Abstract
Objectives:
Platinum compounds cause several adverse events, such as nephrotoxicity, gastrointestinal toxicity, myelosuppression, ototoxicity, and neurotoxicity. We evaluated the incidence of renal impairment as adverse events are related to the administration of platinum compounds using the Japanese Adverse Drug Event Report database.
Methods:
We analyzed adverse events associated with the use of platinum compounds reported from April 2004 to November 2016. The reporting odds ratio at 95% confidence interval was used to detect the signal for each renal impairment incidence. We evaluated the time-to-onset profile of renal impairment and assessed the hazard type using Weibull shape parameter and used the applied association rule mining technique to discover undetected relationships such as possible risk factor.
Results:
In total, 430,587 reports in the Japanese Adverse Drug Event Report database were analyzed. The reporting odds ratios (95% confidence interval) for renal impairment resulting from the use of cisplatin, oxaliplatin, carboplatin, and nedaplatin were 2.7 (2.5–3.0), 0.6 (0.5–0.7), 0.8 (0.7–1.0), and 1.3 (0.8–2.1), respectively. The lower limit of the reporting odds ratio (95% confidence interval) for cisplatin was >1. The median (lower–upper quartile) onset time of renal impairment following the use of platinum-based compounds was 6.0–8.0 days. The Weibull shape parameter β and 95% confidence interval upper limit of oxaliplatin were <1. In the association rule mining, the score of
Conclusion:
Our findings suggest a potential risk of renal impairment during cisplatin use in real-world setting. The present findings demonstrate that the incidence of renal impairment following cisplatin use should be closely monitored when patients are hypertensive or diabetic, or when they are co-administered furosemide, loxoprofen, or pemetrexed. In addition, healthcare professionals should closely assess a patient’s background prior to treatment.
Keywords
Introduction
Platinum-based compounds that are widely used in the treatment of testicular, ovarian, breast, cervical, bladder, and lung cancers include cisplatin, carboplatin, oxaliplatin, and nedaplatin.1-3 These compounds cause adverse events (AEs) such as nephrotoxicity, gastrointestinal toxicity, myelosuppression, ototoxicity, and neurotoxicity. Although platinum-based compounds have some structural similarities, their AE profiles differ. Cisplatin causes severe renal tubular damage and reduces glomerular filtration. 3 One of the dose-limiting AEs of cisplatin is nephrotoxicity. Among the platinum-based compounds approved for use, cisplatin causes the most severe nausea and vomiting, which are usually prevented or managed with current antiemetic regimens.4,5 Carboplatin is a second-generation platinum-based drug. It is a prodrug of cisplatin and a more stable platinum-based analog than cisplatin. 6 Carboplatin-treated patients experience lower incidences of nausea, vomiting, and renal toxicity than cisplatin-treated patients.6,7 Nedaplatin is significantly less nephrotoxic than cisplatin or carboplatin.8,9 Oxaliplatin is a third-generation platinum drug that is generally used for standard treatment together with 5-fluorouracil/leucovorin. 10 The incidence of neurotoxicity resulting from the co-therapy increases with the addition of oxaliplatin. 10 Therefore, the benefits of these frequently prescribed drugs are compromised by the severe AEs they cause.
The analysis of spontaneous reporting systems (SRSs) has served as a valuable tool in post-marketing surveillance that reflects the realities of clinical practice. The Pharmaceuticals and Medical Devices Agency (PMDA), a regulatory authority in Japan, receives voluntary AE reports directly from healthcare professionals and consumers, and has released the Japanese Adverse Drug Event Report (JADER) database as an SRS. The JADER database files are openly available on the PMDA website (www.pmda.go.jp). Several pharmacovigilance indices, such as reporting odds ratio (ROR), have been developed for the detection of drug-associated AEs. 11 It has been proposed that the time-to-onset analysis using the Weibull shape parameter (WSP) of AEs could be a useful tool for signal detection.12–19 Furthermore, association rule mining has been proposed as a new analytical approach for discovering undetected relationships such as the possible risk factors between variables in large databases.18–22
In this study, we aimed to assess renal impairment (RI) caused by platinum-based compounds by analyzing data from the JADER database. Analyses of the time to onset of RI using the JADER database are rare, and to the best of our knowledge, this is the first study to use association rule mining to detect the association rules between platinum-based compounds and RI.
Materials and methods
Data from April 2004 to November 2016 were extracted from the JADER database on the PMDA website (www.pmda.go.jp). The data comprised cases mainly spontaneously reported by pharmaceutical industries, healthcare professionals, and consumers. All data from the JADER database were fully anonymized by the PMDA before we used them. The database consists of four tables: patient demographic information such as sex, age, and reporting year (DEMO); drug information such as drug name and start and end dates of administration (DRUG); AEs and onset date (REAC); and primary disease (HIST). We built a relational database that integrated the four tables using FileMaker Pro 12 software (FileMaker, Inc., Santa Clara, CA, USA). Four platinum-based compounds (cisplatin, oxaliplatin, carboplatin, and nedaplatin) were assessed in the analysis. In case of drug involvement, drugs reported as the DRUG file contained the following role codes assigned to each drug: suspected drug, concomitant drug, and interacting drugs (
Preferred terms (PTs) from the Medical Dictionary for Regulatory Activities (http://www.meddra.org/, version 19.0) were used to define medical terminologies in the JADER database. The following six PTs were used to extract cases of platinum compound–induced RI from the JADER database: “acute kidney injury,” “renal impairment,” “renal failure,” “renal disorder,” “renal function test abnormal,” and “renal tubular disorder.”
We used ROR to analyze the association between the use of platinum-based compounds and RI. ROR represents the odds of a specific AE caused by the drug of interest compared to the odds of a specific AE caused by all other drugs, and is calculated based on the two-by-two contingency table (Figure 1). 23 RORs are expressed as point estimates with 95% confidence intervals (CIs). The signal was considered positive when the lower limit of 95% CI was >1 and the number of reports was ≥2.23,24

Two-by-two contingency table for analysis.
Time-to-onset duration was calculated from the time of the patient’s first prescription to the occurrence of RI. The records with completed AE occurrence and prescription start date were used for the time-to-onset analysis. It was necessary to consider right truncation when evaluating the time to onset of AEs. We determined an analysis period of 90 days after the start of administration to focus on the onset of AEs within 3 months after the patients’ first prescription. The median duration, quartiles, and WSPs were used to evaluate the time-to-onset data. The scale parameter α of the Weibull distribution determines the scale of the distribution function. A larger scale value (α) stretches the distribution, whereas a smaller scale value (α) shrinks the data distribution. The shape parameter β of the Weibull distribution determines the shape of the distribution function. Larger and smaller shape values produce left- and right-skewed curves, respectively.
In the analysis of SRS, the shape parameter β of the Weibull distribution was used to indicate the level of hazard over time without a reference population. When β was 1 (random failure type), the hazard was considered to be constant over time. When β was >1, the hazard was considered to increase over time (wear-out failure type). In contrast, when β was lower than 1, the hazard was considered to decrease over time (initial failure type).12–19
The time-to-onset analysis was performed using the JMP software version 11 (SAS Institute, Cary, NC, USA).
Association rule mining is focused on finding frequent co-occurring associations among a collection of items. Given a set of transactions
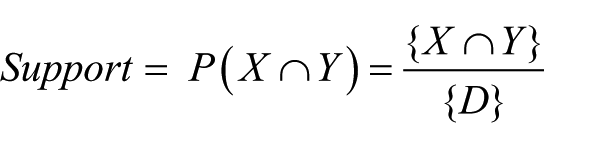
where
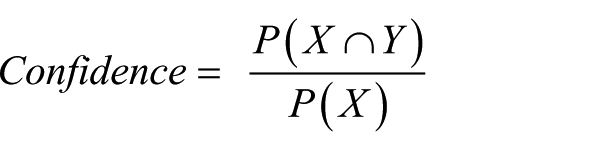
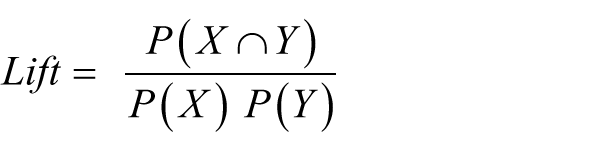
When the
The association rule mining was performed using the
Results
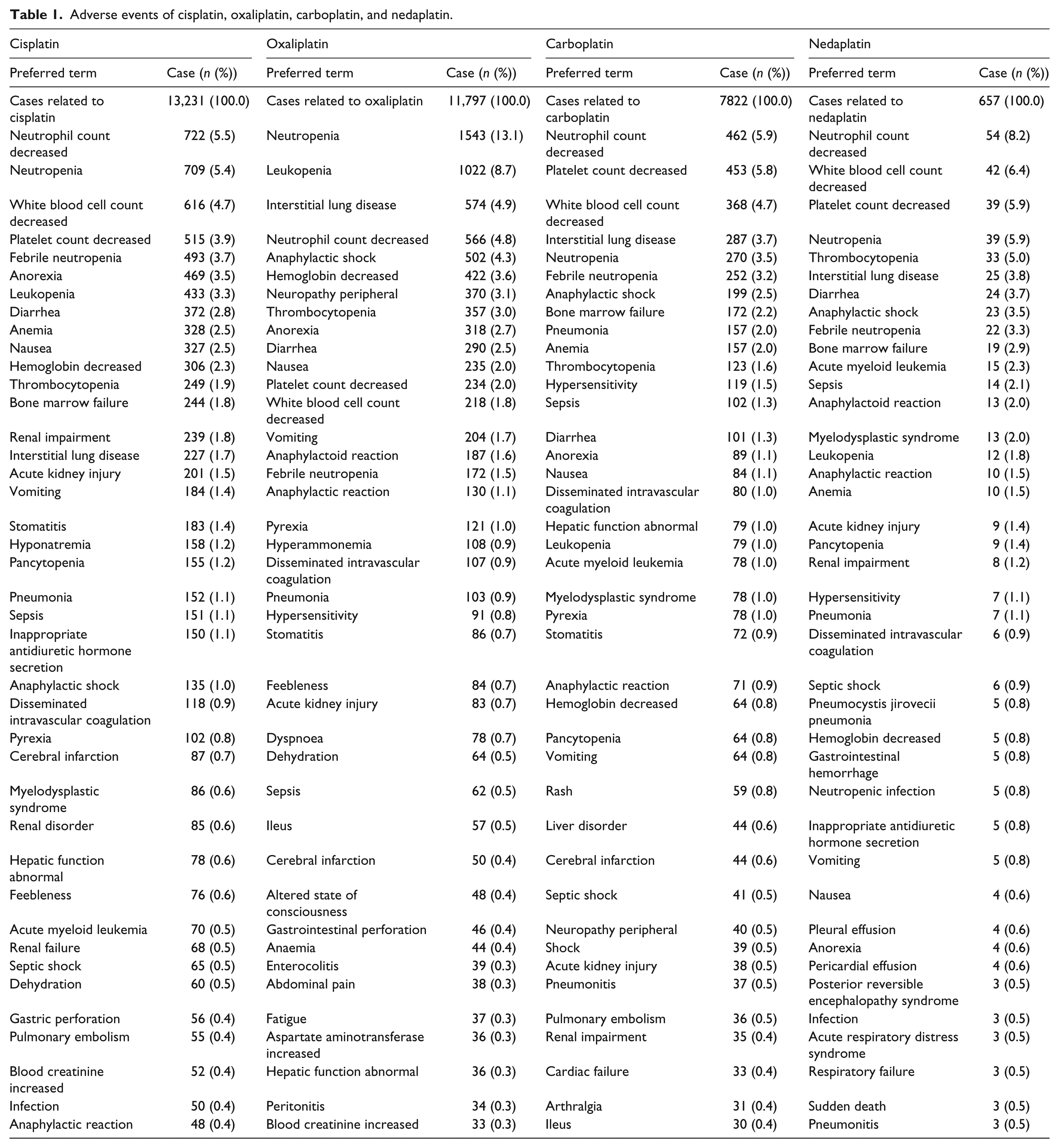
The JADER database contained 430,587 reports from April 2004 to November 2016. The number of cases of RI incidences was 14,872, and the cases related to the use of platinum-based compounds are summarized in Table 1. The table lists the 50 largest PTs in the reporting of the number of AEs. Cisplatin caused the highest number of RI events (“renal impairment” and “acute kidney injury”) among the four platinum-based compounds studied. The RORs (95% CI) for RI following the use of cisplatin, oxaliplatin, carboplatin, and nedaplatin were 2.7 (2.5–3.0), 0.6 (0.5–0.7), 0.8 (0.7–1.0), and 1.3 (0.8–2.1), respectively (Table 2). The lower limit of the ROR (95% CI) for cisplatin was >1.
Adverse events of cisplatin, oxaliplatin, carboplatin, and nedaplatin.
Number of reports and the ROR for renal impairment by platinum-based compounds.

ROR: reporting odds ratio; CI: confidence interval.
The median (lower–upper quartile) onset time of RI after the use of platinum-based compounds was 6.0–8.0 days (Table 3 and Figure 2). We noted that 58.9% (313 out of 532 cases) of RI events were observed within 7 days of drug administration; however, 41.1% were reported after 7 days of drug administration. The WSP β and 95% CI upper limit of oxaliplatin were <1, indicating a significant association between oxaliplatin and RI.
The medians and Weibull parameter of each drug for renal impairment.
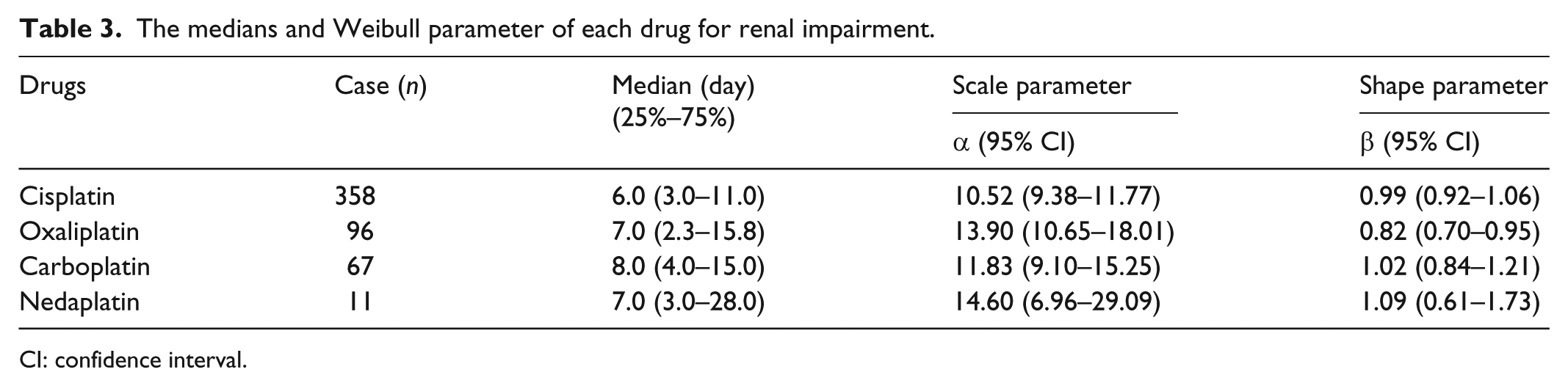
CI: confidence interval.

Histogram and Weibull shape parameter of renal impairment for (a) cisplatin (β = 0.99 (95% CI: 0.92–1.06)), (b) oxaliplatin (β = 0.82 (95% CI: 0.70–0.95)), (c) carboplatin (β = 1.02 (95% CI: 0.84–1.21)), and (d) nedaplatin (β = 1.09 (95% CI: 0.61–1.73)).
We evaluated the possible associations between RI and demographic data. The result of the mining algorithm was a set of 31 rules (Table 4). The
Association parameters of rules (sort by lift).
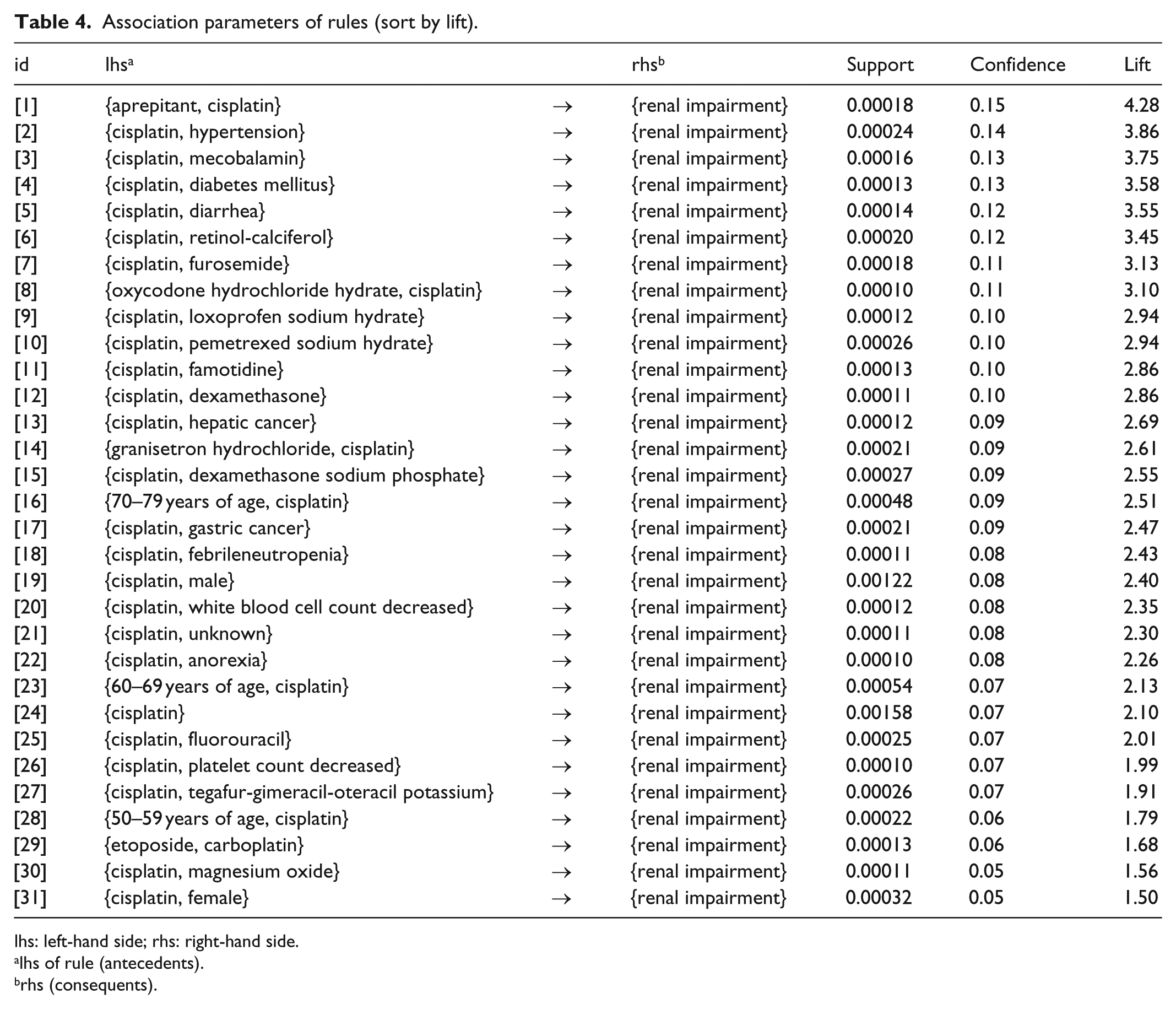
lhs: left-hand side; rhs: right-hand side.
lhs of rule (antecedents).
rhs (consequents).
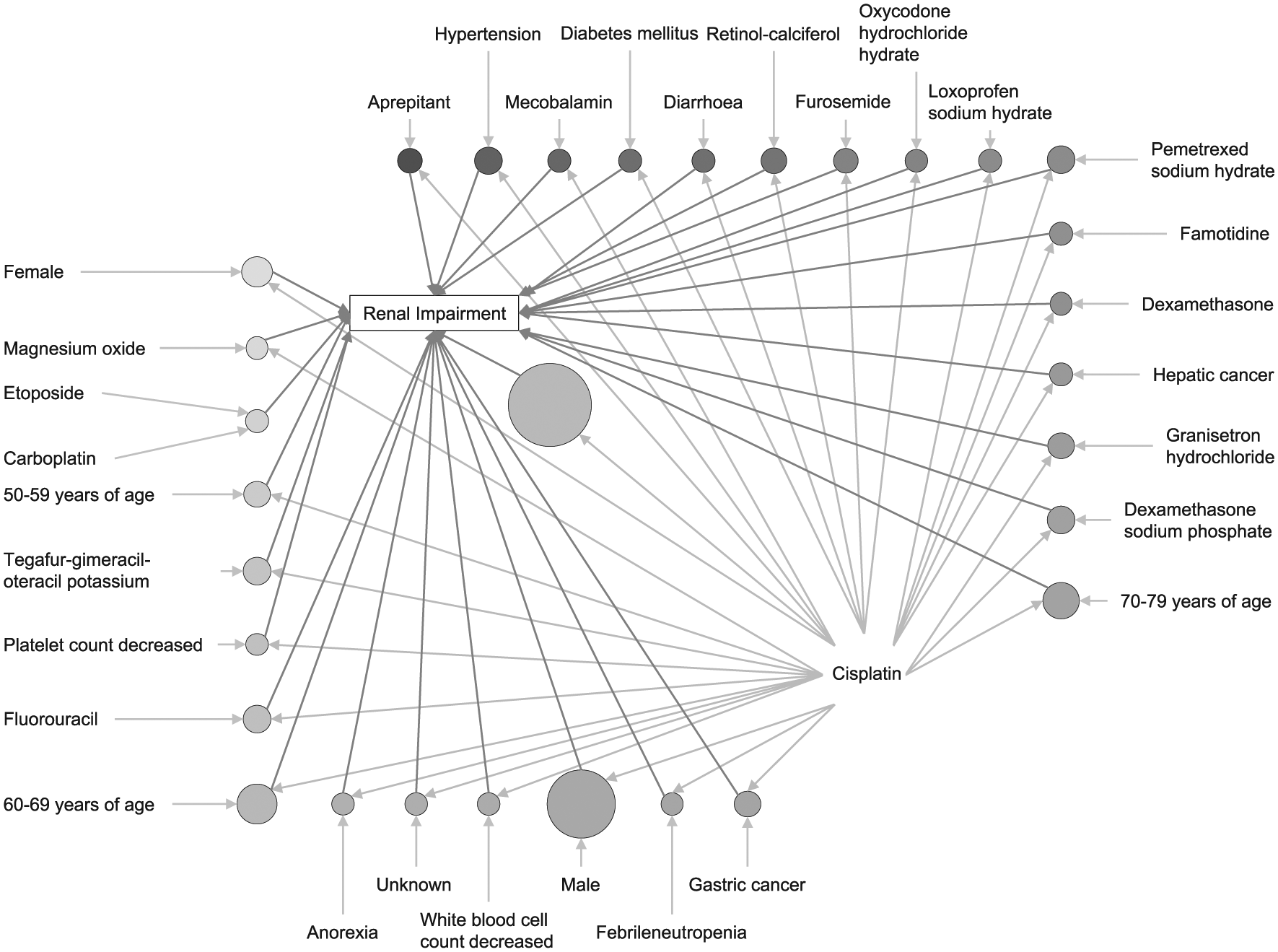
Association rules for renal impairment (RI) based on JADER database from April 2004 to November 2016. Plot represents items and rules as vertices connected with directed edges. Relation parameters are typically added to the plot as labels on the edges or by varying the color or width of the arrows indicating the edges.
Discussion
The RI signal was detected for cisplatin but not for the other platinum-based compounds in the JADER database. This result agrees with those of previous studies.28–30 Approximately 40% of the RI cases were observed 1 week after treatment in the clinical settings. This indicates that health professionals should closely monitor patients for several weeks for RI incidence following treatment with platinum-based compounds.
The upper limit of the 95% CI of ROR for oxaliplatin was <1. We do not have a conclusive explanation for this result. However, the upper limit of the 95% CI of WSP β was <1 (Table 3 and Figure 2), and the hazard was considered to decrease over time (initial failure type; Table 3). We considered that the risk of RI by oxaliplatin should not be ignored:
The association rule mining revealed that the incidence of RI with primary disease–related items such as hypertension or diabetes mellitus was high because of the
The
The findings of several clinical studies indicate that the incidence of cisplatin-induced nephrotoxicity is higher in older patients than in younger patients. 34 The results of the association rule mining confirmed age as a risk factor for cisplatin-induced nephrotoxicity.
The
The risk of developing nephrotoxicity has been reported to be higher in women than in men.34,37,48 In contrast, several reports indicate that women are at a lower risk of developing cisplatin-induced nephrotoxicity than men.
49
The
Our study had some limitations that are worth mentioning. First, the JADER database does not contain detailed background information on medical history (e.g. treatment regimen). Second, SRS has several limitations, including underreporting, overreporting, missing data, comorbidities, and the exclusion of healthy individuals as a reference group.
11
Third, in the association rule mining, the researcher determined the parameters (
Conclusion
This study is the first to evaluate the correlation between platinum-based compounds and RI using ROR, time-to-onset analysis, and association rule mining technique based on the JADER database. Despite the inherent limitations of SRS, we have shown the potential risk of RI during the clinical use of cisplatin. The present analysis demonstrates that the incidence of RI associated with cisplatin use should be closely monitored when patients are hypertensive or diabetic and are co-administered furosemide, loxoprofen, or pemetrexed. We believe that the data presented in this study will help healthcare professionals improve the care of patients undergoing chemotherapy with platinum-based compounds.
Supplemental Material
180324supplementary1Table_02] – Supplemental material for Analysis of adverse events of renal impairment related to platinum-based compounds using the Japanese Adverse Drug Event Report database
Supplemental material, 180324supplementary1Table_02 for Analysis of adverse events of renal impairment related to platinum-based compounds using the Japanese Adverse Drug Event Report database by Misa Naganuma, Yumi Motooka, Sayaka Sasaoka, Haruna Hatahira, Shiori Hasegawa, Akiho Fukuda, Satoshi Nakao, Kazuyo Shimada, Koseki Hirade, Takayuki Mori, Tomoaki Yoshimura, Takeshi Kato and Mitsuhiro Nakamura in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for this study because the study was an observational study without any research subjects.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by JSPS KAKENHI grant number, 17K08452. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
