Abstract
Steroid cell tumor not otherwise specified (SCT-NOS) is a rare type of sex cord-stromal tumor with malignant potential. A 19-year-old woman underwent laparoscopic bilateral cystectomy, and postoperative pathology showed bilateral ovarian SCT-NOS. She had recurrence of the right tumor 8 years after the surgery, with shortened menstrual cycles, elevated testosterone and prolactin concentrations, and impaired glucose metabolism. We performed a laparoscopic right salpingo-oophorectomy. Testosterone and prolactin concentrations rapidly decreased and returned to the normal range after surgery. Subsequently, she had regular menstrual cycles and good glycemic control. The findings in our case suggest that there is a possibility of late recurrence in SCT-NOS. Therefore, we suggest that the postoperative follow-up period should be 10 years for this condition.
Background
Steroid cell tumor (SCT) is a rare type of sex cord-stromal tumor, which accounts for only 0.1% of ovarian tumors. Steroid cell tumor not otherwise specified (SCT-NOS) is a special subtype with malignant potential, and only 6% are bilateral. 1 The main treatment for SCT-NOS is surgery. We present the case of a young woman aged 19 years who underwent laparoscopic bilateral cystectomy after bilateral ovarian masses were discovered. Postoperative pathology showed bilateral ovarian SCT-NOS. She had recurrence of the right tumor 8 years after surgery, with a shortened menstrual cycle, elevated testosterone and prolactin concentrations, and impaired glucose metabolism. We performed laparoscopic right salpingo-oophorectomy. Postoperatively, testosterone and prolactin concentrations rapidly decreased and returned to the normal range. She then had a regular menstrual cycle and good glycemic control.
Case presentation
At the age of 19 years, a patient experienced a shortened menstrual cycle of approximately 15 days over 7 months without any obvious reasons. An ultrasound examination showed bilateral ovarian masses (3.6 × 3.2 × 2.7 cm on the left side and 2.1 × 1.8 × 1.7 cm on the right side), and she was referred to our hospital. We found that the testosterone concentration was elevated at 3.82 nmol/L, the fasting insulin concentration was abnormally elevated at 87.6 μU/mL (Table 1), and the fasting blood glucose concentration and tumor markers were in the normal range. A physical examination showed that the external genitalia of the patient was normal, and there was no obvious facial hirsutism or a thickened voice. Laparoscopic bilateral cystectomy was performed because the patient was a young non-pregnant woman with fertility requirements. Postoperative pathology showed bilateral ovarian SCT-NOS. Immunohistochemistry showed the following: vimentin (+), inhibin (+), partial cytokeratin (+), epithelial membrane antigen (±), chromogranin A (−), synaptophysin (−), S100 (−), scattered CD68 (+), and desmin (+). After the procedure, testosterone and fasting insulin concentrations rapidly declined to normal (Table 1).
Laboratory examinations of the patient.
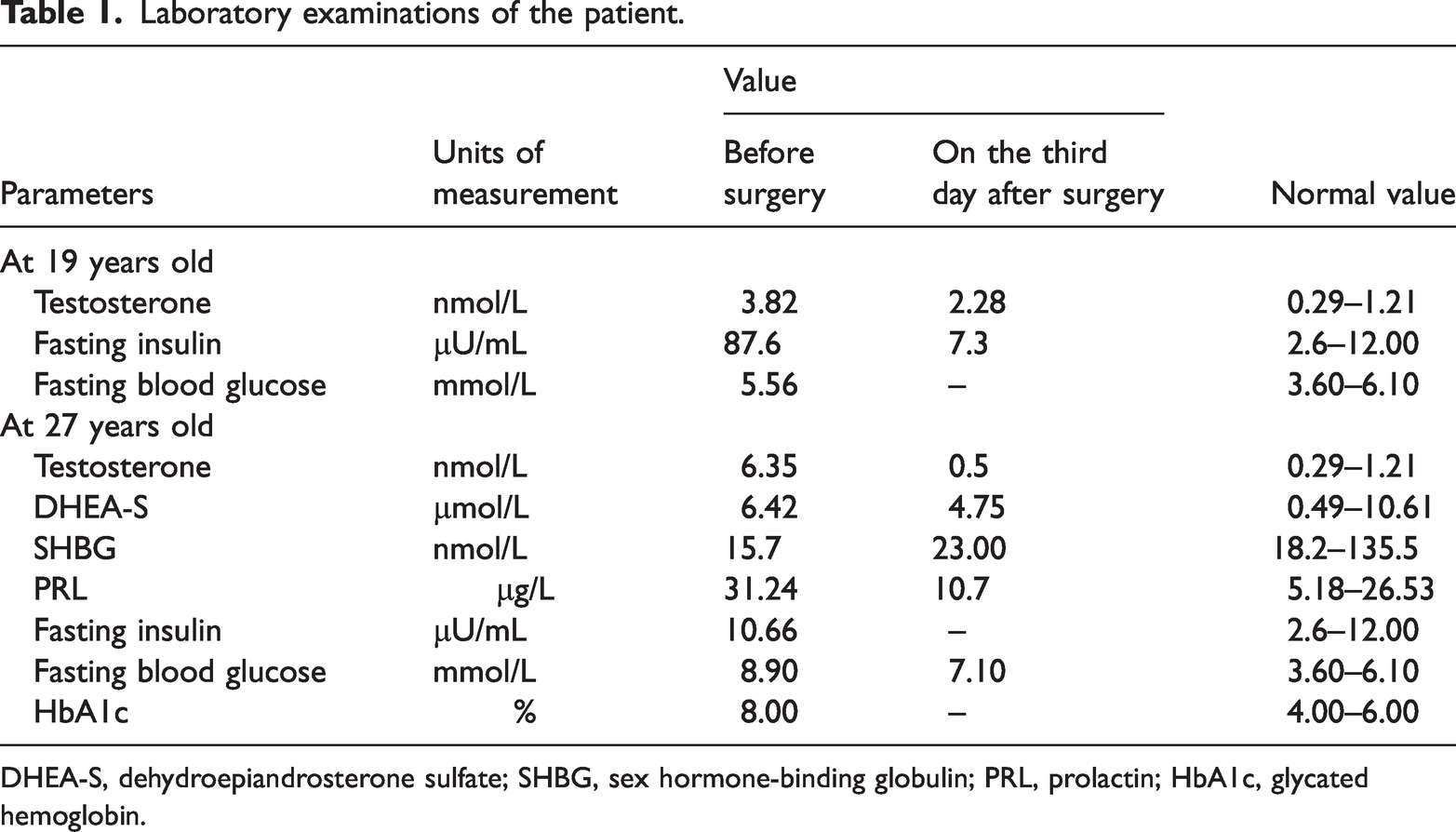
DHEA-S, dehydroepiandrosterone sulfate; SHBG, sex hormone-binding globulin; PRL, prolactin; HbA1c, glycated hemoglobin.
We recommended bilateral oophorectomy because NOS has the potential to be malignant 2 . However, the patient was only 19 years old and she had a strong desire to preserve fertility. If we had performed bilateral oophorectomy for her at that time, she would have directly entered the menopausal state, requiring long-term oral estrogen, progesterone, and other drugs to alleviate perimenopausal syndrome. This situation would have been difficult for the patient. Therefore, she declined additional surgery and requested active follow-up.
After the operation, the patient had a regular menstrual cycle with an interval of 28 days and a duration of 5 days. No masses were found in either of the ovaries by an ultrasound examination every year. When the patient was 27 years of age, the menstrual cycle was shortened with an interval of approximately 15 days for 2 months without any obvious triggers. A sex hormone examination at the local hospital showed elevated testosterone and slightly elevated prolactin concentrations. An ultrasound scan showed a heterogeneous mass in the right adnexal region (Figure 1). The patient returned to our department for treatment. She had received a diagnosis of diabetes 5 years previously, and for 5 years she had maintained a diet that kept her blood glucose concentrations within the normal range. Approximately half a month previously, her fasting blood glucose concentration was 15 mmol/L, and her physician advised her to take an oral metformin hydrochloride sustained release tablet every night.

Transvaginal ultrasonographic findings in the patient. The arrow indicates the ovarian tumor.
She was 160 cm in height and 70 kg in weight. Her body mass index was 27.3 kg/m2 and she was overweight. A physical examination revealed acne on her face and hypertrichosis on her lower legs. A laboratory examination showed normal values of a blood count, follicle-stimulating hormone, luteinizing hormone, estradiol, progestogen, adrenocorticotropic hormone, cortisol, and 17-hydroxyprogesterone. The total serum testosterone concentration was 6.35 nmol/L (normal: 0.29–1.21 nmol/L), serum dehydroepiandrosterone sulfate concentration was 6.42 μmol/L (normal: 0.49–10.61 μmol/L), serum sex hormone-binding globulin was 15.7 nmol/L (normal: 18.2–135.5 nmol/L), and serum prolactin concentration was 31.24 μU/mL (normal: 2.6–12.00 μU/mL). Her fasting insulin concentration was within the normal range, but her fasting blood glucose concentration was 8.9 mmol/L (normal: 3.60–6.10 mmol/L) (Table 1). Additionally, the patient’s glycated hemoglobin A1c value was slightly elevated (8.00%). Therefore, on the advice of an endocrinologist, we added sitagliptin hydrochloride 100 mg once a day orally during the perioperative period. We monitored the patient’s blood glucose concentrations after fasting and three meals.
A pelvic magnetic resonance imaging (MRI) unenhanced scan showed a mass with an abnormal signal shadow in the right adnexal area, and the signal was not uniform. T1-weighted imaging (T1WI) showed equal intensity with multiple low signal areas within the mass, and T2WI showed hyperintensity with a higher signal within the mass. The boundary of the mass was well defined, and its size was 59 × 53 mm. Diffusion was limited and an enhanced scan was markedly enhanced. There was no obvious mass in the rectum, and the surrounding fat space was clear. The rectum, uterus, and bladder were clearly demarcated (Figure 2).

Magnetic resonance imaging findings in the patient. Arrows indicate the ovarian tumor. (a) T1-weighted image, (b) T2-weighted image, and (c) T1-weighted image with contrast medium.
An unenhanced computed tomography (CT) scan of the whole abdomen and pelvis showed a mass of mixed density in the right adnexal area, with a size of 61 × 45 mm. The CT value of the solid component was 19 HU, while that of the cystic component was −7 HU. The mass was well circumscribed and showed markedly heterogeneous enhancement on enhancement. The adjacent ovarian veins were considerably thickened and early developed in the arterial phase. CT also showed a neoplastic lesion in the right adnexal area with arteriovenous fistula formation (Figure 3).
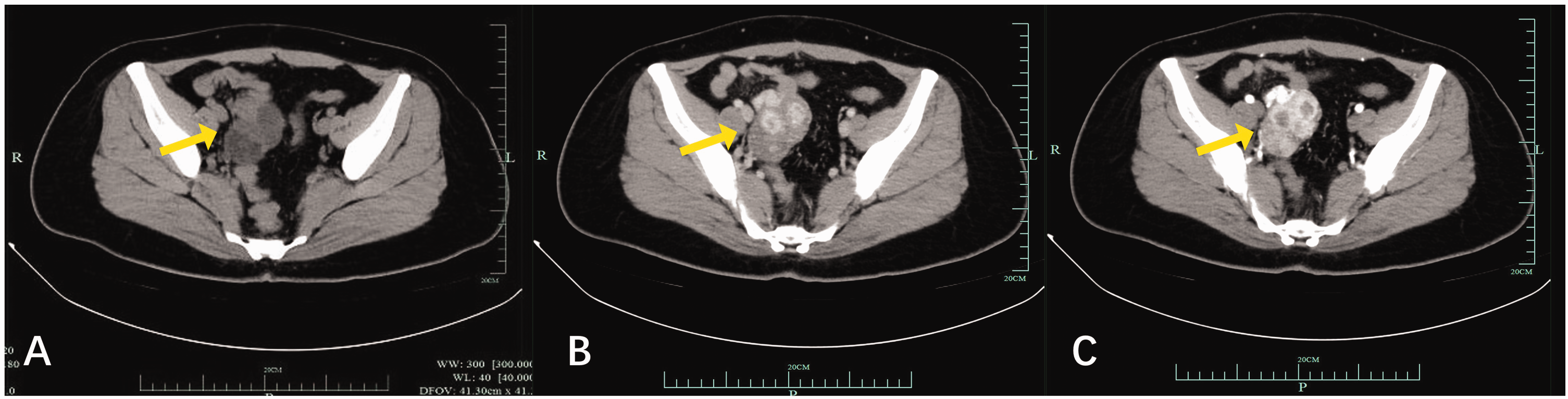
Computed tomography (CT) findings. Arrows indicate the ovarian tumor. (a) Unenhanced CT image, (b) venous phase enhanced CT image, and (c) arterial phase enhanced CT image.
On the basis of the imaging and laboratory examinations, we considered the possibility of a right ovarian sex cord-stromal tumor. We also considered the possibility of recurrence of SCT-NOS because of the patient’s medical history 8 years previously. The patient was an unmarried and non-pregnant young woman who desired to remain fertile, and a radiographic evaluation showed that the tumor was confined to the right ovary, with no evidence of metastasis. Therefore, we chose to perform laparoscopic right salpingo-oophorectomy.
Under laparoscopy, we observed a small amount of light and yellow fluid in the abdominal cavity, and the effusion was withdrawn for a cytological examination. The uterus and bilateral ovaries were closely adhered to the bowel and omentum. The right ovary was enlarged, approximately 6 × 5 × 5 cm in size, and hyperemic on the surface. The right ovary was tightly adhered to the intestine and was surrounded by the right fallopian tube (Figure 4a). The volume of the left ovary was approximately one third to one quarter of normal. No metastatic lesions were found in the liver or pelvis. After careful separation of the adhesions, we laparoscopically removed the right fallopian tube and ovary without rupture and placed them into an endoscopic bag and removed them from the original right lower abdominal incision. The mass was yellow, nodular, solid, and hypervascular, friable to the touch, and resembled the corpus luteum (Figure 4b).
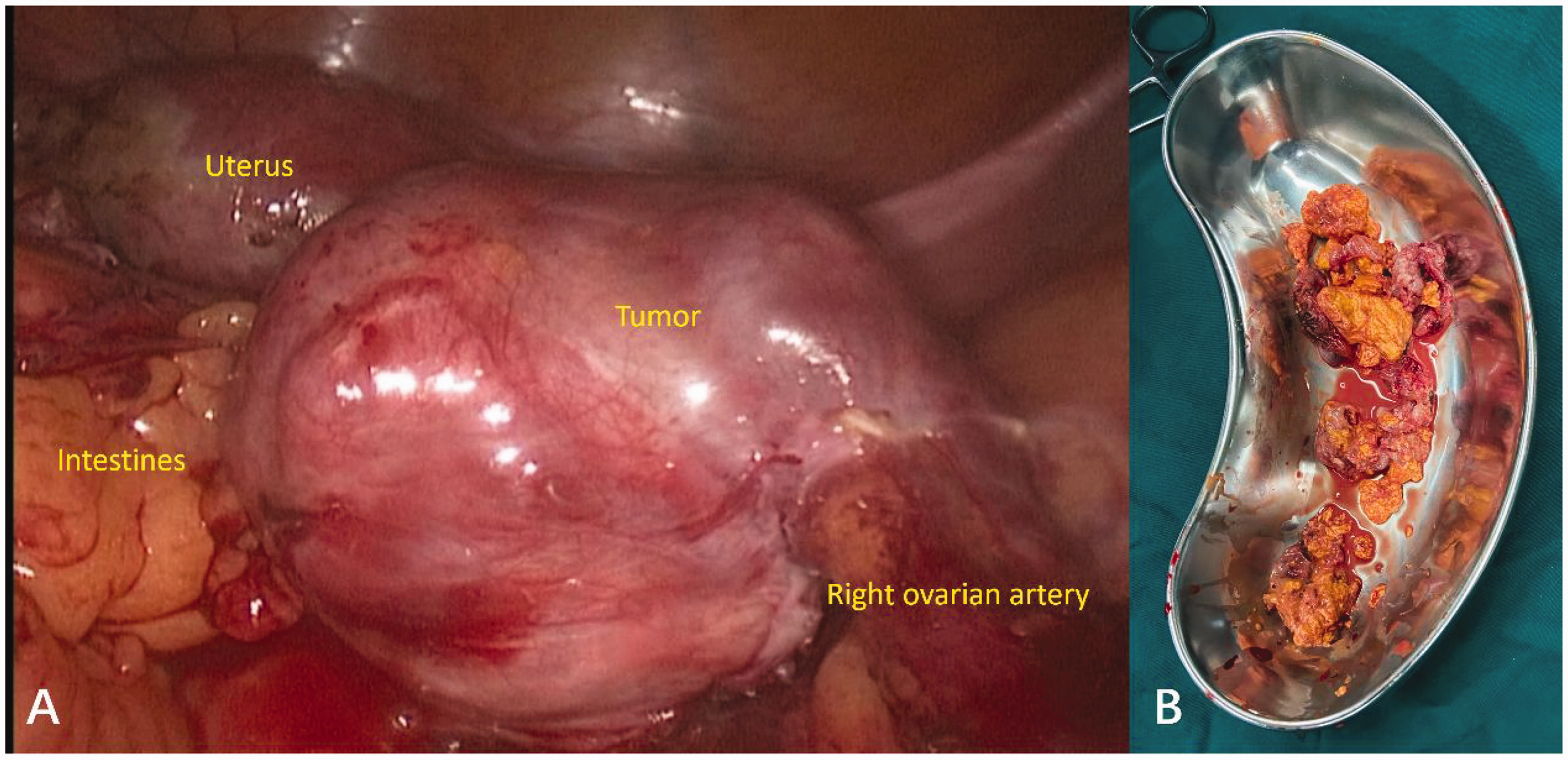
(a) Laparoscopic operative procedure. Images show the right ovarian tumor and (b) macroscopic appearance of the tumor.
Postoperative pathology showed no obvious cancer cells in the peritoneal fluid. Microscopy showed that the tumor cells were round, with an abundant cytoplasm, and were rich in fat. The nuclei were round and small. The tumor cells were arranged in nests and intersected by delicate fibrous bands. These features excluded the possibility of an adrenal tumor and clear cell carcinoma. A histopathological examination revealed sex cord-stromal markers, namely inhibin, calretinin, and vimentin, and <5% Ki67-positive cells. However, Melan-A was negative. PX8 and CD10 were negative, which excluded the possibility of a renal tumor. These results were consistent with ovarian SCT-NOS, which tends to be benign (Figure 5).

Microscopic appearance of the tumor. (a) A histological specimen shows a large aggregation of tumor cells with an abundant clear cytoplasm and small round nuclei. Fibrous septa can be between the tumors (hematoxylin and eosin staining, ×200). (b) Immunohistochemical staining shows that there is a diffuse positive reaction for inhibin in the tumor cells (×200). (c) Immunohistochemical staining shows a diffuse positive reaction of the tumor cells for calretinin (dark red) (×200).
We reviewed the patient’s sex hormone concentrations on the third postoperative day and found that testosterone and prolactin concentrations had rapidly decreased and returned to the normal range (Table 1). The patient was followed up 4 weeks after surgery. At this time, prolactin and testosterone concentrations were within the reference range, and a transvaginal ultrasound examination showed no obvious abnormalities in the uterine appendages. Her facial acne was improved, and her blood glucose concentrations were more easily controlled. The patient’s menstruation returned to a regular cycle 3 months after the operation.
Discussion
SCT is a rare type of sex cord-stromal tumor, and it can be divided into three subtypes regarding their cell of origin: NOS, stromal luteoma, and Leydig cell tumor. NOS accounts for 56% of all cases. Patients with SCT-NOS range in age from 2 to 80 years, with a mean age of 43 years. 3 SCT-NOS is usually solid and well circumscribed, occasionally lobulated, and only 6% are bilateral. In our case, the patient was 19 years old and had bilateral SCT-NOS. The clinical manifestations of SCT-NOS are mostly ambiguous, but the most common features are abdominal pain and distention. Changes in androgens are found in approximately half of SCT-NOS cases and persist over many years. These changes often occur with amenorrhea, oligomenorrhea, and virilizing features, such as a reduced breast size, hirsutism, facial acne, male pattern baldness, and clitoral hypertrophy. 4
In SCT-NOS, hormones other than testosterone are sometimes elevated. Estradiol concentrations are elevated in 6% to 23% of patients and can lead to menorrhagia, postmenopausal bleeding, and even endometrial cancer. 5 In 6% to 10% of cases, the clinical presentation is associated with Cushing’s syndrome with elevated glucocorticoid concentrations. However, approximately 25% of SCT-NOS cases are not associated with hormone production. 3
When our patient was 19 years old, her fasting insulin concentration was high, while her fasting blood glucose concentration was within the normal range, and after surgery, fasting insulin concentrations rapidly decreased. These findings suggest that tumors may increase fasting insulin expression by decreasing insulin sensitivity. Her blood glucose concentrations were well controlled with dietary management for 8 years. When the patient was 27 years old, her fasting insulin concentration was in the normal range, but her glycated hemoglobin A1c and fasting blood glucose values were high, which indicated that glucose metabolism was more impaired than previously. This finding suggests that tumors may impair glucose metabolism. The patient’s blood glucose concentrations were well controlled after surgery. SCT-NOS was found twice in our patient, and it was accompanied by elevated testosterone concentrations and a shortened menstrual cycle. After surgery, testosterone concentrations returned to normal and the menstrual cycle resumed its original rhythm, suggesting that SCT-NOS increases the secretion of testosterone and affects the menstrual cycle. When the patient was 27 years of age, laboratory tests showed a slight increase in the prolactin concentration, which rapidly declined after surgery. Prolactin is partially synthesized in the ovary, and elevated prolactin concentrations may be associated with an increased risk of ovarian cancer, especially in women with a body mass index ≥25 kg/m2. 6 In cases of granulosa cell tumor, which is another type of sex cord-stromal tumor, elevated prolactin concentrations have also been found in some cases. 7 We speculate that SCT-NOS regulates prolactin secretion.
The diagnosis of SCT-NOS should be based on clinical virilization, laboratory analysis, imaging studies, and a pathological examination. Ovarian SCTs are characterized by an abundance of intracellular lipids. 8 Lipid has a characteristic appearance and can be identified by ultrasound, CT, and MRI. Fat-rich tumors appear hyperechoic on ultrasound and hypodense on CT, similar to subcutaneous fat. On MRI, fat-rich tumors show higher intensity on T1WI. 9 Under the microscope, SCT-NOS is usually solid, composed of polygonal or round cells with well-defined cell boundaries and an abundant cytoplasm, and lipofuscin is present in the cytoplasm in approximately one third of cases. NOS lacks Reinke’s crystals and thus can be distinguished from Leydig cell tumors. 3 The positive rates of α-inhibin and calretinin in SCT-NOS are 80% to 100% and 90% to 100%, respectively. Calretinin is more sensitive than α-inhibin in ovarian sex cord-stromal tumors, but has less specificity. 10 The positive rate of Melan A is 77.8% to 100% in sex cord-stromal tumors, but the staining is weaker than that of α-inhibin and shows focal expression. A Melan A reaction is only seen in the tumor area with steroid cell differentiation. 11 Additionally, 75% of SCTs are positive for vimentin. Calretinin and α-inhibin are thought to be sensitive markers of ovarian SCT-NOS. Melan A and vimentin are also helpful for the diagnosis of SCT-NOS. In the present case, tissue of the excised tumor expressed inhibin, calretinin, and vimentin, but not Melan A, which is in line with previous studies.3,10,11
Approximately one third of SCTs are malignant. Their degree of malignancy can be judged by the pathological characteristics of the tumor, including two or more mitoses per 10 high-power fields, necrosis, the size of the tumor (>7 cm), hemorrhage, and grade 2 or 3 nuclear atypia. 3 Clinical detection of metastatic lesions can also be used to evaluate benign and malignant tumors, mainly pelvic metastasis,12,13 and occasionally uterine metastasis 5 and liver metastaees. 14 Therefore, before surgery, patients with SCT-NOS should be evaluated for metastatic disease by full abdominal CT or pelvic magnetic resonance imaging.
There is no clear standard treatment for ovarian SCT-NOS because it is rare. Surgery is the main treatment for this condition. Complete removal of the tumor facilitates the resolution of symptoms and a reduction in testosterone concentrations. The surgical method that should be used is determined according to the patient’s age, fertility requirements, and whether it is a benign or malignant tumor. The mainstay treatment of ovarian SCTs is total abdominal hysterectomy with bilateral salpingo-oophorectomy. Because SCT-NOS is mostly benign, unilateral oophorectomy may also be performed in young women with early progression of this condition who want to preserve reproductive function. According to a literature review, eight cases of SCT-NOS underwent laparoscopic oophorectomy, and only four cases underwent partial oophorectomy. 15 The patients who underwent partial resection were followed up for <5 years and had no recurrence of SCT-NOS. However, no previous reports showed a longer follow-up than this time. At present, there is no clear recommendation for the follow-up time of patients undergoing laparoscopic partial resection. The findings in our case suggest that SCT-NOS has a higher than 5-year survival rate, but there is a possibility of late recurrence, similar to granular cell tumors. Therefore, we suggest that the postoperative follow-up period for SCT-NOS should be 10 years.
Patients with malignant tumors should be treated with chemotherapy and radiotherapy after surgery, but there is no optimal adjuvant regimen because SCT-NOS is rare and mostly benign, with little metastasis and recurrence. The recommended adjuvant chemotherapy regimens are as follows: bleomycin, etoposide, and cisplatin; cisplatin, adriamycin, and cyclophosphamide; taxane and platinum; and bleomycin, vinblastine, and cisplatin. 13 Gonadotropin-releasing hormone agonists also inhibit ovarian steroidogenesis, and they have been used to successfully reduce serum testosterone concentrations and shrink recurrent tumors. 16
Conclusion
SCT-NOS is a special subtype of SCT with malignant potential. The main treatment of SCT-NOS is surgery. The preservation of fertility should be considered when the patient is of childbearing age, depending on the degree of malignancy. If fertility-sparing surgery is performed, owing to the possibility of late recurrence, the patient must be followed up carefully for a long time, possibly for longer than 5 years. A disturbance in the menstrual cycle, elevated testosterone concentrations, and other endocrine metabolic disturbances, such as hyperprolactinemia and impaired glucose metabolism, may indicate tumor recurrence.
Footnotes
Acknowledgement
We appreciate Dr Jian Wu who helped us improve the quality of the pathological figures in the manuscript.
Author contributions
Dr. Hong-Kai Shang designed the study and Yuan Xu (postgraduate) drafted the manuscript. All authors read and approved the final manuscript.
Data availability statement
Data from this report are openly available in a public repository.
Disclosure statement
The reporting of this study conforms to the CARE guidelines. 17
Declaration of conflicting interest
All authors declare that there is no conflict of interest.
Ethics statement
The patient’s details have been de-identified in the manuscript. The patient provided written informed consent for her data to be submitted or published. Approval from an ethics review board was not required because this was a case report.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
