Abstract
Objective
We evaluated the discriminatory ability of variations in lymphocyte, D-dimer, C-reactive protein (CRP), and lactate dehydrogenase (LDH) serum levels at 48 to 72 hours of hospitalization compared with baseline measurements to predict unfavorable clinical outcomes in patients with COVID-19.
Methods
We analyzed diagnostic test results based on a retrospective cohort to determine the ability of variations (gradients or ratios) in patients’ lymphocyte, D-dimer, CRP, and LDH serum levels taken 48 to 72 hours after hospital admission to predict adverse outcomes such as death, mechanical ventilation, or intensive care unit (ICU) admission developing.
Results
Among 810 patients (56.1% men, age 61.6 ± 16.2 years), 37.5% had at least one adverse outcome; 28.2% required ICU admission, 26.5% required mechanical ventilation, and 19.4% died during hospitalization. In comparing baseline measurements with measurements at 48 to 72 hours, D-dimer, lymphocyte delta, LDH, and CRP had similar discriminatory ability (area under the receiver operating characteristic curve [AUC] 0.57 vs. 0.56, 0.53 vs. 0.57, 0.64 vs. 0.66, and 0.62 vs. 0.65, respectively).
Conclusions
Measuring serum risk markers upon hospital admission can be used to evaluate risk of adverse outcomes in hospitalized patients with COVID-19. Repeating these measurements at 48 to 72 hours does not improve discriminatory ability.
Introduction
On March 11, 2020, the World Health Organization declared COVID-19, an infection caused by SARS-CoV-2, to be a pandemic. 1 As of September 2022, over 603 million people worldwide have been confirmed to be infected. 2 This staggering number has triggered an unparalleled health, political, and economic crisis.
With saturation of emergency and hospitalization services, it is essential to identify effective predictors that permit the early identification of patients with a greater risk of developing complications, such as the need for admission to an intensive care unit (ICU), mechanical ventilation, or death. 3 The presence of lymphopenia (<1000 cells),4,5 elevation of D-dimer above 1000 ng/mL, C-reactive protein (CRP) above 10 mg/dL, and lactate dehydrogenase (LDH) above 350 IU/L,4,6,7 are known markers of poor prognosis, among others.
National and international guidelines recommend measuring the levels of these markers at hospital admission;8–10 however, it is common practice to assess these in follow-up at 48 to 72 hours, with no evidence that variations in these parameters predict the development of adverse outcomes in patients hospitalized with COVID-19.
The objective of this study was to evaluate the discriminatory ability of variations (gradients or ratios) in the serum levels of lymphocytes, D-dimer, CRP, and LDH at 48 to 72 hours of hospitalization in comparison with baseline measurements to predict unfavorable clinical outcomes such as admission to the ICU, mechanical ventilation, or death.
Methods
We investigated the results of diagnostic tests based on a retrospective cohort of patients diagnosed with COVID-19 who were hospitalized at the Hospital Universitario San Ignacio in Bogota (Colombia) between March 2020 and January 2021. We included patients older than 18 years with confirmed infection via reverse transcription polymerase chain reaction and available lymphocyte, D-dimer, LDH, and CRP measurements at admission and at 48 to 72 hours after hospitalization. We excluded patients who required care in the ICU and/or mechanical ventilation at admission and patients with unknown outcomes (i.e., those who were referred to another hospital or requested voluntary discharge). All data were anonymous and all patient details were deidentified. The conduct of this study adhered to the ethical principles outlined in the Declaration of Helsinki. The ethics committees of the Hospital Universitario San Ignacio and the Faculty of Medicine of the Pontificia Universidad Javeriana approved the study in June 2021 (code MI 020-2021). Owing to the retrospective nature of the study, the ethics committees of the hospital and the university waived the requirement for patient informed consent.
Data collection, calculation, and outcomes
The data were obtained from the institutional registry database of patients diagnosed with COVID-19. To develop the registry, we identified patients from the daily lists managed by internal medicine, pulmonology, and infectious diseases physicians. We collected information on various sociodemographic variables, such as age, sex, and race and ethnicity, as well as the time from symptom onset to the time of hospital admission, comorbidities, and the National Early Warning Score (NEWS),11,12 which is used to determine the degree of illness and to prompt critical care intervention. We also systematically recorded the treatment received and patient outcomes in a standardized format using the RedCap® application. To ensure the quality of the data, we monitored the rate of missing data and verified the information when extreme data values were found. Furthermore, we integrated information from the clinical laboratory for measurements of severity markers at admission and at 48 or 72 hours after hospitalization.
The transfer of patients to the ICU was determined by a team of healthcare professionals, including the attending physician and at least two additional physicians. This team assessed the patient's clinical status and the availability of resources before making a decision. To determine variations in the biomarker levels, we calculated the ratio between the measurement at 48 to 72 hours and the baseline measurement for each of the four selected biomarkers. Additionally, we calculated the lymphocyte gradient by subtracting the values at 48 to 72 hours from the admission values. We defined a composite outcome as the occurrence of at least one of the three outcomes of interest: death, mechanical ventilation, or admission to the ICU.
Statistical analysis
Categorical variables are described as absolute and relative frequencies. Continuous variables are presented as mean and standard deviation or as median and interquartile range, depending on the distribution of the data. The Shapiro–Wilk test was used to evaluate the assumption of normality. Receiver operating characteristic (ROC) curves were used to analyze the discriminatory ability of both the baseline measurements and variations in levels of the biomarkers; the area under the ROC curve (AUC) was compared using the method proposed by De Long. 13 The same method was used to calculate the sensitivity and specificity of the different variables and of their ratio (48–72 hours/baseline). Liu’s method 14 was used to define the point with the greatest discriminative ability. Values of p < 0.05 were considered statistically significant. Statistical analyses were performed with Stata software, Release 16 (StataCorp LLC, College Station, TX, USA).
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 15
Results
We identified 1837 patients who were hospitalized between March 2020 and January 2021. After excluding 964 patients who did not have at least two measurements of any risk markers of interest, 26 patients who required care in the ICU or mechanical ventilation at admission, and 26 patients who were referred to another hospital, a total of 810 patients remained for inclusion in the analysis. The baseline characteristics of these patients are shown in Table 1. Most patients were men (56.1%) with an average age 61.6 ± 16.2 years. The main comorbidity was hypertension (41.4%). Most patients were classified as having moderate illness severity (74.9%) according to the NEWS score. Nearly all patients (99.3%) received dexamethasone according to the RECOVERY trial, 16 and no specific antiviral therapy was administered.
Demographic characteristics, variables at admission, and outcomes in patients with COVID-19.
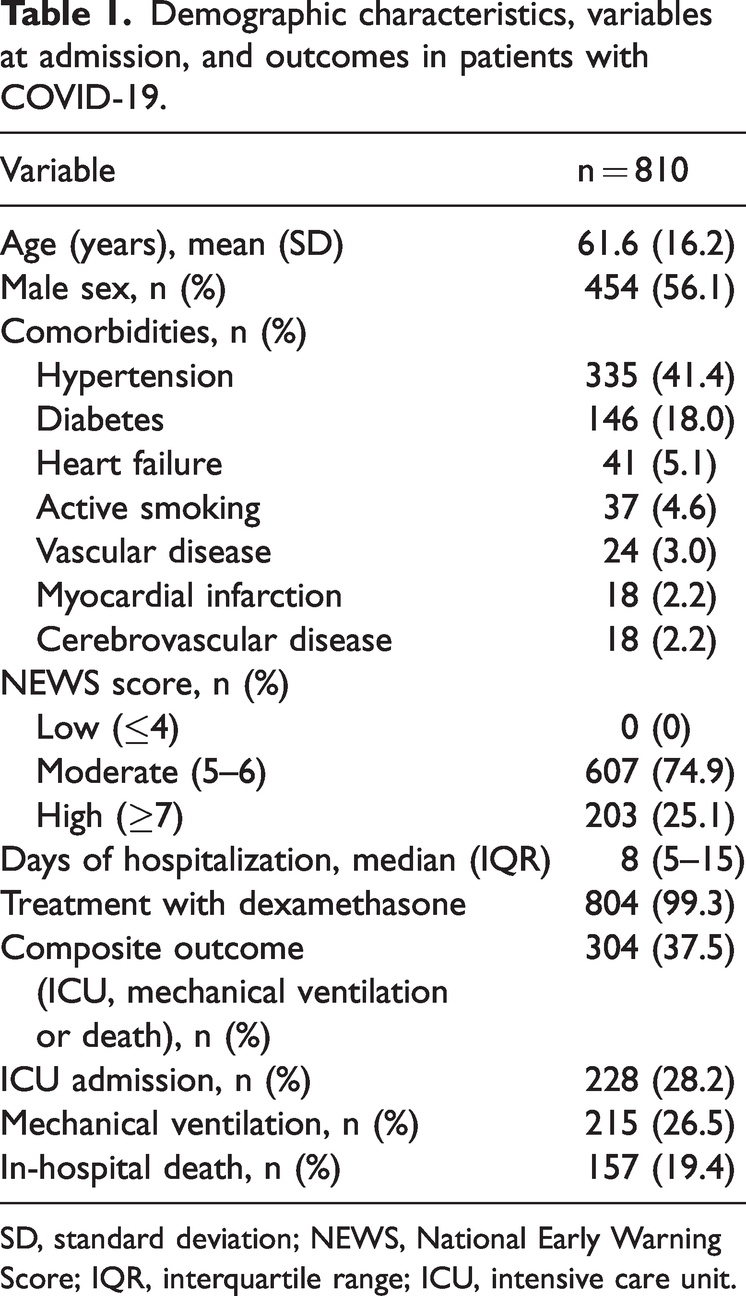
SD, standard deviation; NEWS, National Early Warning Score; IQR, interquartile range; ICU, intensive care unit.
The composite outcome occurred in 37.5% of patients. Of these, 28.2% required ICU admission, 26.5% required mechanical ventilation, and 19.4% died during hospitalization. The median levels of lymphocytes, D-dimer, CRP, and LDH at admission and at 48 to 72 hours of hospitalization are shown in Table 2. A significant change was found in the values of lymphocytes, LDH, and CRP (p < 0.001), but not in D-dimer values.
Values of severity markers at admission and at 48 to 72 hours.

IQR, interquartile range; LDH, lactate dehydrogenase; CRP, C-reactive protein.
Table 3 presents the optimal cutoff points for each risk marker. The cutoff points identified for the baseline measurements were similar to those reported in previous studies, with a lymphocyte count of 1050 cells/µL, D-dimer of 837 ng/mL, CRP of 10.34 mg/dL, and LDH of 359 IU/L. To assess the variations in these markers at 48 to 72 hours, we selected the ratio (measurement at 48–72 hours/baseline measurement) for D-dimer, CRP, and LDH, and the lymphocyte count delta (measurement at 48–72 hours−baseline measurement). The best cutoff points were a decrease in lymphocyte count of 199 cells/µL and a D-dimer ratio of 0.66. Table 3 presents the sensitivity and specificity of each value. Overall, the baseline measurements and the variations at 48 to 72 hours had very similar operating characteristics, with sensitivities ranging between 60% and 75% and specificities between 50% and 65%.
Optimal cutoff points of admission values and their variations to predict the composite outcome of death, mechanical ventilation, or admission to the ICU.

LDH, lactate dehydrogenase; CRP, C-reactive protein.
Figure 1 shows the results of analysis regarding the ability of risk marker measurements at admission to identify patients with outcomes of in-hospital mortality. LDH (AUC 0.66; 95% confidence interval [CI] 0.60, 0.72), CRP (AUC 0.68; 95% CI 0.62, 0.73), and D-dimer (AUC 0.68; 95% CI 0.62, 0.72) had a similar discriminatory ability. However, absolute lymphocyte count had the poorest discriminatory ability (AUC 0.61; 95% CI 0.56, 0.66).
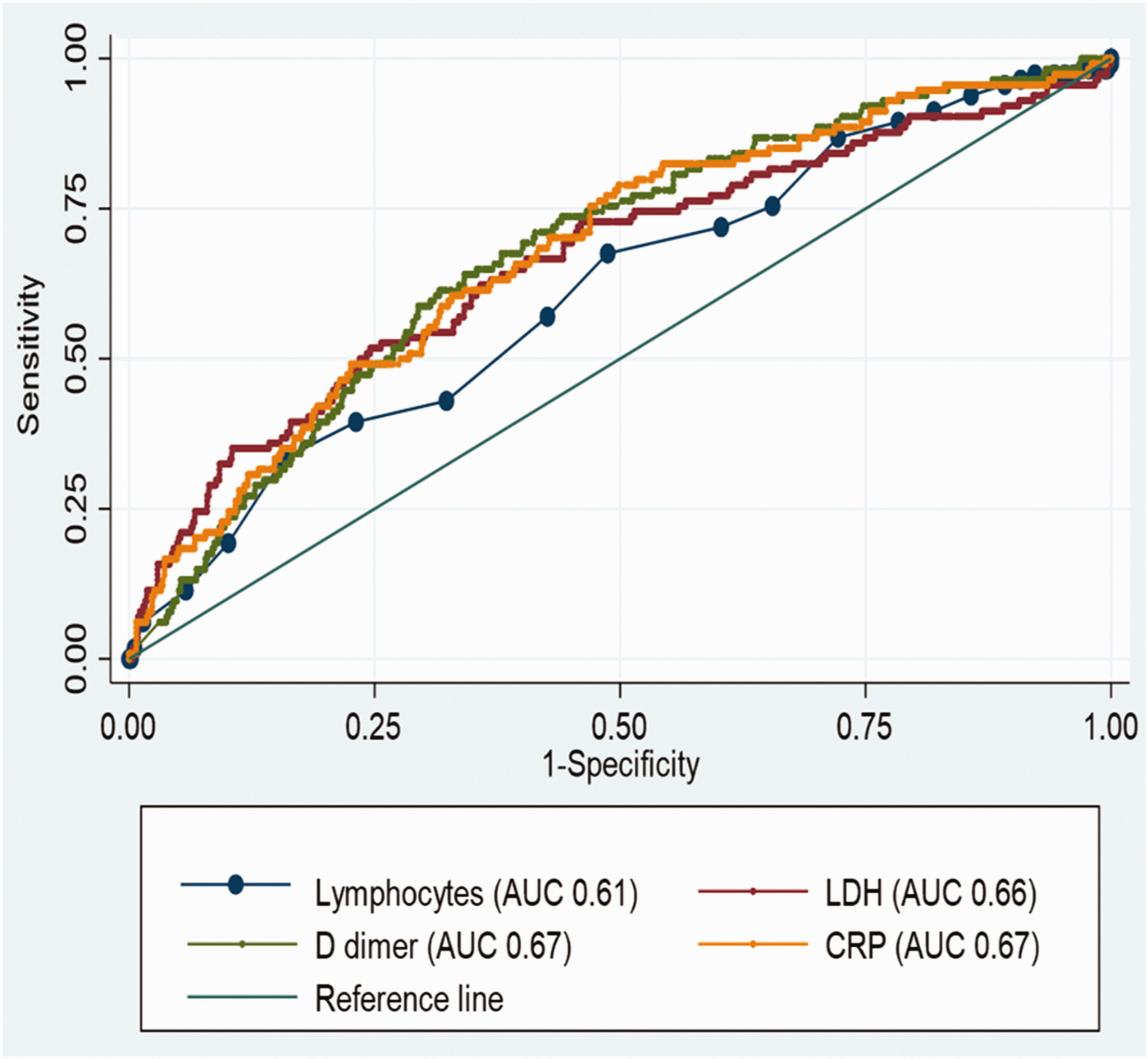
Discriminatory ability of severity parameters at admission.
We compared the discriminatory ability for mortality of the measurements at admission with the variations at 48 to 72 hours (delta and/or ratio) (Figure 2). All markers had similar discriminatory ability: D-dimer (AUC 0.57 vs. 0.56) (Figure 2a), lymphocyte count delta (AUC 0.53 vs. 0.57) (Figure 2b), CRP (AUC 0.62 vs. 0.65) (Figure 2c), and LDH ratio (AUC 0.64 vs. 0.66) (Figure 2d).

Comparison of discriminatory ability for mortality of measurements at admission with variations at 48 to 72 hours (delta and/or ratio). Continued.(a) Discriminatory ability of D-dimer at admission compared with D-dimer ratio; (b) discriminatory ability of lymphocytes at admission compared with delta lymphocytes; (c) discriminatory ability of CRP at admission compared with CRP ratio and (d) discriminatory ability of LDH at admission compared with LDH ratio.
In the sensitivity analysis, taking into account the composite outcome, the discriminatory ability of the different markers was again similar, with the exception of D-dimer, where the basal value showed better discrimination than the measurement at 48 to 72 hours (AUC 0.69 vs. 0.53, p = 0.014) (Figure 3).

Discriminatory ability of D-dimer to predict composite outcome.
All four tests were found to have similar discriminatory ability for patients admitted to the hospital less than 7 days from symptom onset compared with those admitted 7 days or more after symptom onset.
A subgroup analysis revealed that CRP, LDH, and D-dimer had similar discriminatory abilities for mortality in patients with or without diabetes and hypertension in comparison with the general population. However, lymphocyte count exhibited a better discriminatory ability in patients without comorbidities (Supplementary Table 1). Similarly, the follow-up measurements of CRP, LDH, and D-dimer did not enhance the discriminatory ability compared with baseline measurements in the various comorbidity subgroups.
Discussion
In our setting, the repeated measurement of some prognostic markers of severity in COVID-19 is a common practice, despite no studies having evaluated the predictive ability of these measurements beyond baseline measurements. Our study suggests that repeated measurements of lymphocytes, D-dimer, CRP, and LDH do not significantly improve the ability to identify patients who could develop adverse outcomes in comparison with baseline measurements.
The results of the predictive cutoff points for each of the studied variables at admission were similar to those reported in various publications.5–7 Of the four variables analyzed, the one with the best ability to predict adverse outcomes upon patient admission was CRP, followed by LDH. In a study by Koozi et al., CRP levels greater than 10 mg/dL on admission were associated with mortality in the ICU and at 30 days. 17 In a pooled analysis, Henry et al. demonstrated that elevated LDH levels are associated with worse outcomes, specifically, a six-fold increased likelihood of severe disease and a 16-fold increased risk of dying. 18
Some publications have reported differences in the values of these tests during hospitalization and according to the severity of disease between patients with COVID-19 who died and survivors,19–22 but not when variations in these values during the first hours of care could predict the risk of the composite outcome. In our study, when evaluating the values at 48 to 72 hours for lymphocyte count, D-dimer, and CRP, the discriminatory ability did not improve significantly. The only marker that was slightly better in discriminating patients with in-hospital mortality was the LDH ratio, with a cutoff point of 0.89. However, the magnitude of the improvement was minimal, so the clinical utility of these serial measurements is debatable. According to our review of the published literature, this is the first study with this finding. Thus, additional studies will be required to validate the present results.
In this study, the D-dimer ratio was the least accurate marker in predicting adverse patient outcomes. Our data suggest that it is not useful to take new measurements at 48 to 72 hours and that the baseline values should be interpreted carefully, considering higher cutoff points for suspected pulmonary embolism, as other authors have suggested.23,24
Regarding the time to hospital admission from the onset of symptoms (fewer than or more than 7 days), there was no difference in the predictive ability of these measurements compared with measurements at admission, which suggests that the usefulness of these markers does not depend on this variable.
A strength of this study is the sample size, which was larger than previously published series; this allowed us to accurately estimate the characteristics of the studied markers. However, there are several study limitations to consider. The collected data were mostly for the year 2020, when the vaccination program had not yet begun in Colombia. New studies should be conducted to evaluate whether the discriminatory ability of these risk markers changes in vaccinated patients or in those infected with new strains of SARS-CoV-2.
In conclusion, the measurement of four serum risk markers at hospital admission could predict the risk of ICU admission, mechanical ventilation, and death among hospitalized patients with COVID-19 pneumonia. Repeating these measurements 48 to 72 hours after admission did not improve the predictive ability and could unnecessarily increase health care costs or lead to confusion in decision-making by clinicians.
