Abstract
We report a 53-year-old man with metastases of alveolar soft-part sarcoma originated from the mediastinum. He was hospitalized due to lower extremities’ paralysis. Computed tomography scan findings revealed multiple nodules of bilateral lungs, swollen mediastinal lymph nodes, and osteolysis of thoracic vertebrae. We performed spinal decompression and biopsy from vertebra. And, we finally diagnosed this case as metastases of mediastinal alveolar soft-part sarcoma which was removed 10 years ago. Alveolar soft-part sarcoma is rare tumor accounted for 0.5%–1.0% of soft tissue sarcoma that often occurs primarily in the lower extremities and trunk. It is difficult to distinguish between alveolar soft-part sarcoma and paraganglioma, renal cell carcinoma and granular cell tumor morphologically. Periodic acid–Schiff stain and immunohistochemical staining of ASPL-TFE3 are useful in making a definitive diagnosis of alveolar soft-part sarcoma. This case is a rare case of alveolar soft-part sarcoma originated in the mediastinum with local recurrence and distant metastases 10 years after the initial surgery.
Introduction
Alveolar soft-part sarcoma (ASPS) is rare soft tissue sarcoma that often occurs primarily in the lower extremities and trunk. Surgical resection is the only curative treatment for ASPS, and prognosis in patients with metastases is very poor. Although it is difficult to distinguish between ASPS and morphological similar diseases, immunostain using anti-TFE3 antibody is useful for definitive diagnosis. We report a rare case of ASPS originated in the mediastinum with local recurrence and distant metastases 10 years after the initial surgery.
Case
A 53-year-old man was hospitalized for paralysis in the lower extremities. Computed tomography (CT) and magnetic resonance imaging (MRI) findings revealed multiple bilateral lung nodules, swollen mediastinal lymph nodes, and osteolysis of thoracic vertebrae (Figure 1). A spinal decompression was performed, and vertebral biopsies were taken. Before 10 years, a round mediastinal tumor, 3.5 cm in diameter and proximate to the 11th thoracic vertebra, had been removed from the patient and was diagnosed as a benign paraganglioma of the mediastinum. Comparison of the vertebral biopsy specimen with tumor tissue from the initial surgery revealed that both specimens were pathologically identical, and the vertebral tumor was diagnosed as a metastasis from the mediastinal tumor. Histologically, the tumors showed a characteristic pseudoalveolar pattern, composed of large polygonal tumor cells with abundant cytoplasm, vesicular nuclei, marked nucleoli, and low nuclear cytoplasmic ratio (Figure 2(a)). The tumor tissue was re-evaluated using immunohistochemical (IHC) staining. The results were positive for TFE3 (Figure 2(b)) and S100 protein and very focally positive for CD10. However, staining was negative for the neuroendocrine markers, chromogranin A, dopamine beta-hydroxylase, synaptophysin, and CD56. The final diagnosis was revised to alveolar soft-part sarcoma (ASPS) of the mediastinum, with multiple metastases in the lungs, lymph nodes, and vertebrae. The patient died 4 months following biopsy.
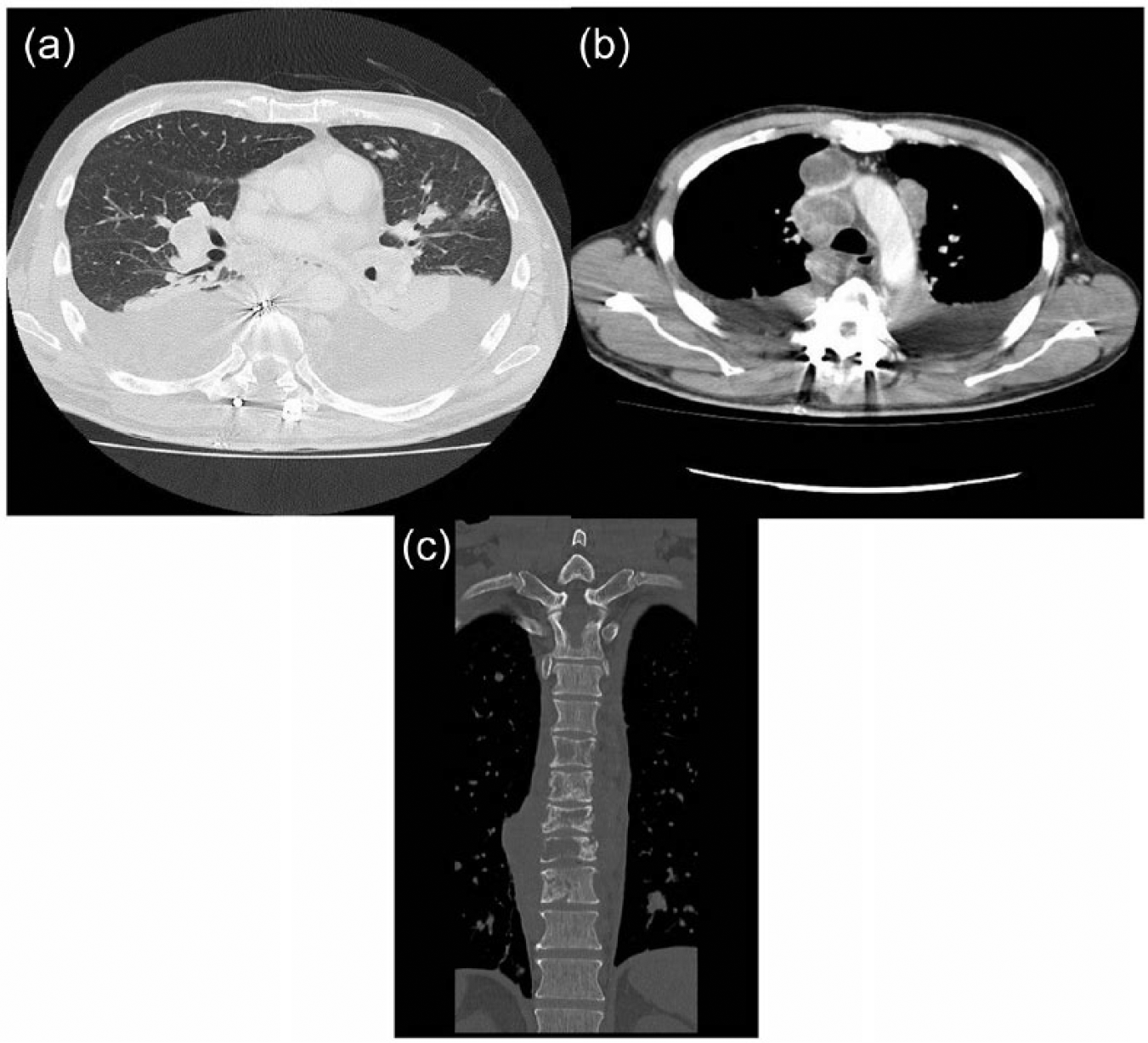
Chest CT scan revealed multiple tumors in bilateral lung: (a) pleural effusion, (b) swollen mediastinal lymph nodes, and (c) destructed thoracic vertebrae.
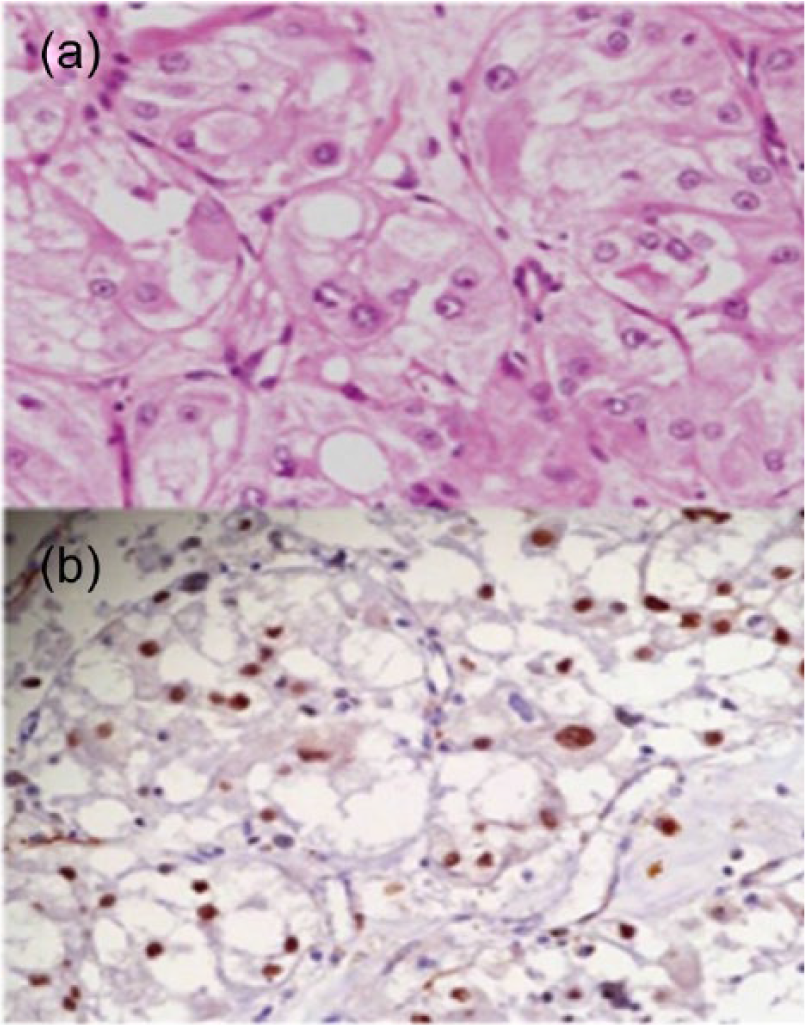
Histopathological finding of hematoxylin and eosin stained section: (a) pseudoalveolar arrangement of large and polygonal cells with abundant cytoplasm and (b) immunostain using anti-TFE3 antibody were positive for most of tumor cells.
Discussion
ASPS is rare, presenting as slow-growing tumors accounting for only 0.5%–1.0% of soft tissue sarcomas and occurs primarily in the lower extremities and trunk of young people and grows slowly. 1 Only two cases of ASPS originating from the mediastinum have been reported. 2 It has been reported that the 5-year survival rate, following surgical resection in patients without metastasis, is 87%. However, the 5-year survival rate is only 20% in patients with metastasis, 1 and it has been reported that metastasis is present in 30%–65% of cases at the time of diagnosis.1,3 Pseudoalveolar arrangement of large polygonal cells with abundant cytoplasm is the typical histopathological finding of ASPS. The differential diagnoses of ASPS are renal cell carcinoma and paraganglioma. Although it is difficult to distinguish between ASPS and these tumors morphologically, Argani et al. have reported that diastase–periodic acid–Schiff (d-PAS) staining can identify diastase-resistant rod-shaped crystals in ASPS, and IHC staining of TFE3 is useful in making a definitive diagnosis. 4 ASPL-TFE3 is the fusion gene product of the chromosomal translocation, der(17)t(X;17)(p11;q25), and is considered to be the most reliable diagnostic feature of ASPS. In this case, although the cytoplasmic crystal structures were not identified by d-PAS staining, IHC staining for TFE3 was positive. As the patient had no history of renal cell carcinoma, and a diagnosis of paraganglioma was immunohistochemically ruled out, the tumor was finally diagnosed as ASPS of the mediastinum. In this case, diagnosis using TFE3 was not made in those days at the initial diagnosis. Nowdays, anti-TFE3 antibody is useful in establishing the diagnosis of ASPS.
Therefore, both PAS stain and immunostain of anti-TFE3 antibody have to be done at the time of initial diagnosis when large and polygonal cells with abundant cytoplasm are observed in the tumor at the initial specimen.
ASPS is not responsive to conventional chemotherapy. Surgical resection is the only curative treatment for primary and metastatic ASPS. Notably, in the recent studies, a use of antiangiogenic agents, such as bevacizumab and tyrosine kinase inhibitors, such as sunitinib, reported as treatment for metastatic ASPS.5–8 Stacchiotti et al. 7 reported nine patients with progressive metastatic ASPS who received oral administration of sunitinib 37.5 mg/day. The results were partial response in five cases, stable disease in three, and progression in one. At present, sunitinib may be the most promising among these therapies.
Footnotes
Acknowledgements
The authors thank Dr Tomoko Mitsuhashi, Hokkaido University Hospital, for immunostain of anti-TFE3 antibody.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
