Abstract
Background
Endoscopic resection (ER) is a proven treatment for gastric gastrointestinal stromal tumors (gGISTs). We aimed to assess the learning curve (LC) associated with ER for gGISTs and identify determinants.
Methods
We conducted an analysis of 289 patients who underwent the ER of gGISTs by an experienced endoscopist. To characterize the LC, we employed cumulative sum analysis of the duration of surgery. The participants were divided into an early phase (cases 1–50) and a later phase (case 51–289), which were compared. Furthermore, we identified risk factors for the conversion from endoscopic to laparoscopic resection (LR).
Results
The durations of surgery and hospitalization were shorter, and there were fewer complications and fasting days in the later phase. The conversion rates to LR were 6.0% and 2.5% in the early and later phases, respectively. The tumor diameter (≥3.0 cm) and invasion beyond the muscularis propria were significant risk factors for conversion to LR (odds ratio 17.92, 95% confidence interval 2.66–120.87; and 58.03, 6.40–525.84; respectively).
Conclusions
The LC for ER of gGISTs lasts for approximately 50 cases. In addition, tumors ≥3.0 cm in diameter and those that invade beyond the muscularis propria are more likely to require conversion to LR.
Keywords
Introduction
Endoscopic resection (ER) is a widely used treatment for early gastric cancers (EGCs) and gastric submucosal tumors (SMTs).1,2 Gastric gastrointestinal stromal tumors (gGISTs) are the most frequently encountered type of SMT in the upper digestive tract. 3 ER has been demonstrated to be an efficient and safe method of managing gGISTs, offering advantages such as swift postoperative recovery, a shorter hospital stay, and lower costs.4,5 However, owing to the malignant potential of gGISTs, regular surveillance is required for lesions <2 cm, which can impose a psychological burden on patients. Consequently, there has been a gradual increase in the number of ER procedures performed for the treatment of gGISTs during recent years. 6
Performing ER for gGISTs requires advanced endoscopic skills, and therefore inexperienced endoscopists may require assistance. This procedure carries inherent risks, including of severe intraoperative hemorrhage, perforation, and the need for conversion to laparoscopic resection (LR). Older patients, and in particular those with comorbidities, are at a higher risk of complications, including perforation, during endoscopic procedures. Furthermore, when patients with gastrointestinal stromal tumors present with acute gastrointestinal bleeding, the challenges associated with endoscopic treatment become even greater, and the risk of significant bleeding during the procedure is higher. 7 Therefore, the development of guidelines for endoscopists to achieve proficiency in ER for gGIST is necessary to improve patient safety, given the complexity of the procedure and the potential complications associated.
A study conducted by Sun et al. 8 showed that the performance of approximately 32 procedures is required for an endoscopist to complete the LC for the ER of gastric SMTs. Similarly, Yoshida et al. 9 found that novice endoscopists achieve a stable state after performing approximately 30 ER procedures on gastric lesions. However, it is important to note that the sample sizes in these studies were relatively small. Moreover, the LC for the ER for gGISTs performed by a single endoscopist in a high-volume center has not been studied. To address these limitations, we aimed to study a larger number of cases and to comprehensively evaluate the LC for the ER of gGISTs, thereby providing more robust and reliable insights into this LR and improving understanding of the optimal way of implementing this procedure in an institution.
Material and methods
Patients
We conducted a retrospective study of patients who underwent preoperative computed tomography (CT) and endoscopic ultrasonography (EUS) examinations to evaluate the location, size, invasion depth, and presence of metastasis of their tumor. In addition, comprehensive preoperative testing, including a complete blood count, coagulation profile, and electrocardiography, were performed. The inclusion criteria were as follows: tumor <5.0 cm in diameter, without lymph node or distant metastasis. Consecutive patients who underwent the ER of gastric gastrointestinal stromal tumors (gGISTs) by a single endoscopist between August 2012 and December 2022 were studied. All the procedures were performed by a single endoscopist (DTS), who had performed approximately 500 colonic endoscopic mucosal resections and 100 gastric endoscopic submucosal dissections (ESD) before performing their first ER procedure for gGIST. The demographic, intraoperative, and postoperative data were retrospectively collected for each participant. The study protocol was approved by the ethics committee of the First Affiliated Hospital of Soochow University, and all the participants provided their written informed consent for the procedure and to participate in the study. The reporting of this study conforms to STROBE guidelines. 10
Endoscopic resection procedures and equipment
A general anesthetic was administered to all the participants, and they all underwent endotracheal intubation. We used two ER techniques: ESD and endoscopic full-thickness resection (EFTR). ESD was used for the treatment of gGISTs that originated from the muscularis mucosa (MM) or muscularis propria (MP) and protruded into the lumen, whereas GISTs originating from deep to the MP that showed extraluminal growth and those that could not be separated from the serous layer during ESD were treated using EFTR. The ER procedures were performed as previously described.11,12 Figure 1 illustrates the process of EFTR for gGISTs. We used a single-channel endoscope (GIF-Q260J, Olympus, Tokyo, Japan) with a transparent cap attached to the endoscope tip. The other equipment used included dual knives (KD-650L; Olympus, Japan), insulated-tip knives (KD-611L; Olympus), a high-frequency generator device (ERBE VIO 200D, Erbe Elektromedizin, Tübingen, Germany), a carbon dioxide insufflator, hot biopsy forceps, injection needles, metallic clips, over-the-scope-clips (OTSCs), and nylon loops.

Illustration of the stages of EFTR for the treatment of a gGIST. (a) Tumor located in the fundus; (b) The mucosal layer was incised using a dual knife; (c) Peripheral incision of the mucosal layer; (d) Exposure of the tumor body; (e) Incision through the full thickness of the gastric wall; (f) Complete excision of the tumor using an IT knife; (g) Closure of the wound surface using an OTSC and (h) Resected gGIST.
Definitions
The duration of surgery was defined as the time from the start of submucosal injection until the closure of the defect was complete. The origin of the tumor was identified by preoperative EUS, and its size and location were evaluated using preoperative abdominal-enhanced CT or EUS. The most common intraoperative complication was severe intraoperative bleeding, defined as repeated endoscopic hemostasis with a postoperative decrease in circulating hemoglobin concentration of >2 g/dL. 13 The postoperative complications encountered included delayed hemorrhage, delayed perforation, and postoperative infection. A diagnosis of delayed hemorrhage was confirmed by emergency endoscopy, and a diagnosis of delayed perforation was confirmed by X-ray or CT. Postoperative infection was defined as a postoperative body temperature >37.5°C, accompanied by indicators of inflammation on laboratory testing (routine blood data, high C-reactive protein concentration, or high calcitonin concentration. 14 An R0 resection was defined on the basis of histological examination of the lateral and vertical margins, which showed no residual tumor. 15
Postoperative management
The tissue removed during surgery was fixed in a 10% formalin solution and subjected to immunohistochemical staining for markers including CD117, CD34, and Dog-1, to confirm the diagnosis. Following surgery, the participants typically underwent nasogastric decompression to prevent postoperative complications and were instructed to fast for 2 days. For those who underwent EFTR, the fasting period was extended to 3 days or more, according to their postoperative condition. Post-surgical routine blood measurements, and the measurement of circulating CRP and/or calcitonin concentration was performed, and all the participants were administered a proton pump inhibitor, gastric mucosal protective agents, nutritional support, and fluid replacement. When a participant experienced abdominal pain or muscle tension, a CT or orthostatic X-ray scan was performed to rule out postoperative perforation. Antibiotic therapy was provided or further surgery was performed based on the condition of the participant. When participants experienced intraoperative perforation or postoperative infection, antibiotics were prescribed.
Follow-up
To evaluate wound healing, the participants underwent endoscopic examinations 1, 3, 6, and 12 months after their procedure, and recurrence or residual tumor was identified using endoscopic ultrasonography. Subsequently, re-examinations were conducted annually, when an abdominal contrast-enhanced CT scan and a chest CT scan were performed to identify lymph node and distant metastases.
Learning curve analysis
Cumulative sum (CUSUM) analysis has previously been used to quantitatively evaluate the LC for a number of endoscopic procedures.16,17 In the present study, we evaluated the relationship between the experience of the endoscopist and the duration of surgery, and the CUSUM method was used to analyze the LC for the ER of gGISTs. The formula used to calculate CUSUM is as shown below, in which Xi represents the duration of surgery for each participant, u represents the mean duration of surgery for all the participants, and n represents the participant’s serial number.
Statistical analysis
Categorical datasets are expressed as frequencies and percentages and were compared using the chi-square or Fisher’s exact tests. Normally distributed continuous datasets are expressed as mean ± standard deviation and were compared using Student’s t-test. Non-normally distributed continuous datasets are expressed as median and interquartile range (IQR) and were compared using the Mann–Whitney U-test. Univariate analysis was performed for the Conversion and Non-conversion (to LR) groups, and variables with P < 0.1 on univariate analysis were included in a multivariate regression analysis. p < 0.05 was considered to represent statistical significance, and all analyses were performed using SPSS version 26 (IBM Corp., Armonk, NY, USA).
Results
Patient demographics
During the study period, 297 patients with 300 stomach lesions underwent ER. We excluded five patients who had simultaneous lesions at other sites and three who had multiple lesions in their stomach. Following this, data from 289 participants with 289 lesions remained for analysis. Table 1 shows the demographics of the participants who underwent ER for the treatment of gGISTs. There were 149 men (51.6%) and 140 women (48.4%), with a median age of 59.8 ± 9.4 years. The median (IQR) tumor diameter was 1.5 (1.0, 2.0) cm, and the diameter ranged from 0.3 to 5.0 cm. Most of the lesions were within the MP (87.5%). The demographics of the participants during the two phases were similar (Table 1).
Demographics of the participants during the two phases.

SD: standard deviation; IQR: interquartile range; BMI: body mass index; MP: muscularis propria; NIH: National Institutes of Health; ASA: American Society of Anesthesiologists; N/A: not available. Data were analyzed using #Student’s t-test; $the Mann–Whitney U-test; &the chi-square test, or *Fisher’s exact test.
Procedures and outcomes for ER of gGISTs
The median (IQR) duration of surgery was 59.0 (47.5, 75.0) minutes, and conversion to LR was required in nine patients (3.1%). The intraoperative and postoperative complication rates were 2.4% and 9.0%, respectively. A total of 273 participants (94.5%) underwent R0 resection. The median (IQR) durations of postoperative fasting and hospitalization were 3.0 (2.0, 3.0) and 5.0 (5.0, 6.0) days, respectively. Twenty-six participants experienced postoperative complications, comprising two cases of postoperative hemorrhage, 25 cases of postoperative infection, and one case of postoperative perforation. The two participants who experienced postoperative hemorrhage were successfully treated by emergency endoscopic hot biopsy forceps hemostasis. Of the 25 participants who experienced postoperative infection, 14 presented with fever but no increases in inflammatory marker concentrations and were not administered antibiotics, and 11 had high inflammatory marker concentrations and fever, but recovered after receiving antibiotic treatment. The participant who experienced postoperative perforation was successfully treated by supportive nutritional therapy and nasogastric decompression. During the follow-up period, no lymph node or distant metastasis was identified.
Follow-up analysis
A total of 242 participants were successfully followed up, with a median duration of follow-up of 45.0 months (IQR 18.0 to 82.0 months). Seventeen of the participants were classified as intermediate- or high-risk, according to the modified National Institutes of Health (NIH) criteria, 18 all of whom underwent postoperative treatment with imatinib. Only one participant, who had an intermediate risk score following surgery, experienced recurrence. This patient discontinued imatinib treatment after 6 months but underwent laparoscopic surgery upon identification of the recurrence. No further recurrence was identified during the subsequent follow-up period.
Learning curve analysis
The maximum CUSUM duration of surgery occurred at approximately the 50th case. After this, the CUSUM decreased, except when six participants required conversion to LR (Figure 2). Based on the CUSUM chart, the participants were divided into early (cases 1 to 50) and later (cases 51 to 289) phases of the LC. The durations of surgery, postoperative hospitalization, and postoperative fasting, and the incidences of intraoperative and postoperative complications, during the early phase were significantly lower than those during the later phase (P < 0.05) (Table 2).

Cumulative sum (CUSUM) of the duration of surgery plotted against case number.
Intraoperative and postoperative data for the two phases.

ESD: endoscopic submucosal dissection; EFTR: endoscopic full-thickness resection; Others: over-the-scope-clips or surgical sutures. $Mann–Whitney U-test, &chi-square test, *Fisher’s exact test.
Risk factors for conversion to laparoscopic resection
The prevalences of conversion to LR during the early and later phases were 6.0% and 2.5%, respectively (P > 0.05) (Table 2). The reasons for conversion to LR were as follows: severe intraoperative hemorrhage that could not be controlled endoscopically (n = 6), failure to close the perforation (n = 2), and the tumor falling into the abdominal cavity (n = 1). Multivariate analysis revealed that the risk factors associated with conversion to LR were tumor size (≥3.0-cm diameter, odds ratio [OR] 17.92, 95% confidence interval [CI] 2.65, 120.87, P = 0.003) and invasion beyond the MP (OR 58.03, 95% CI 6.40, 525.84, P < 0.001) (Table 3).
Risk factors for the conversion to laparoscopic resection.
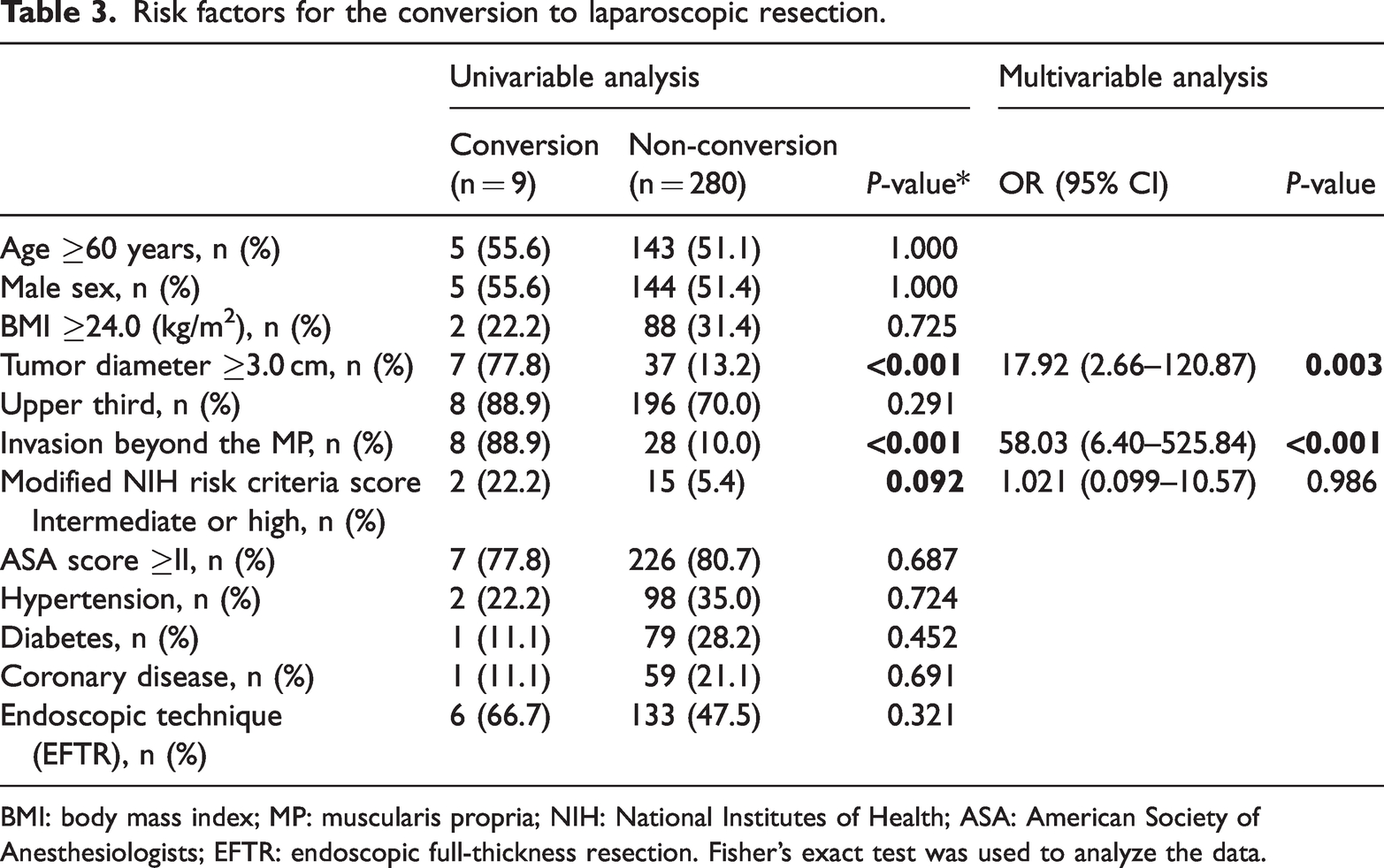
BMI: body mass index; MP: muscularis propria; NIH: National Institutes of Health; ASA: American Society of Anesthesiologists; EFTR: endoscopic full-thickness resection. Fisher’s exact test was used to analyze the data.
Discussion
Although ER for gGIST had previously been performed by a small number of specialists in high-volume centers, the technique has recently been introduced to many less-busy hospitals. Owing to the difficulty of the technique and the risk of intraoperative complications, it may be important to assign cases according to the characteristics of the surgical procedure and the skill level of each endoscopist to ensure the safety of the procedure. Therefore, in the present study, we aimed to objectively evaluate the technical ability of an endoscopist by studying the LC for the ER of gGISTs.
In our previous study, we evaluated the LC for the ER of gGISTs in a cohort of 100 patients, and found that the treatment of approximately 30 patients was sufficient for an endoscopist to acquire the skills necessary to minimize the risks of complications and conversion associated with the ER procedure. We estimated that the treatment of 60 patients would be necessary to complete the LC for the ER of gGISTs. 19 However, it is important to note that this study was of a relatively small number of patients with tumor diameters >3.0 cm, which may have been relatively simple to treat surgically. In addition, the small sample size might have influenced the accuracy of the results. To address these limitations, in the present study, we included 289 patients, of whom only 14.9% (43/289) had tumors >3.0 cm in diameter (Supplementary Table 1). The findings indicate that approximately 50 cases must be treated by an endoscopist before they reach the appropriate level of skill required to successfully perform the ER of gGISTs.
Sun et al. 8 reported that approximately 32 patients should be treated for endoscopists who are competent in basic endoscopic intervention skills to acquire the skills necessary for the successful ER of gastric SMTs. However, this was a small study, and therefore there was a high risk of bias. In addition, gastric SMTs include many types of lesions, including gGISTs, leiomyomas, ectopic pancreas, schwannomas, hemangiomas, and inflammatory fiber polyps.2,3,20 SMTs differ in the sites at which they develop, their shapes, their growth types, and their boundaries with surrounding tissues. Therefore, the difficulty of the ER of the various SMTs differs, and therefore the LC for the ER of specific SMTs differs. Numerous previous studies have consistently shown that the LC for the ER of gastric lesions is approximately 30 cases,9,21–23 which is shorter than the LC calculated in the present study for the ER of gGISTs. We believe that the longer LC for the ER of gGISTs than for the other gastric lesions may be attributable to the following factors. (1) GISTs are located deeper within the gastric wall than other gastric lesions, and closer to the muscular or serosal layer. This necessitates deeper incisions and adds to the complexity of GIST resection. (2) GISTs can exhibit irregular shapes and may be associated with adhesions to surrounding tissues, whereas other SMTs, such as leiomyomas, tend to have more regular shapes. This more complex morphology of GISTs poses challenges during the resection process. (3) GISTs have a rich blood supply, predisposing them toward intraoperative bleeding. In comparison, other gastric lesions, such as leiomyomas, are less likely to hemorrhage. Consequently, a meticulous approach is required to control hemorrhage and ensure surgical safety during the resection of GISTs.
The median duration of surgery was significantly shorter during the later phase than the early phase (55.0 vs. 79.0 min, P < 0.001), principally because of an improvement in the proficiency of the endoscopist. With the increase in the number of procedures being performed, the technique with which intraoperative hemorrhage was dealt with improved, which reduced the duration of surgery. Furthermore, the accumulation of experience led to a decrease in the incidence of postoperative complications, and the durations of postoperative fasting and hospitalization were also shorter during the later phase.
During the present study, nine participants had their procedures converted to LR. Because this is associated with a longer duration of surgery, we also aimed to identify the risk factors for this conversion. To the best of our knowledge, the difficulty associated with performing the ER of gastric lesions is largely related to the tumor size, location, and depth of invasion.24–27 Therefore, we chose to interrogate these variables as potential risk factors, and found that tumor diameter >3.0 cm and invasion beyond the MP were associated with conversion. We also found that participants with larger gGISTs were more likely to experience severe intraoperative hemorrhage than those with tumors <3 cm (14.0% vs. 0.4%, P < 0.001), as were those with gGISTs showing invasion beyond the MP (1.6% vs. 8.3%, P = 0.044) (Supplementary Table 1). When severe intraoperative hemorrhage could not be endoscopically controlled, conversion to LR was required.
The present study had some limitations. First, it was a single-center retrospective study, and the LC of a single endoscopist was studied. Therefore, a prospective multi-center study involving multiple endoscopists should be performed to validate our results. Second, the number of participants that required conversion was low (n = 9; 3.1%), which may have affected the accuracy of the results. However, previous studies have shown that the conversion rate for the ER of gGISTs is low.5,13 With improvements in the technical proficiency of the endoscopist, the incidence of conversion gradually decreased. Therefore, an increase in sample size may not necessarily increase the number of patients requiring conversion. Third, given the differences in the procedures, it is likely that the difficulty of and the LCs associated with ESD and EFTR differ. In the present study, we studied the LC for both ER techniques together. However, it will be important to conduct further research regarding the LCs specific to each endoscopic technique in the future.
In conclusion, we have shown that the LC for the ER of gGISTs lasts for approximately 50 cases, and tumors >3.0 cm in diameter and those that invade beyond the MP are associated with higher risk of conversion to LR. Thus, novice endoscopists should aim to accumulate surgical experience and select cases suitable for their technical proficiency before embarking on surgical procedures.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231194448 - Supplemental material for Assessment of a Real-world Learning Curve for the Endoscopic Resection of Gastric Gastrointestinal Stromal Tumors
Supplemental material, sj-pdf-1-imr-10.1177_03000605231194448 for Assessment of a Real-world Learning Curve for the Endoscopic Resection of Gastric Gastrointestinal Stromal Tumors by Ying Sun, Luojie Liu, Dongtao Shi, Chao Ma and Xiaodan Xu in Journal of International Medical Research
Footnotes
Author Contributions
Ying Sun and Luojie Liu collected the data. Ying Sun interpreted the data. Luojie Liu drafted the manuscript. Dongtao Shi and Chao Ma revised the manuscript. Xiaodan Xu conceived and designed the study.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: a grant from the Health Personnel Training Project of Soochow (number GSWS2020109) and a grant from the Changshu Science and Technology Project (number CS202116).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

