Abstract
Objective
Endoscopic resection (ER) of gastric gastrointestinal stromal tumors (gGISTs) is a commonly used treatment; however, it is associated with a risk of conversion to laparoscopic resection (LR). This study was performed to identify factors influencing conversion from ER to LR and the effects of conversion on outcomes.
Methods
The clinicopathological features of patients treated for gGISTs from March 2010 to May 2021 were retrospectively collected. Endpoints included the determination of risk factors associated with LR conversion, with comparisons of surgical outcomes with and without conversion. Propensity score matching was performed to compare the two groups.
Results
In total, 371 gGISTs were analyzed. Sixteen patients required conversion from ER to LR. Propensity score matching demonstrated that invasion depth (muscularis propria with exophytic growth) and gGIST size (≥3 cm) were independent risk factors for conversion to LR. The procedure duration (median, 160.5 vs. 60.0 minutes), postoperative hospitalization duration (median, 8 vs. 6 days), and postoperative fasting duration (median, 5 vs. 3 days) were significantly longer in patients who underwent conversion to LR.
Conclusions
Accurate preoperative measurements of tumor size and invasion depth may help determine more appropriate surgical approaches for patients with gGISTs.
Keywords
Introduction
Typical treatments for gastric gastrointestinal stromal tumors (gGISTs) have historically included gastrectomy or wedge resection.1,2 With advanced endoscopic techniques and accumulated surgical experience, endoscopic resection (ER) of gGISTs is being increasingly performed. Several cohort studies have shown that when compared with laparoscopic resection (LR), ER of gGISTs causes less trauma, results in faster postoperative recovery times, and has lower hospitalization costs.3–7 However, perioperative complications may occur even when ER is performed by experienced surgeons, potentially requiring time-consuming conversion to LR. Although many studies have reported complications such as perforation and hemorrhage,8,9 few have outlined the risks of unintended conversion from ER to LR during treatment of gGISTs. Therefore, we sought to identify the incidence and risk factors associated with conversion from ER to LR during treatment of gGISTs and to elucidate patients’ clinical outcomes.
Methods
Patients
We performed a retrospective study using the medical records of 421 patients with gGISTs who underwent ER at the First Affiliated Hospital of Soochow University, Suzhou, China from March 2010 to May 2021. The inclusion criteria were a pathological and immunohistochemical diagnosis of gGIST; normal routine preoperative blood tests, coagulation profile, and electrocardiogram; no lymph node or distant metastases; and the presence of solitary lesions in the stomach. All demographic information, perioperative parameters, and outcomes were obtained from the patients’ electronic medical records. The primary endpoint was the rate of conversion from ER to LR during treatment of gGISTs. The secondary endpoints were risk factors associated with this conversion and detailed analyses of factors associated with surgical outcomes in patients without conversion to LR. All patients provided written informed consent before surgery, and all patients’ details have been de-identified. The study protocol was approved by the ethics committee of The First Affiliated Hospital of Soochow University. The reporting of this study conforms to the STROBE guidelines. 10
Endoscopic equipment and procedures
All patients received general anesthesia and endotracheal intubation. ER was performed using a dual knife (KD-650L; Olympus, Tokyo, Japan), an insulated-tip knife (Erbe, Tübingen, Germany), or a combination. Other equipment included injection needles, hot biopsy forceps, metallic clips, snares, nylon loops, and an Over-the-Scope-Clip System (Ovesco Endoscopy, Tübingen, Germany). We measured the largest tumor diameter and classified the gGISTs using the modified National Institutes of Health (NIH) risk criteria. 11 Two types of ER were performed: endoscopic submucosal dissection (ESD) and endoscopic full-thickness resection (EFTR). These endoscopic techniques have been described in previous reports.12,13
Data collection
The following patient information was obtained from the electronic medical records: age, sex, body mass index, tumor size, tumor location, invasion depth, primary symptoms, American Society of Anesthesiologists score, procedure duration, endoscopic method, modified NIH classification, postoperative hospitalization duration (days), postoperative fasting duration (days), intraoperative complications, postoperative complications, en bloc R0 resection rate, follow-up duration (months), postoperative mortality rate, and recurrence rate.
Definitions
We defined the procedure duration as the amount of time between marking the lesion circumference and completing gastric defect closure after all materials were removed. Resection of the tumor in its entirety, with no residual tumor tissue at the lateral or vertical margins, was defined as en bloc R0 resection. 14 Intraoperative complications were defined as perforations and severe intraoperative bleeding (requirement for repeated endoscopic hemostasis with a postoperative hemoglobin drop of ≥2 g/dL or requirement for surgical assistance). 15 Postoperative complications were defined as delayed bleeding episodes confirmed by emergency endoscopy, delayed perforation confirmed by X-ray or computed tomography, and postoperative infection. All procedures were performed by endoscopists with different experience levels. Experience was defined by the number of surgical cases performed: <50 was defined as inexperienced, and ≥50 was defined as experienced.
Statistical analysis
Categorical variables are expressed as frequency and percentage and were analyzed using the chi-square test or Fisher’s exact test. Continuous variables are expressed as median and interquartile range and were compared using the Mann–Whitney U test. Patients in the conversion group were paired 1:4 to patients in the non-conversion group using propensity score matching (PSM) to balance potential covariates. Univariable analyses were performed between the non-conversion and conversion groups. Univariate analyses were performed, and variables with statistically significant differences (p < 0.05) were included in the multivariate regression analyses. All analyses were performed with SPSS version 26 (IBM Corp., Armonk, NY, USA).
Results
Patients’ baseline characteristics
A flow chart of the study protocol is shown in Figure 1. The patients’ characteristics and clinicopathologic factors are highlighted in Table 1. The patients comprised 146 men and 225 women with a median age of 60 years (interquartile range, 53–66 years). A total of 309 (83.3%) patients had tumors of <3 cm, while 62 (16.7%) had tumors of >3 cm. Approximately two-thirds (64.4%) of gGISTs were located at the fundus of the stomach. ESD and EFTR procedures were performed in 45.6% and 54.4% of patients, respectively. Approximately 50% of patients had a body mass index within the normal range. The majority of gGISTs (87.6%) originated within the muscularis propria (MP), and 258 (69.5%) procedures were performed by experienced endoscopists. A total of 91.1% of patients had mild to moderate systemic disease (American Society of Anesthesiologists score of ≤2). The main reasons for gastroscopy were abdominal discomfort (50.9%) and physical examination (44.7%).

Study design flow chart.
Patients’ clinicopathologic characteristics (conversion vs. non-conversion group) before and after PSM. Data are presented as median (interquartile range) or n (%).
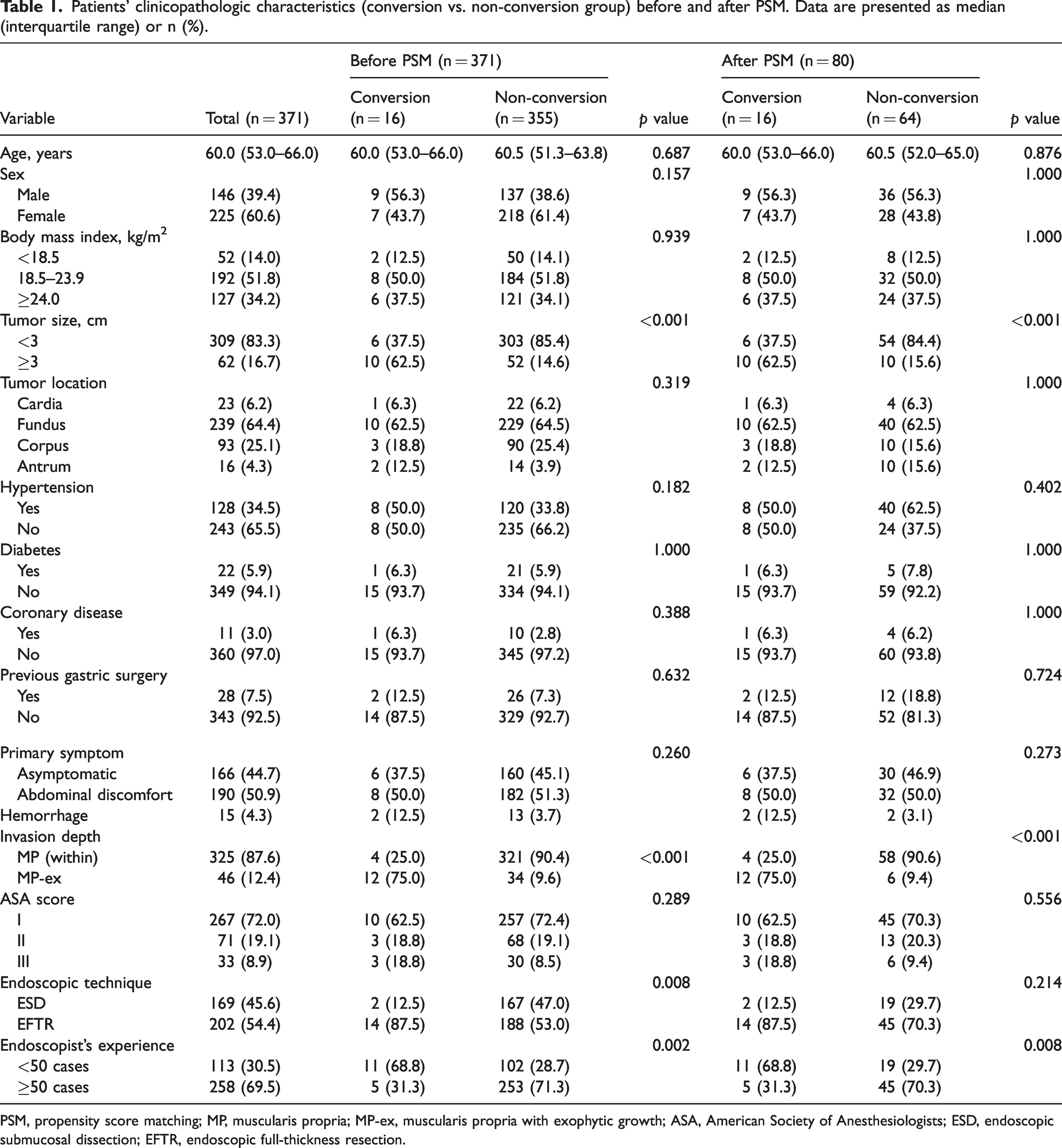
PSM, propensity score matching; MP, muscularis propria; MP-ex, muscularis propria with exophytic growth; ASA, American Society of Anesthesiologists; ESD, endoscopic submucosal dissection; EFTR, endoscopic full-thickness resection.
Risk factors for conversion from ER to LR
Of 371 patients, 16 (4.3%) underwent conversion from ER to LR. The most common reason for conversion was severe intraoperative bleeding that could not be controlled by endoscopy (56.3%), failure to close a gastric perforation (31.3%), and accidental dropping of a tumor into the abdominal cavity (12.5%). Before PSM, univariate analyses revealed that tumor size, invasion depth, endoscopic technique, and endoscopist’s experience were risk factors for conversion to LR (Table 1). The proportion of >3 cm tumors was significantly higher in the conversion group than in the non-conversion group (62.5% vs. 14.6%, respectively; p < 0.001). Patients treated by EFTR were more prone to require conversion to LR (p = 0.008). In terms of gGIST invasion depth, 12 of the 16 patients in the conversion group had lesions with an invasion depth that reached the MP with exophytic growth (MP-ex). In the non-conversion group, however, 34 patients had lesions with an invasion depth that reached the MP-ex, accounting for only 9.6%. This difference was statistically significant (p < 0.001). In the conversion group, 5 (31.3%) procedures were performed by experienced endoscopists, whereas in the non-conversion group, 253 (71.3%) procedures were performed by experienced endoscopists. This difference was also statistically significant (p = 0.002). Multivariate analyses (Table 2) showed that gGISTs originating from MP-ex exhibited a significantly higher possibility of conversion to LR than superficial gGISTs (odds ratio (OR), 16.026; 95% confidence interval (CI), 2.949–87.092; p < 0.001), and tumor size of ≥3 cm was an independent risk factor for conversion to LR (OR, 6.747; 95% CI, 2.062–22.071; p = 0.002). After PSM, univariate analyses revealed that tumor size, invasion depth, and endoscopist’s experience were risk factors for conversion to LR (Table 1). Multivariate analyses (Table 2) showed that gGISTs originating from MP-ex exhibited a significantly higher possibility of conversion to LR than superficial gGISTs (OR, 18.146; 95% CI, 3.135–92.172; p = 0.001), and tumor size of ≥3 cm was an independent risk factor for conversion to LR (OR, 5.667; 95% CI, 1.982–18.310; p = 0.002).
Multivariate analysis of conversion to laparoscopic resection of gastric gastrointestinal stromal tumors before and after PSM.

PSM, propensity score matching; OR, odds ratio; CI, confidence interval; MP, muscularis propria; MP-ex, muscularis propria with exophytic growth; ESD, endoscopic submucosal dissection; EFTR, endoscopic full-thickness resection.
Comparison of operative data and postoperative outcomes in patients with and without conversion from ER to LR
Comparisons of the operative data and postoperative outcomes between the two patient groups are shown in Table 3. Before PSM, no significant between-group differences were identified regarding postoperative complications, en bloc R0 resection, or recurrence. The procedure duration was significantly longer for patients requiring conversion to LR (median, 160.5 vs. 58.0 minutes for patients without conversion, p < 0.001). The proportion of patients with severe intraoperative bleeding was significantly higher in the conversion group than in the non-conversion group (50.0% vs. 0.3%, respectively; p < 0.001). The rate of intraoperative perforation was also significantly higher in the conversion group than in the non-conversion group (87.8% vs. 53.0%, respectively; p = 0.008). The postoperative hospitalization duration (median, 8 vs. 6 days; p < 0.001) and postoperative fasting duration (median, 5 vs. 3 days; p < 0.001) were significantly longer in the conversion than non-conversion group. In terms of the modified NIH risk criteria, the conversion group was dominated by intermediate- or high-risk cases (68.8%), and the non-conversion group was dominated by very low- or low-risk cases (95.2%) (p < 0.001). In total, 305 patients were successfully followed up (12 in the conversion group and 293 in the non-conversion group). No significant between-group difference was found in the follow-up duration (median, 42 vs. 42 months). After PSM, the operative data and postoperative results between the two groups were similar to those before PSM.
Operative data and postoperative outcomes (conversion vs. non-conversion group) before and after PSM. Data are presented as median (interquartile range) or n (%).
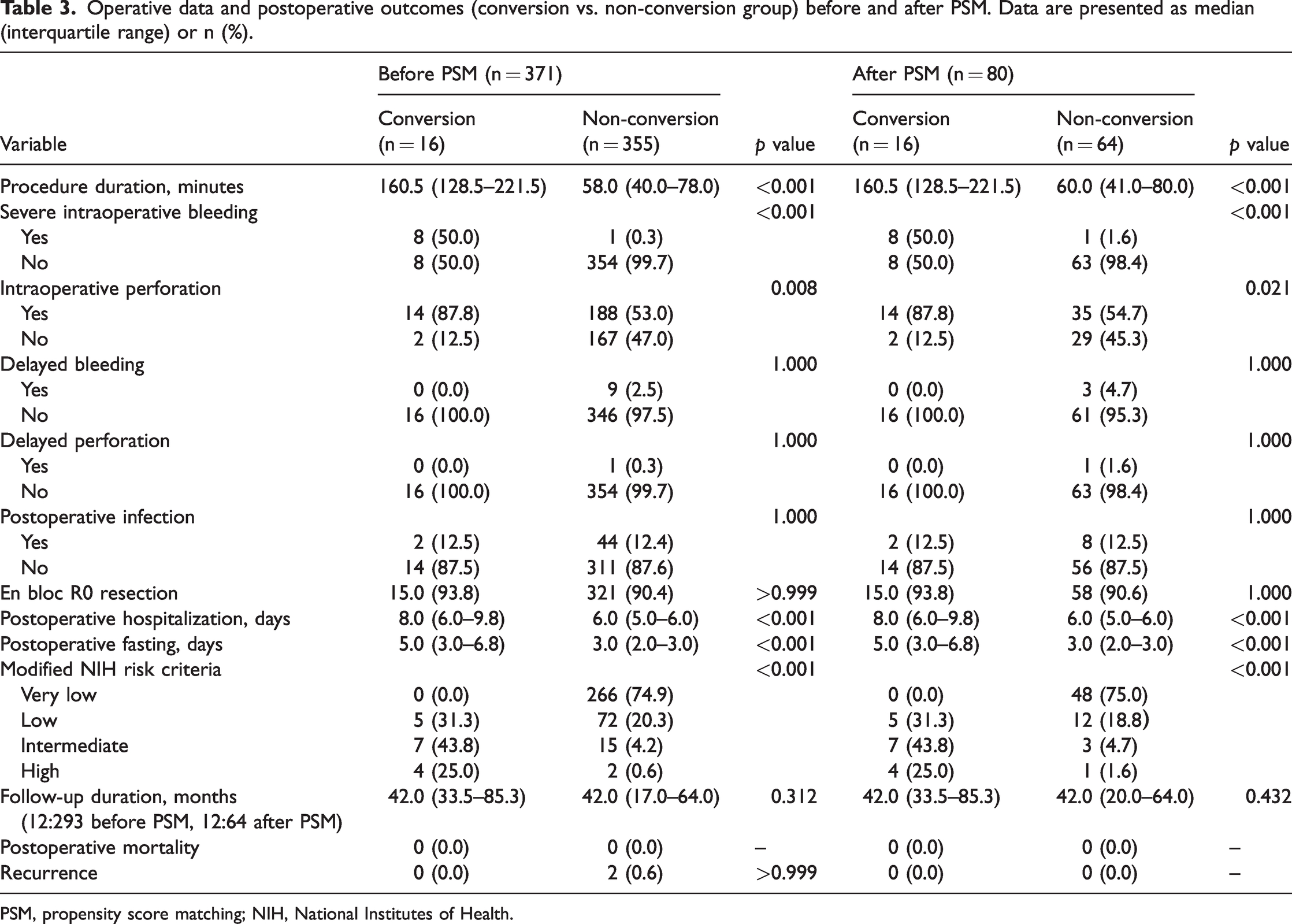
PSM, propensity score matching; NIH, National Institutes of Health.
Distribution of clinical complications across different gGIST sizes and invasion depths
Larger gGISTs (≥3 cm) were more vulnerable to severe intraoperative bleeding (9.7% vs. 1.0%, p = 0.001), intraoperative perforation (74.2% vs. 50.5%, p < 0.001), and delayed bleeding (6.5% vs. 1.6%, p = 0.046) when compared with tumors of <3 cm (Table 4). However, no significant differences were observed in terms of delayed perforation or postoperative infection. The probabilities of severe intraoperative bleeding and perforation in gGISTs originating from MP-ex were significantly higher than for superficial gGISTs (15.2% vs. 0.6%, p < 0.001; 100.0% vs. 48.0%, p < 0.001). No significant between-group differences were identified in terms of postoperative complications.
Distribution of clinical complications according to different gastric gastrointestinal stromal tumor sizes and invasion depths. Data are presented as n (%).

MP, muscularis propria; MP-ex, muscularis propria with exophytic growth.
Discussion
Because of the malignant potential of GISTs, European and Japanese guidelines recommend resection once a histological diagnosis of a GIST has been confirmed, regardless of its diameter.16,17 With the improved development and application of endoscopic ultrasound and minimally invasive endoscopic technologies, more gGISTs are being detected and effectively treated.17,18 A recent Chinese consensus document provided two endoscopic indications for gGIST treatment 19 : (1) GISTs of ≤2 cm with a short period of tumor enlargement or a strong patient desire for resection and (2) low-risk GISTs of 2 to 5 cm. However, ER of a gGIST is more challenging than ER of a mucosal lesion and requires substantial skills because of the risk of requiring conversion to LR. Few studies to date have reported risk factors associated with conversion from ER to LR for gGISTs; therefore, we identified and characterized these risk factors in the present study.
Previous studies have revealed variable incidence rates of conversion from ER to LR for gGISTs.6,19–21 Jeong et al. 20 reported that 5 of 32 (15.6%) patients with gGISTs undergoing ER were converted to LR. Dai et al. 6 reported that the rate of conversion from ER to LR was 0.8% (2/262) in their cohort. Huang et al. 21 and Wu et al. 5 reported that none of their patients underwent conversion to LR. In our study, the percentage of patients requiring conversion to LR was 4.3% of 371 patients. We identified two possible reasons for the differences in these incidence rates. First, in the study by Jeong et al., 20 the ESD technique was used to remove tumors, whereas Huang et al. 21 and Wu et al. 5 used the EFTR technique. The EFTR technique has a wider range of indications, and even if the tumor adheres to the gastric wall, it does not require conversion to LR. Second, the different incidence rates may have been related to the small sample sizes. Thus, increasing the sample sizes may generate more accurate results.
In our study, gGISTs of ≥3 cm and an invasion depth to MP-ex were independent risk factors for conversion to LR. We found that 16.1% (10/62) of gGISTs measuring ≥3 cm were converted to LR. In contrast, this proportion was only 1.9% (6/309) for gGISTs measuring <3 cm. Additionally, larger gGISTs were more vulnerable to severe intraoperative bleeding and perforation than tumors of <3 cm (9.7% vs. 1.0%, p = 0.001; 74.2% vs. 50.5%, p < 0.001) (Table 4). When severe intraoperative bleeding could not be endoscopically controlled and intraoperative endoscopic perforation repair failed, conversion to LR was required. We observed that 26.1% (12/46) of patients with gGISTs originating from MP-ex were converted to LR, while only 1.2% (4/325) of patients with gGISTs originating within the MP were converted to LR. gGISTs with MP-ex invasion had a higher probability of severe intraoperative bleeding and perforation when undergoing ER than did gGISTs with MP invasion (15.2% vs. 0.6%, p < 0.001; 100.0% vs. 48.0%, p < 0.001) (Table 4). Deep gGISTs were removed using the EFTR technique, and repair of large perforations was difficult; these factors increased the risk of conversion to LR.
Undoubtedly, learning curves can objectively reflect an endoscopist’s experience.22,23 Sun et al. 24 reported that for endoscopists competent with basic endoscopic intervention skills, the learning curves for ESD of submucosal tumors should be achieved after approximately 32 cases. In our study, the endoscopists’ experience was differentiated according to the number of ER procedures performed for gGISTs (50 cases). Although the univariate analyses showed that the endoscopist’s experience affected conversion to LR, the multivariate analyses showed that previous experience was not an independent risk factor for conversion. This may be because when inexperienced endoscopists encounter intraoperative bleeding or perforation that cannot be resolved, they seek help from experienced endoscopists, thereby reducing the risk of conversion to LR. Additionally, we observed that the endoscopic method was not associated with conversion to LR, although the specific steps varied. Thus, even if an intraoperative perforation occurred, as long as the perforation was not too large and no serious bleeding occurred, it could be well-managed under endoscopy.
Wu et al. 5 reported that laparoscopic surgery had a higher risk of conversion to open laparotomy when gGISTs were located in the posterior wall of the gastric fundus near the cardia. The location of the gGIST is a key factor affecting LR difficulty. However, we observed that the requirement for conversion from ER to LR was not associated with the location of the gGIST. In our study, nine patients required conversion to LR because of difficulties with intraoperative bleeding control by endoscopy. Five patients underwent conversion to LR because of failure to close the perforation. In two cases, conversion to LR was performed because the tumor accidentally fell into the abdominal cavity and could not be found by endoscopy. Jeong et al. 20 reported that 15.6% (5/32) of patients with gGISTs treated with ESD underwent conversion to LR because the tumor had adhered to the gastric wall. However, Huang et al. 21 and Wu et al. 5 reported no cases of conversion to LR for gGISTs with the EFTR approach. When compared with surgery, the EFTR technique is safe, generates less intraoperative blood loss, provides faster postoperative recovery of bowel function, and is relatively inexpensive.25,26 Therefore, with the development of improved endoscopic technology, tumor adhesion to the gastric wall is no longer a risk factor for conversion from ER to LR during treatment of GISTs.
In our study, the procedure, postoperative hospitalization, and postoperative fasting durations were significantly longer in patients who required conversion to LR. The proportion of patients with severe intraoperative bleeding was significantly higher in the conversion group than in the non-conversion group (50.0% vs. 0.3%, respectively; p < 0.001). The intraoperative perforation rate was also significantly higher in the conversion group than in the non-conversion group (87.8% vs. 53.0%, respectively; p = 0.008). However, no between-group differences were observed in postoperative complications, en bloc R0 resection, follow-up duration, or recurrence. Although the long-term clinical efficacy was comparable between the two groups, the non-conversion group had obvious advantages in terms of short-term efficacy.
Our study had two main limitations. First, this was a single-center retrospective study with a relatively small sample size, and only 16 patients underwent conversion to LR. The review of the medical records may have been associated with recall bias, potentially affecting the results. A prospective multicenter large cohort study will be required to validate our results. However, we used the PSM method to balance the potential covariates to some extent. Second, the median follow-up time was 42 months (range, 3–139 months), and only two recurrences were recorded during this period; therefore, we cannot draw conclusions related to recurrence.
In summary, ER is a common treatment strategy for patients with gGISTs. Because conversion from ER to LR can result in compromised surgical outcomes, an awareness of risk factors contributing to conversion to LR is crucial for the surgical management of patients with gGISTs. Tumor size (≥3 cm) and invasion depth (MP-ex) were independent risk factors for conversion from ER to LR during treatment of gGISTs in this study. Our experience suggests that a precise preoperative assessment of tumor size and invasion depth is important to avoid the need for conversion to LR.
Footnotes
Acknowledgment
The authors thank the patients with gastric stromal tumors for their participation and cooperation.
Author contributions
Luojie Liu and Ye Ye collected the data. Luojie Liu and Xiaodan Xu interpreted the data. Luojie Liu drafted the manuscript. Rui Li and Dongtao Shi revised the manuscript. Rui Li and Weichang Chen conceived and designed the study.
Data availability statement
The data that support the findings of this study are available on request from the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Health Personnel Training Project of Soochow (GSWS2020109) and the Changshu Science and Technology Project (CS202116).
