we conclude that nebulized inhaled heparin is safe at the current doses, which supports the beneficial nature of nebulized heparin for the clinical management of patients.
There are some limitations to the present meta-analysis. First, substantial heterogeneity was observed among the included trial participants, and the limited number of previously published trials factored against further subgroup analysis for aetiology and dose of nebulized heparin. Secondly, the doses of nebulized heparin used in the included studies also varied, which may have been associated with different clinical effects. Thirdly, the present meta-analysis focused on the effect of nebulized heparin on the clinical management of mechanically ventilated patients and did not include other anticoagulants for comparison; hence, the potential efficacy and safety of any other anticoagulant could not be determined.
Conclusion
Nebulized inhaled heparin reduced the duration of mechanical ventilation and shortened ICU length of stay in mechanically ventilated patients without increasing the incidence of adverse events, such as bleeding; however, it had no effect on patient mortality or length of hospitalization.
Footnotes
Author contributions
Jun Li and Zhijiang Qi designed this study, and contributed to interpretation of findings. Yi Zhang participated in literature search and data analysis, and drafted the manuscript. Qiankun Li participated in literature search and data analysis. Changan Sun and Yue Gu contributed to literature screening and quality assessment.
Data availability statement
All data that support the study’s results are available upon request from the corresponding author.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
