Abstract
Objective
To investigate sex hormone and blood lipid levels in patients with lifelong premature ejaculation (LPE) in China.
Methods
Sex hormone and blood lipid levels were measured in 156 patients with LPE and 76 healthy controls. The Premature Ejaculation Diagnostic Tool (PEDT) and Chinese Index of Sexual Function for Premature Ejaculation-5 Questionnaires (CIPE-5) were applied to diagnose and grade LPE.
Results
PEDT and CIPE-5 scores were significantly altered in the LPE group compared with the control group. Free testosterone levels were significantly higher in the LPE group than in the control group. Free testosterone levels were also significantly higher in the mild, moderate, and severe LPE subgroups than in the control group. Total testosterone and prolactin levels tended to be lower in the control group than in the LPE group. Very low-density lipoprotein levels were significantly lower in the LPE group and LPE subgroups than in the control group. Triglyceride levels were highest in controls and decreased with progression of LPE.
Conclusions
Patients with LPE have higher free testosterone levels and lower very low-density lipoprotein levels than controls. These findings indicate that these factors might be indices for LPE. However, the reasons for these phenomena need to be further investigated.
Keywords
Introduction
Premature ejaculation (PE) is the most common male sexual dysfunction, and the prevalence of PE worldwide is 20% to 30%.1,2 The most widely accepted definition of PE was established by the International Society for Sexual Medicine and includes three characteristics. 2 PE affects intimacy of couples, undermines self-confidence, and leads to anxiety, embarrassment, and depression. 3 Among four subgroups, lifelong PE (LPE) has a prevalence of 2.3% to 3%. 1
The cause of PE is unclear and its complexity stems from the possible involvement of psychogenic factors, such as early sexual experience, 4 anxiety 5 and frequency of sexual activity. 6 Biogenic factors, such as endocrinopathy (including hormones),7,8 genetic predisposition, 9 and 5-hydroxytryptamine receptor dysfunction,10–12 and some diseases, such as chronic prostatitis and erectile dysfunction, are associated with PE. 13 Psychological factors, organic factors, and various diseases affect each other, causing difficulty for people to understand the exact cause of PE.
Testosterone modulates the function of different tissues (muscle, adipose, and bone) and cell types (epithelial, endothelial, and hematopoietic) and regulates the metabolism of lipids, carbohydrates, and proteins.14,15 Sex hormones (especially testosterone and prolactin) affect erectile dysfunction.16,17 Higher testosterone levels may result in PE, whereas reduced testosterone levels may be associated with delayed ejaculation. 8 A previous clinical study showed that some patients with LPE had higher free testosterone (FT) levels. 18 Testosterone is closely related to blood lipid metabolism. Long-term testosterone therapy improves the lipid profile by reducing total cholesterol (TC), low-density lipoprotein (LDL) cholesterol, and triglyceride (TG) levels, and increasing high-density lipoprotein (HDL) cholesterol levels compared with baseline levels. 19 Although testosterone levels in LPE vary among studies,7,8 there have been few studies on the role of these factors (e.g., testosterone, blood lipids) in LPE at the same time.
This case–control study compared serum levels of sex hormones and blood lipids between patients with LPE and healthy controls in different populations in China, and determined the relationship between these factors and LPE.
Material and methods
Subjects
Based on the incidence of LPE and the conditions of a case–control study, 182 patients with LPE who were diagnosed with PE in andrology clinics between September 2017 and March 2018 were included in the study. The control group included 86 healthy men who were selected from the Physical Examination Center, which provides a routine physical examination for people. The medical and sexual history was collected before the study, and both groups underwent the same tests after meeting the inclusion and exclusion criteria defined by the same physician. The intravaginal ejaculation latency time (IELT) was measured by self-reporting. A flowchart of the study is shown in Figure 1.

Study flowchart.
All procedures were approved by the Ethics Committee of the Second Affiliated Hospital, Xi’an Jiaotong University (No: 2017-0029). Our study adhered to the code of ethics of the World Medical Association (Declaration of Helsinki) for experiments involving human subjects and complied with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Written consent was obtained from the participants who provided samples.
Inclusion and exclusion criteria
The inclusion criteria for patients with LPE were patients who complained of always or usually ejaculating within 2 minutes before or after vaginal penetration (with every or nearly every sexual partner and from the first sexual encounter onwards), with a heterosexual relationship for at least 6 months. The inclusion criteria for controls were healthy individuals who underwent a physical examination at the same hospital during the same period as patients with LPE and an IELT >5 minutes (i.e., ejaculating after more than 5 minutes of vaginal penetration). A history of cerebrovascular disease, neurological disease, thyroid disease, hypertension, diabetes mellitus, endocrine pathology, psychiatric disorders, and erectile dysfunction were factors of exclusion for both groups.
Study design
The history of LPE was obtained from all patients. Both study groups completed the Premature Ejaculation Diagnostic Tool (PEDT) 20 and Chinese Index of Sexual Function for Premature Ejaculation-5 Questionnaire (CIPE-5). 20 The PEDT was primarily used to diagnose PE, whereas the CIPE-5 was used to grade PE. After 12 hours of fasting, blood samples were collected to measure levels of sex hormones, lipids, and glucose. Sex hormones were measured using a chemiluminescent immunoassay (Roche Diagnostics, Basel, Switzerland).
Indicators
The following PEDT scores were used: ≤8, absence of PE; 9 to 10, possible PE; and ≥11, presence of PE. 20 Patients with LPE were divided into three groups on the basis of the total CIPE-5 score as follows: mild, ≥15 points; moderate, 10 to 14 points; and severe, 5 to 9 points. 21
Statistical analysis
Statistical analysis was performed using SPSS software version 18.0 (SPSS Inc., Chicago, IL, USA). Quantitative variables are expressed as mean ± standard deviation or percentage, when appropriate. The Kolmogorov–Smirnov test was used to assess normality of the data, and the Wilcoxon rank sum test of two independent samples was used to compare PEDT and CIPE-5 scores between the LPE and control groups. The independent-samples t-test was used to compare the IELT, body mass index (BMI), sex hormones, and blood lipids between the two groups. Analysis of variance was used to compare the means of multiple samples in the LPE group. Statistical significance was defined as P < 0.05.
Results
Subjects’ characteristics
A total of 156 patients with LPE and 76 controls were eligible for the study. Of these, 26 patients in the LPE group and 10 patients in the control group were excluded because of incomplete data or abnormal blood glucose levels.
The patients in the LPE group were aged 21 to 48 years (mean, 30.36 ± 5.09 years), including 145 men aged <40 years and 11 men aged ≥40 years. The disease course varied from 1.5 to 16.0 years (mean, 4.70 ± 4.05 years). The IELT in the LPE group ranged from 0.50 to 2.00 minutes (mean, 1.46 ± 0.56 minutes). The subjects in the control group were aged 22 to 45 years (mean, 31.29 ± 5.01 years), including 71 men aged < 40 years and 5 men aged ≥40 years. The IELT in the control group ranged from 5.00 to 20.00 minutes (mean, 8.50 ± 3.89 minutes). There was no significant difference in age between the two groups.
The levels of education of patients with LPE and their partners were primary (0.64% and 1.28%), secondary (56.41% and 58.98%), higher (41.03% and 38.46%), and graduate (1.92% and 1.28%), respectively. The levels of education in the control group and their partners were primary (1.32% and 0.00%), secondary (56.58% and 59.21%), higher (39.47% and 38.16%), and graduate (2.63% and 2.63%), respectively. The level of education was not significantly different between the two groups. The rate of circumcision in the LPE group was not significantly different to that in the control group (48.72% versus 43.42%). The BMI in the LPE group was not significantly different from that in the control group (23.64 ± 3.50 versus 24.08 ± 3.25 kg/m2).
PEDT and CIPE-5 scores
The PEDT scores (D = 0.109, P < 0.001) and CIPE-5 scores (D = 0.089, P < 0.05) in the LPE group followed a non-normal distribution, and therefore, the Wilcoxon rank sum test was used for analysis. The PEDT score was significantly higher (P < 0.001) and the CIPE-5 score was significantly lower (P < 0.001) in the LPE group than in the control group (Table 1). The diagnostic sensitivity of the PEDT in the LPE group was 71.79% and diagnostic specificity in the control group was 77.63%. The CIPE-5 was not able to be used to diagnose LPE and the CIPE-5 scores in the control group were ≥15 points. The percentage of patients with LPE with mild, moderate, and severe dysfunction was 11.54%, 51.28%, and 37.18%, respectively.
PEDT and CIPE-5 scores in the groups
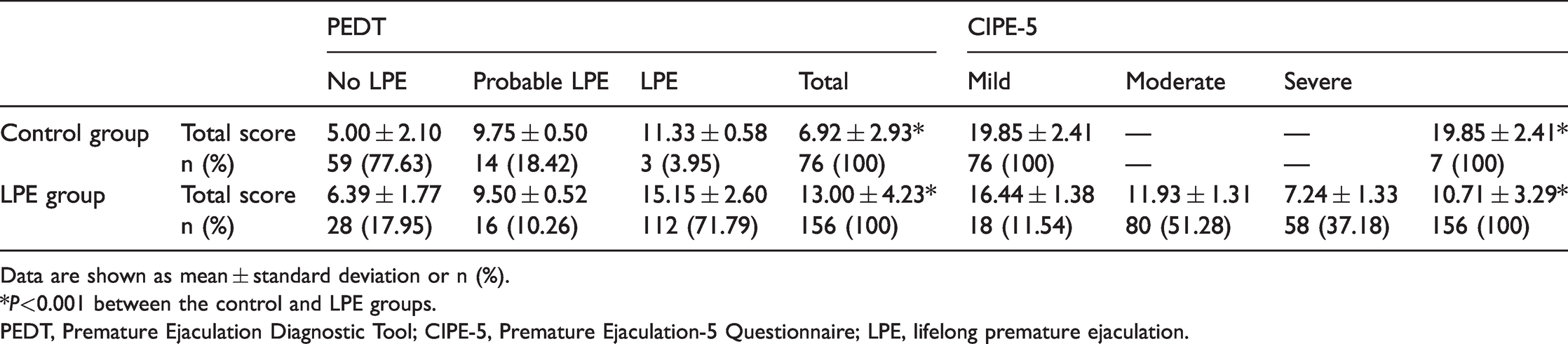
Data are shown as mean ± standard deviation or n (%).
*P<0.001 between the control and LPE groups.
PEDT, Premature Ejaculation Diagnostic Tool; CIPE-5, Premature Ejaculation-5 Questionnaire; LPE, lifelong premature ejaculation.
Sex hormones
The mean FT level was significantly higher in the LPE group than in the control group (P < 0.001). However, there were no significant differences in the levels of estradiol, total testosterone (TT), luteinizing hormone, follicle-stimulating hormone (FSH), progesterone, and prolactin between the two groups. Sex hormone levels were compared in relation to CIPE-5 scores. Only FT levels were significantly higher in patients with mild, moderate, and severe LPE than in the control group (all P < 0.001). However, analysis of variance indicated that there was no significant difference in sex hormones among the three LPE subgroups (Table 2).
Hormonal and lipid status in the groups.
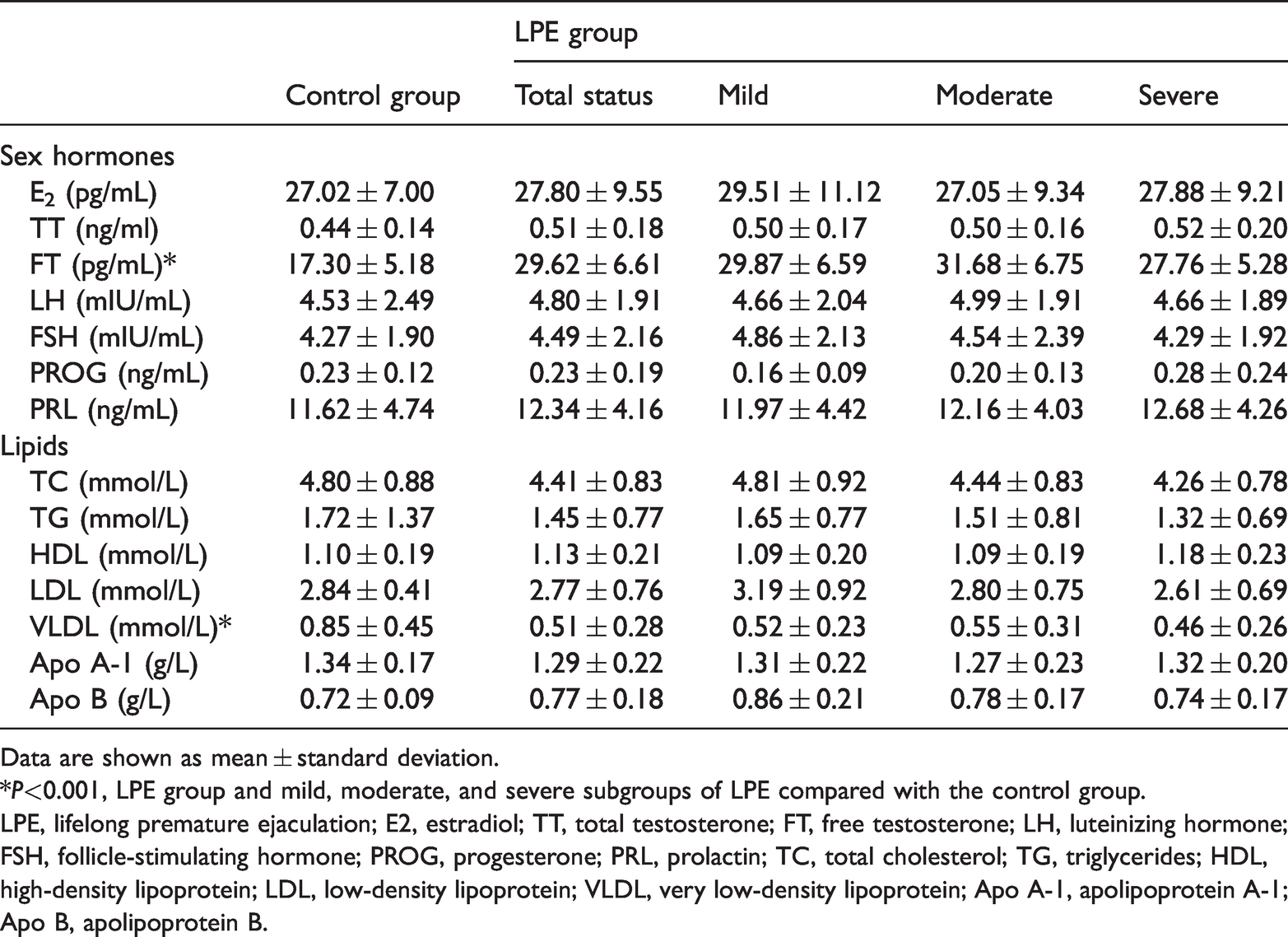
Data are shown as mean ± standard deviation.
*P<0.001, LPE group and mild, moderate, and severe subgroups of LPE compared with the control group.
LPE, lifelong premature ejaculation; E2, estradiol; TT, total testosterone; FT, free testosterone; LH, luteinizing hormone; FSH, follicle-stimulating hormone; PROG, progesterone; PRL, prolactin; TC, total cholesterol; TG, triglycerides; HDL, high-density lipoprotein; LDL, low-density lipoprotein; VLDL, very low-density lipoprotein; Apo A-1, apolipoprotein A-1; Apo B, apolipoprotein B.
Sex hormones levels varied between the control and LPE subgroups. TT and prolactin levels tended to be lower in a healthy condition than with LPE. The mean progesterone level appeared to be higher in the control group than in the mild LPE group. Progesterone and prolactin levels tended to increase and FSH levels tended to decrease with worsening of LPE (Figure 2).

TT, PRL, PROG, FSH, TC, TG, HDL, and LDL levels in the group with lifelong premature ejaculation and the subgroups with mild, moderate, and severe premature ejaculation.
Blood lipids
The mean very low density lipoprotein (VLDL) level was significantly lower in the LPE group than in the control group (P < 0.001) (Table 2). However, there were no significant differences in TC, TG, HDL, LDL, apolipoprotein A-1, and apolipoprotein B levels between the two groups. According to CIPE-5 scores, VLDL levels were significantly lower in patients with mild, moderate, and severe (all P < 0.001) LPE than in controls, with no significant difference among the subgroups.
Blood lipid levels varied between the control group and LPE subgroups. TG levels were highest in the control group and decreased from mild to severe LPE. In the LPE group, HDL levels increased, whereas TC and LDL levels decreased with progression of LPE (Figure 2).
Discussion
The diagnosis of LPE is primarily based on the IELT and medical history at present. Our study showed that FT levels, but not TT levels, were significantly higher in the LPE group than in the control group. Moreover, FT levels were significantly higher in the PE subgroups than in the control group, although there was no significant difference in FT levels among the PE subgroups. Additionally, VLDL levels were significantly lower in the LPE group than in the control group, which to the best of our knowledge, has not been previously reported.
Testosterone is a major regulator of male sexual behavior, including gender recognition, sexual arousal, erection, and ejaculation. 22 Keleta et al. showed that long-term treatment with testosterone significantly reduced 5-hydroxytryptamine levels in the brain of rats, 23 which suggested that testosterone might be involved in PE. Nonetheless, the relationship between serum testosterone levels and PE is unclear. Previous studies showed that patients with PE had higher FT and FSH levels than healthy men; however, there was no significant difference in TT levels between these two groups. 24 Testosterone and FT are associated with sexual arousal, erection, and ejaculation, and patients with LPE tend to present with a hyperexcitable ejaculatory reflex and hyperarousability. 8 An increase in FT levels may be the cause of these two conditions in these patients. The reasons for higher FT levels in some patients with LPE require further investigation.
Testosterone controls body functions in men.14,24 In patients with testosterone deficiency, testosterone therapy improves sexual desire, erectile function, energy, mood, and vigor, reduces TC, LDL cholesterol, and TG levels, and increases HDL cholesterol levels. 15 Blood lipids affect sexual function and erectile dysfunction is ameliorated by reducing hypercholesterolemia. 25 In our study, FT levels were higher and VLDL levels were lower in the LPE group than in the control group, which suggested a negative correlation between FT and VLDL levels. The reason for the absence of significant intergroup differences in the levels of other blood lipids and the mechanisms underlying regulation of FT and VLDL are not fully understood.
Local anesthetics are the first-line treatment for PE in Europe. 26 Neurophysiological analysis has shown that some patients with PE have penile hypersensitivity, 27 suggesting that, to some extent, PE might be caused by neurological disease. Bronner et al. described eight patients with Parkinson’s disease who complained of PE, although he believed that PE was caused by a medication effect. 28 We speculate that PE in some patients may be caused by nerve sheath disease via abnormal blood lipid metabolism, leading to penile hypersensitivity and premature ejaculation. Current research has shown that blood lipids may help control integrity of the myelin sheath, which may affect nerve conduction.25,29,30 The role of VLDL in PE was not examined in detail in the present study. The underlying mechanisms of this role need to be determined using animal models and large, randomized, controlled trials.
Abu El-Hamd and Farah investigated serum testosterone, gonadotropin and prolactin levels in 90 patients with PE and 90 controls, and found no differences in these factors between the two groups. 31 Our study showed that FT levels were not significantly different among the LPE subgroups. Additionally, TT, prolactin, and progesterone levels tended to increase, whereas FSH levels tended to decrease as dysfunction worsened. These phenomena may have been caused by clinical factors (anxiety and depression) in patients with PE, 32 changes resulting from an increase in FT levels, bias induced by the small sample size, the effect of the test method (chemiluminescent immunoassay), and other factors.
The PEDT is the most commonly used scale for diagnosing PE. The Chinese version of the PEDT scale is useful for screening PE and has an excellent correlation with clinical diagnoses. 20 In this study, the PEDT scale was used to diagnose LPE and it had a false-negative rate of 23.10% and false-positive rate of 17.60%. Similarly, a previous study showed that the false-negative and false-positive rates of this scale were 17.95% and 22.37%, respectively. 33 A possible reason for the high rates in our study is because the evaluated populations were different (individuals with and without LPE) and self-reporting was used. The IELT tends to be higher with self-reporting than with using a stopwatch. 33
The advantage of the CIPE-5 is the ability to grade severity of LPE. In our sample, the percentage of patients with LPE with mild, moderate, and severe dysfunction as assessed by CIPE-5 scores was 11.54%, 51.28%, and 37.18%, respectively. A Chinese study evaluated 167 patients with LPE and showed that the rates of mild, moderate, and severe dysfunction using the same instrument were 19.8%, 62.8%, and 16.7%, respectively. 20 In our study, the rate of severe LPE was higher than that in the previous Chinese study. This discrepancy between studies might be related to the strict inclusion criteria, and patients who did not meet these criteria or did not have a definite diagnosis were excluded from the study.
The IELT of LPE is defined by the International Society for Sexual Medicine as < 1 minute. The IELT in our study was defined within 2 minutes, but not 1 minute, because some studies have shown that 10% to 20% of LPE occurs between 1 and 2 minutes,34,35 and the definition of < 2 minutes could maximize inclusion of patients with LPE. The inclusion criteria in this study were men whose core symptoms were early ejaculation at nearly every intercourse, within 2 minutes, with every or nearly every sexual partner, and from the first sexual encounter onwards. These features distinguished LPE from acquired PE. Additionally, self-reporting of the IELT in our study should have been effective because there is a strong association between self-reporting and using a stopwatch for determining ejaculatory latency.20,36,37
There are some limitations to our study. Blood lipid metabolism is affected by many factors. Although BMI was not significantly different between the groups in our study, other factors can affect blood lipid metabolism, except for LPE, which require further investigation in an experimental control study. Second, testosterone is strongly bound to sex hormone-binding globulin (SHBG). Measurement of FT may be more appropriate than measuring TT because FT is positively associated with total testosterone and strongly negatively associated with SHBG levels. 28 Higher FT levels indicate lower SHBG levels in serum. SHBG levels are lower in obesity, in type 2 diabetes, hirsutism, and metabolic syndrome, 28 which may lead to higher FT levels. Additionally, detecting bioactive testosterone is complex and has not been used in the clinic. Testosterone and FT, but not bioactive testosterone, were detected in our study, although bioactive testosterone can better reflect the state of testosterone in vivo. Third, although the number of 156 patients with LPEs was sufficient to complete the case–control study, the sample size was small and there was asymmetry of cases between the control and LPE group. These issues are considered as limitations and a reliable conclusion cannot be made. Additionally, according to the definition of LPE, only 10% to 20% of the patients with LPE may ejaculate between 1 to 2 minutes, but most of their IELT values should be < 1 minute. Indeed, the mean IELT in patients with LPE in our study was 1.46 minutes, which suggests that some enrolled patients with LPE might have had acquired PE instead.
The pathophysiology of LPE has not been fully determined. High FT levels and low VLDL levels might be used as an objective indicator to diagnose and classify LPE and be used as an indicator of treatment options (e.g., LPE with low VLDL levels can be directly treated with local anesthetics). Although our findings showed significant changes in some patients with LPE, a larger sample size is necessary to improve the design of different interventions and identify the accuracy of our research results.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211002681 - Supplemental material for Elevated free testosterone levels and reduced very low-density lipoprotein levels in men with lifelong premature ejaculation: a preliminary study
Supplemental material, sj-pdf-1-imr-10.1177_03000605211002681 for Elevated free testosterone levels and reduced very low-density lipoprotein levels in men with lifelong premature ejaculation: a preliminary study by Jun Zhao, Yubo Ma, Xiaoqiang Zhai, Hecheng Li, Ziming Wang and Tie Chong in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Fundamental Research Funds for the Central Universities (xjj 2015085), the Social Development Science and Technology Research project (2016SF-147), and Xi’an Science and Technology +Action Plan (201805096YX4SF30(8)).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
