Abstract
The most common metastatic sites of gastric cancer include the liver, peritoneum, lung, and bone. However, there is a lack of relevant clinical reports regarding rectal metastasis. Herein, we report the rare case of a patient with gastric cancer who developed rectal metastasis. A 57-year-old male patient was diagnosed with gastric cancer and underwent a radical gastrectomy in January 2016, followed by eight cycles of adjuvant chemotherapy. The patient subsequently developed a rectal mass in March 2021. He was diagnosed with rectal adenocarcinoma and underwent surgical resection of the rectal tumor. A mass was then found in the abdominal wall in September 2021 and was resected. Specimens obtained from the three surgeries were reviewed, and the rectal tumor and the mass in the abdominal wall were both found to be metastatic tumors from the gastric cancer. Metastasis of gastric cancer to the rectum is rare, but it is important to differentiate between rectal metastasis and primary rectal cancer to help avoid unnecessary treatment.
Introduction
Gastric cancer is one of the most common tumors worldwide, with an especially high incidence rate in developing countries. 1 Patients with advanced gastric cancer often have distant metastases, especially involving the liver, peritoneum, lung, and bone. 2 Rectal metastasis is relatively uncommon and there is currently a lack of relevant clinical reports. Herein, we report the rare case of a patient with gastric cancer who developed rectal metastasis.
Case presentation
A 57-year-old male patient was diagnosed with gastric cancer (pT4N0M0, stage IIIB) and underwent a radical gastrectomy in January 2016, followed by eight cycles of XELOX chemotherapy. No tumor recurrence was recorded during follow-up. In March 2021, however, the patient was hospitalized with a complaint of poor defecation. Physical examination and colonoscopy revealed a rectal mass located 4 cm from the anus (Figure 1). Magnetic resonance imaging examination also showed a rectal mass suggesting rectal cancer. A biopsy of the mass showed superficial intestinal mucosal tissue, interstitial edema with inflammatory cell infiltration, and mild glandular hyperplasia, but no tumor cells. However, the imaging findings were highly indicative of a tumor. Because no tumor cells were found and the tumor was bleeding, surgical resection without neoadjuvant radiochemotherapy was carried out to obtain a pathologic diagnosis and relieve the bleeding symptoms. The patient underwent transanal total mesorectal excision combined with mesenteric lymph node dissection. Pathological examination of the specimens established a diagnosis of moderate-to-poorly differentiated adenocarcinoma of the rectum. The tumor penetrated the adipose tissue around the rectum. No tumor was found at the surgical margins, but lymphovascular invasion and perineural invasion were confirmed. Fifteen lymph nodes were removed, of which three showed tumor infiltration. The patient was diagnosed with rectal adenocarcinoma (pT3N1bM0, stage IIIB), and had indications for postoperative adjuvant radiotherapy and chemotherapy. However, only one dose of chemotherapy was completed and radiotherapy was discontinued because of severe gastrointestinal adverse events. The patient was followed-up regularly. In September 2021, computed tomography (CT) showed a mass on the right lower abdominal wall, suggesting a metastatic tumor. The patient underwent surgical resection of the mass, and pathological examination suggested a metastatic adenocarcinoma of gastric origin. To further confirm the source of the metastatic tumor, we invited Sun Yat-sen University Cancer Center to conduct a pathological consultation. Specimens obtained from the three surgeries were reviewed. Specimen 1 (from the gastric tumor) was diagnosed as a moderate-to-poorly differentiated adenocarcinoma of mixed type (Lauren classification). The tumor infiltrated the whole gastric wall and invaded the splenic capsule. Lymphatic and venous invasion was identified. The surgical margin was tumor-free. Eleven lymph nodes were removed but none showed signs of invasion. The immunohistochemical (IHC) staining results were matrix metalloproteinase-9(+), proliferating cell nuclear antigen(+), Ki-67(40%+), nm23(+), CD34 (vascular +), epidermal growth factor receptor(+), CerbB-2(0), and vascular endothelial growth factor receptor(+). Specimen 2 (from the rectum) (Figure 2a) was diagnosed as a poorly differentiated adenocarcinoma. The tumor infiltrated the fibrous adipose tissue outside the muscular layer and involved the edge of the smooth muscle layer of the seminal vesicle. Tumor was found at the circumferential margin. Lymphovascular invasion and perineural invasion were identified. Three of the 15 resected lymph nodes showed signs of invasion. IHC staining showed MSH2(+), MSH6(+), MLH1(+), PMS2(+), carcinoembryonic antigen(+), CDX2(+), villin(+), cytokeratin (CK) 20(small foci +), BRAF(−), CK7(weak +), and Ki-67(40%+). Specimen 3 (from the abdominal wall) (Figure 2b) revealed adenocarcinoma infiltration in fibrous adipose tissue. IHC staining showed carcinoembryonic antigen(+), CK7(weak +), CK20(−), and CK(+). The morphology of the tumor tissue in all three specimens was similar. The history, morphology, and IHC changes suggested that the rectal and abdominal wall tumors were metastases from the gastric cancer. In October 2021, CT examination found nodules in the left lower abdomen and left splenic region, which were considered to be metastases from the gastric cancer. The patients underwent PD-1 monoclonal antibody combined with apatinib therapy for 2 months, after which the nodules were in complete remission. However, the patient experienced severe adverse events that were considered to be related to apatinib. Apatinib was therefore discontinued, but PD-1 monoclonal antibody therapy was continued to date. There was no tumor recurrence at the last follow-up in June 2022.

Colonoscopy (March 2021) revealed a rectal mass 4 cm from the anus.

Pathological examination of the rectal tumor (a) and the nodule in the abdominal wall (b) (hematoxylin and eosin staining).
Written informed consent to report the case was obtained from the patient and the patient approved the version to be published. The reporting of this study conforms to CARE guidelines. 3
Discussion
Gastric cancer is one of the most common tumors worldwide, with 1,089,103 new cases and 768,793 deaths in 2020, accounting for 5.6% and 7.7% of the total cancer morbidity and mortality, respectively. 4 Cases of metastatic gastric cancer have increased over the past two decades and now account for about 40% of all gastric cancer cases. 5
Most cases of gastric cancer recurrence after radical resection occur within 4 years, 6 but about 10.8% of patients who survive for 5 years experience recurrence. 7 The dominant recurrence pattern is distant metastasis after 5 years. 7 The most common sites of distant metastasis after complete gastric cancer resection are the liver (37%), lung (16%), bone (16%), lymph nodes (14%), brain (6%), adrenal glands (3%), pleura (2%), and subcutaneous tissue (2%). 6 However, rectal metastasis from gastric cancer is rare. Here, we reported a case of rectal metastasis from gastric cancer, with no peritoneal dissemination.
It is clinically difficult to differentiate intestinal metastasis of gastric cancer from primary colorectal cancer, especially recurrent intestinal metastasis of gastric cancer after radical resection. However, understanding the clinical manifestations, imaging features, and pathological characteristics of intestinal metastasis may help to distinguish between these entities. The clinical manifestations of rectal metastasis are similar to those of primary rectal cancer. The clinical symptoms are non-specific, including abdominal pain, ascites, constipation, and diarrhea, with intestinal obstruction and bleeding in some severe cases.8–10
The typical CT manifestations of intestinal metastasis of gastric cancer are thickening and circular enhancement of the intestinal wall, normally with uniform thickening and layered enhancement. Abnormal thickening of the intestinal wall is caused by the dispersal of cancer cells in the stroma and the formation of fibrous tissues in the intestinal wall. Intestinal metastasis of gastric cancer is often accompanied by ascites, which is also a characteristic of colonic metastasis.11,12
Gastric cancer can invade the colorectum by extension of the primary tumor via the gastrocolic ligament, peritoneal metastasis, hematologic and lymphovascular dissemination. 13 The rectum is the most common site of intestinal metastasis from gastric cancer (37%), followed by the ascending colon (26.7%), transverse colon (24%), and descending colon (5.3%). 12 The uterorectal and bladder rectal crypts are the lowest part of the peritoneal cavity, and cancer cells can easily seed and cause peritoneal implantation metastasis.14–16 Intestinal metastasis of gastric cancer can either occur synchronously at the initial diagnosis or, more commonly, metachronously at relapse. One study of 104 cases of colon metastasis from gastric cancer found that 31% of colon metastases occurred before surgery and 69.2% occurred after surgery. Among the cases with postoperative colon metastasis, 91.3% occurred within 3 years after surgery. The occurrence of colon metastasis 5 years or more after radical surgery is extremely rare. 17 Gastric cancers located at the antrum and cardia of the stomach, tumors confined to the serosal layer (T1-2), tumors larger than 5 cm, and tumors with more than six involved lymph nodes are related to higher risks of colonic metastasis. 18 One study found that 60% of patients with intestinal metastasis had primary tumors larger than 5 cm. 19
The management of intestinal metastasis of gastric cancer should be based on the location, extent of the primary tumor, histological type, and tumor stage. Surgical treatment has certain benefits in patients with intestinal metastasis, with an average survival time of 26 months in patients with colonic metastasis 5 years after radical resection of gastric cancer who received surgical treatment. 9 In addition, surgical resection of intestinal metastases can reduce the risks of bleeding, intestinal obstruction, and perforation. 9 Chemotherapy is recommended for patients with good performance status and extensive metastases that cannot be removed surgically. The chemotherapy regimen includes taxol, platinum, fluorouracil, or a combination of these drugs. 9 However, the small number of cases of intestinal metastasis means that there is currently a lack of large-scale clinical research, and the overall 5-year survival rate has not been reported. We searched the PubMed database and found 11 reported cases of colorectal metastases of gastric origin (Table 1). Four of these 11 cases developed colorectal metastases more than 5 years after the diagnosis of gastric cancer and most underwent resection of the metastases. However, the clinical outcomes of the patients were not reported.8–10,13,20–23
Summary of cases of rectal metastases of gastric origin.
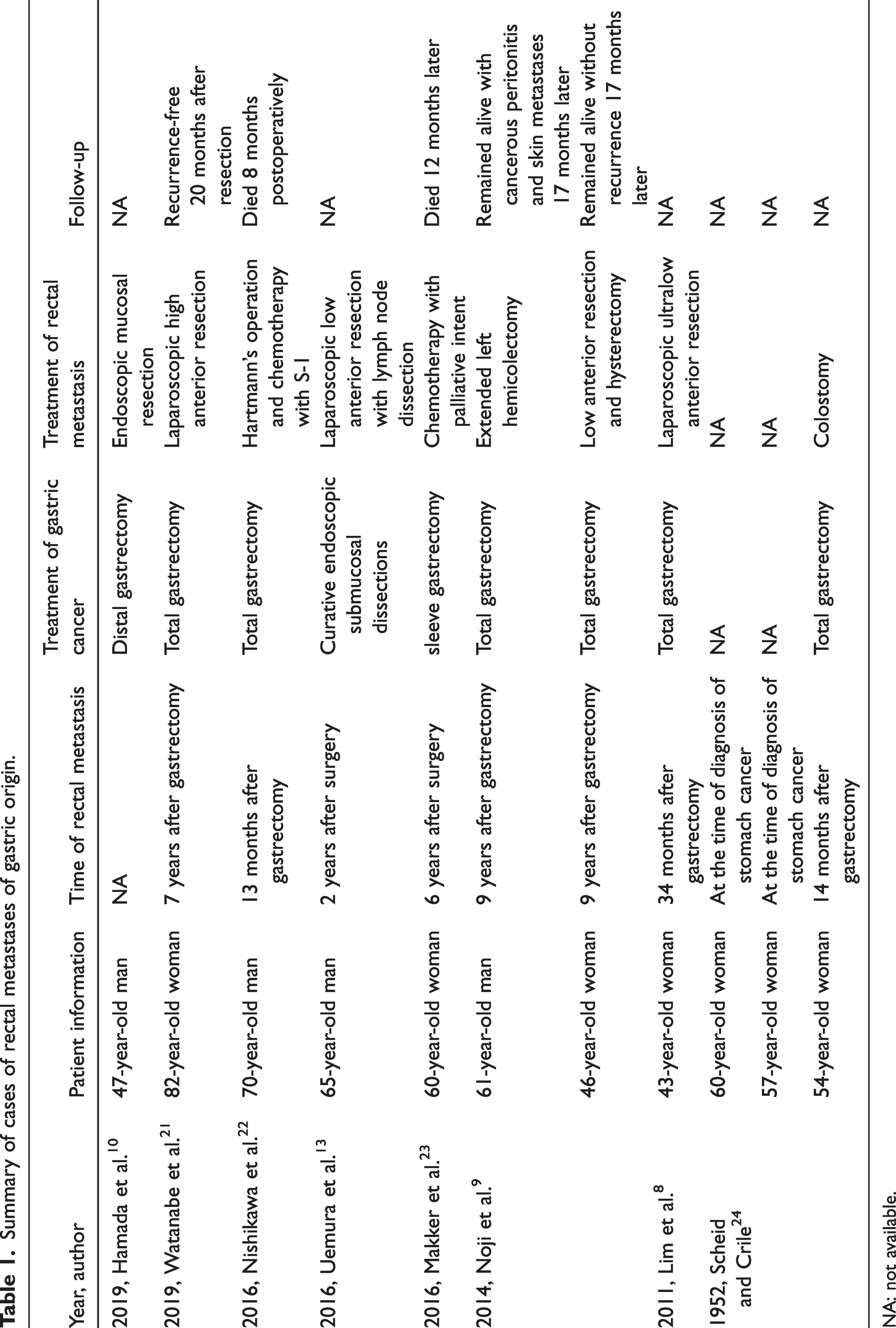
NA: not available.
The current patient developed rectal metastasis 5 years after radical gastrectomy for gastric cancer and was initially misdiagnosed with rectal cancer. The patient was thus treated for rectal cancer and received unnecessary radiotherapy. It is therefore important to differentiate between rectal metastasis and primary rectal cancer to avoid unnecessary treatment.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231198407 - Supplemental material for Rectal metastasis of gastric cancer: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231198407 for Rectal metastasis of gastric cancer: a case report by Lu Tang, Huifen Li, Jing Lv, Cantu Fang, Huatang Zhang and Jincheng Meng in Journal of International Medical Research
Footnotes
Acknowledgements
We thank all those who contributed to this work.
Author contributions
All the authors have made a substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data. All the authors contributed to the writing or revising of the manuscript.
Data availability statement
Data used in the current study are available from the corresponding authors on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
