Abstract
Objective
This study was performed to examine the disease course in geriatric patients with soft tissue sarcoma and determine the risk factors for mortality.
Methods
We retrospectively analyzed patients who were treated at Istanbul University Oncology Institute from January 2000 to August 2021.
Results
Eighty patients were included in the study. The patients’ median age was 69 years (range, 65–88 years). The median overall survival of patients diagnosed between the ages of 65 and 74 years was 70 months, and that of patients diagnosed at the age of ≤75 years was significantly lower at 46 months. The median survival of patients who did and did not undergo surgical resection was 66 and 11 months, respectively, with a significant difference. The median overall survival of patients with positive and negative surgical margins was 58 and 96 months, respectively, also with a significant difference. Age at diagnosis and recurrence/metastasis significantly affected mortality. A 1-year increase in the age at diagnosis increased mortality by 1.147 times.
Conclusion
Age of >75 years, inability to undergo surgery, positive surgical margins, and head and neck location may be associated with a poor prognosis in geriatric patients with soft tissue sarcoma.
Introduction
Soft tissue sarcomas are a heterogeneous group of diseases with more than 50 subtypes, 1 but they constitute <1% of all malignancies in adults. 2 The main treatment is surgery, and both preoperative and postoperative radiotherapy and chemotherapy are frequently performed. Although doxorubicin-based chemotherapy is the main treatment for metastatic disease, multikinase inhibitors such as pazopanib are also routinely administered. The incidence of cancer increases with age. According to the National Cancer Institute Surveillance Epidemiology and End Program data, 54% of malignancies and 70% of malignancy-related deaths occur in patients aged >65 years. 3 In one study, 40% of soft tissue sarcomas were observed in patients aged >65 years. 4 Cancer treatment in geriatric patients is difficult because such patients are often fragile, comorbid diseases are common, and chemotherapy tolerance decreases with age. The present study was performed to examine the disease course in geriatric patients with soft tissue sarcoma and determine the risk factors for mortality. We examined geriatric patients who were treated for soft tissue sarcoma at Istanbul University Oncology Institute from January 2000 to December 2020.
Methods
Study design and patient characteristics
This retrospective study was performed at the Department of Oncology, Istanbul University Institute of Oncology. The inclusion criteria were an age of ≥65 years, diagnosis of soft tissue sarcoma, and follow-up or treatment for soft tissue sarcoma. Patients aged <65 years, patients with bone and cartilage tumors, and patients who had discontinued follow-up were not included in the study. The files of patients from January 2000 to December 2020 were examined, and patients who were deemed suitable for the study were included.
Examined clinical parameters
The patients’ age at the time of diagnosis, comorbid diseases, sex, histological subgroups, tumor location, and disease stage according to the American Joint Committee of Cancer 2017 classification were examined. We also examined whether the patients received neoadjuvant chemotherapy, neoadjuvant radiotherapy, adjuvant chemotherapy, or postoperative radiotherapy; whether local recurrence or distant metastasis developed; the final status; and overall survival. The patients’ privacy was respected, and information regarding their identity was not included in the study.
Statistical analysis
Statistical analyses were performed using the R package program (www.r-project.org). Descriptive statistics of quantitative variables included median, minimum, and maximum values. Qualitative variables are presented as frequency and percentage. The factors affecting disease-free survival and overall survival were analyzed using Kaplan–Meier analysis and the log-rank test. For all statistical analyses, results with a p value of <0.05 were considered statistically significant.
Results
Eighty patients were included in the study. The median follow-up period was 78 months (range, 65.1–90.8 months). Thirty-three (41.25%) patients were female, and 47 (58.75%) patients were male. The patients’ median age was 69 years (range, 65–88 years). Fifty-eight (72.50%) patients were 65 to 74 years old, and 22 (27.50%) patients were ≥75 years. Thirty-nine (48.75%) patients had comorbid disease. Forty-one patients (51.25%) had no comorbid disease. The most common comorbid diseases were hypertension, diabetes mellitus, and atherosclerotic heart disease. In addition to soft tissue sarcoma, four patients had other simultaneous malignancies (bladder cancer, lung cancer, breast cancer, and brain tumor in one patient each). The disease location was the lower extremities in 26 (32.50%) patients, intra-abdominal region in 18 (22.50%), upper extremities in 16 (20.00%), head and neck in 7 (8.75%), lung in 4 (5.00%), chest wall in 3 (3.75%), skin in 3 (3.75%), testis in 2 (2.50%), and back in 1 (1.25%). With respect to histological subgroups, 38 (47.50%) patients had fusiform cell sarcoma (no subtyping), 17 (21.25%) had liposarcoma, 7 (8.75%) had myxofibrosarcoma, 5 (6.25%) had malignant peripheral nerve sheath tumor, 5 (6.25%) had leiomyosarcoma, 3 (3.75%) had angiosarcoma, 2 (2.50%) had epithelioid sarcoma, 1 (1.25%) had fibrosarcoma, 1 (1.25%) had pleomorphic undifferentiated sarcoma, and 1 (1.25%) had schwannoma. With respect to the tumor grade [using the Fédération Nationale des Centres de Lutte Contre Le Cancer (FNCLCC) grading system], no grading was performed in 6 (7.50%) patients, 29 (36.25%) had grade 1 tumors, and 45 (56.25%) had grade 2–3 tumors. With respect to disease stage, 10 (12.50%) patients had Stage 1A, 21 (26.25%) had Stage 1B, 8 (10.00%) had Stage 2, 16 (20.00%) had Stage 3A, 16 (20.00%) had Stage 3B, and 9 (11.25%) had Stage 4. Sixty-nine (86.25%) patients underwent operations and 11 (13.75%) did not. The surgical margin was positive in 14 patients and negative in 29 patients. Information regarding the surgical margin could not be obtained for 26 patients. Adjuvant chemotherapy was applied in 10 patients, and adjuvant radiotherapy was applied in 19 patients. Disease relapse occurred in 25 patients. Distant metastasis occurred in 9 patients and local recurrence in 16 patients. Four of the distant metastases were in the lung. Curative resection was performed in 13 of 16 patients with local recurrence. Eleven patients received metastatic regimen chemotherapy. Four patients received adriamycin and ifosfamide, two patients received single-agent ifosfamide, one patient received single-agent doxorubicin, one patient received temozolomide, and two patients received pazopanib (Table 1).
Patient characteristics.

The median overall survival was 58 months (range, 2–132 months) (Figure 1). The median overall survival in women and men was 61 and` 54 months, respectively, with no significant difference. The median overall survival of patients without and with comorbid disease was 61 months (range, 44.2–77.8 months) and 53 months (range, 23.6–82.3 months), respectively, with no significant difference (Figure 2). Eighteen (46.2%) patients with comorbid disease and 21 (53.8%) without comorbid disease died, also with no significant difference. A survival analysis was performed according to the patients’ age at the time of diagnosis. The median overall survival of 58 patients diagnosed between the ages of 65 and 74 years was 70 months, and that of 22 patients diagnosed at the age of ≥75 years was significantly lower at 46 months (p = 0.030) (Figure 3). The median survival times were evaluated based on tumor localization. The median survival of patients with lower extremity localization was 91 months, that of patients with upper extremity localization was 66 months, that of patients with head and neck localization was 21 months, that of patients with intra-abdominal localization was 43 months, and that of patients with localization in other parts of the body was 96 months (p = 0.019) (Figure 4). A survival analysis was also performed according to the histological subgroup. The median survival was 58 months in patients with fusiform cell sarcoma (not subtyped) histology, 53 months in patients with liposarcoma histology, and 53 months in patients with other histology, with no significant difference. A survival analysis was also performed based on tumor grading. The median overall survival was 96 months in patients with grade 1 tumors and 61 months in patients with grade 2 and 3 tumors, also with no significant difference. Moreover, a survival analysis was performed according to whether surgical resection was performed. The median survival was 66 months in 69 patients who underwent surgical resection and 11 months in 11 patients who did not (p < 0.001) (Figure 5). A survival analysis was also performed according to surgical margins. The median survival was 54 months in patients without surgical margin information, 58 months in patients with positive surgical margins, and 96 months in patients with negative margins, with no significant difference (Figure 6). The median survival of patients who did and did not receive adjuvant chemotherapy was 43 and 74 months, respectively, with no significant difference. Furthermore, a survival analysis was performed according to the tumor stage. The median survival of patients with stage 1 and 2 tumors was 66 months, and that of patients with stage 3 and 4 tumors was 44 months; the difference was not statistically significant (Figure 7). The median survival of patients who did and did not receive adjuvant radiotherapy was 66 months, with no significant difference (Table 2).
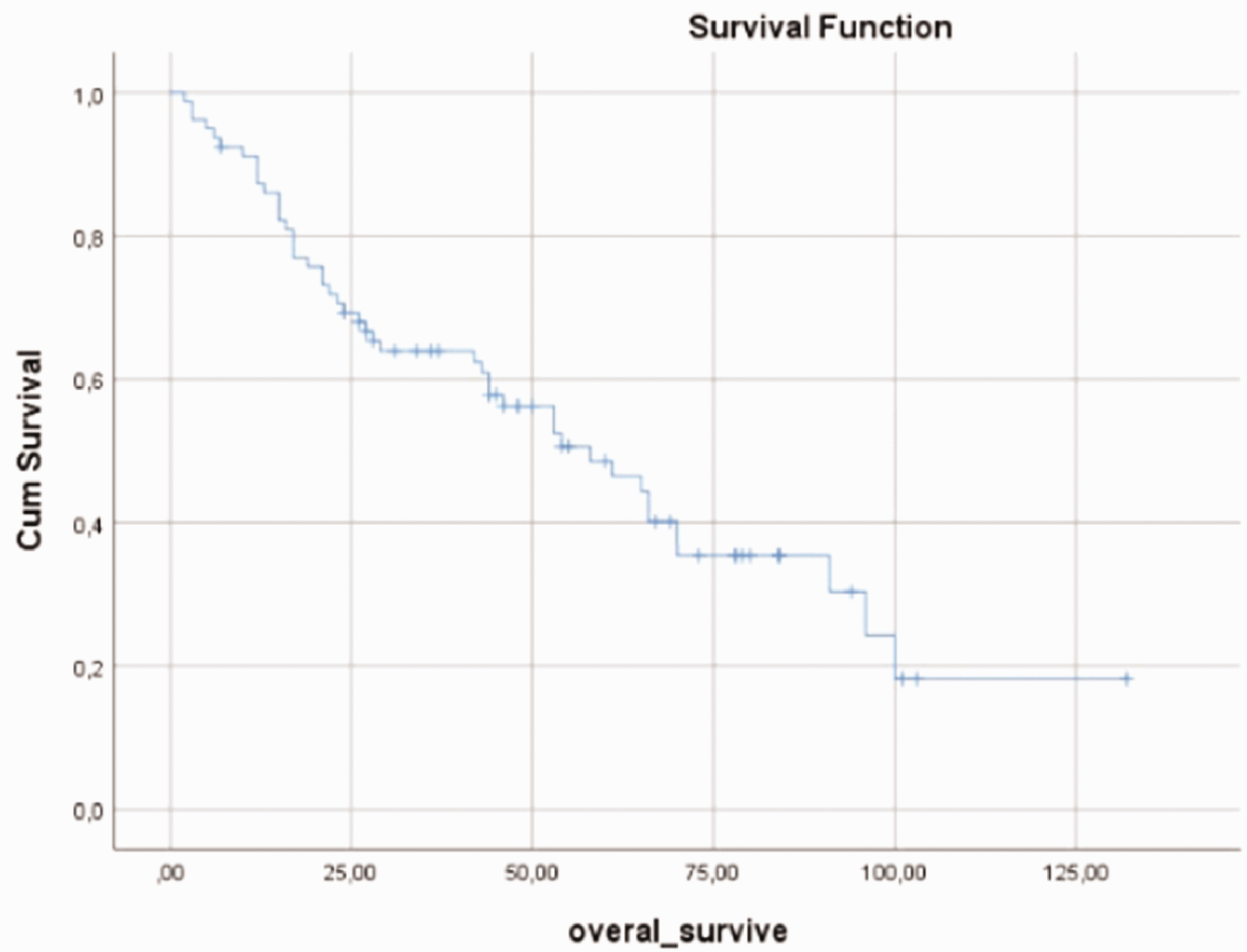
Graph of overall survival of all patients.
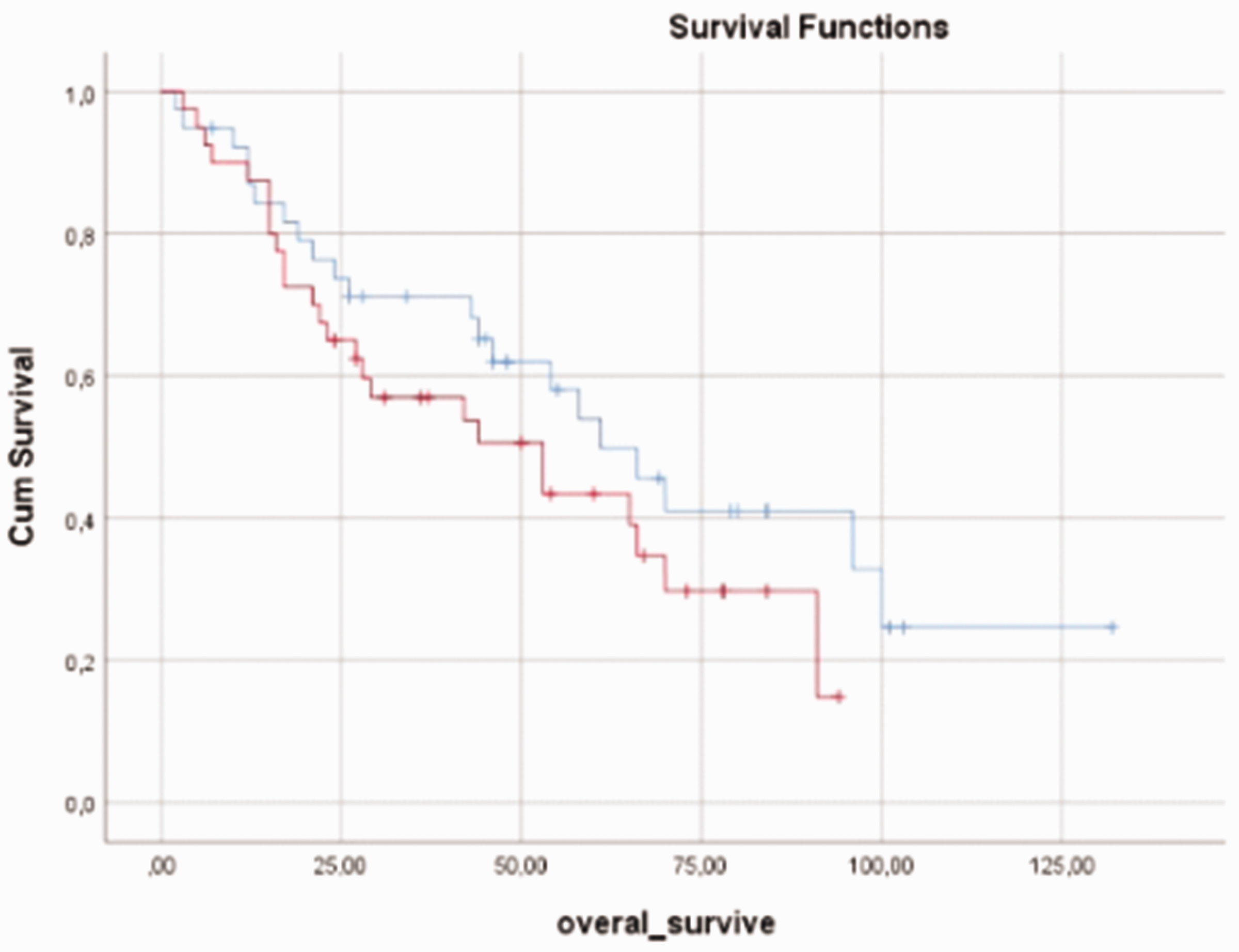
Graph of overall survival according to comorbid disease. Blue, patients with comorbid disease; red, patients without comorbid disease.

Graph of overall survival according to age. Blue, patients aged <75 years; red, patients aged ≥75 years.

Graph of overall survival according to tumor localization. Blue, lower extremity; red, upper extremity; green, head and neck; pink, intra-abdominal; yellow, other localizations.
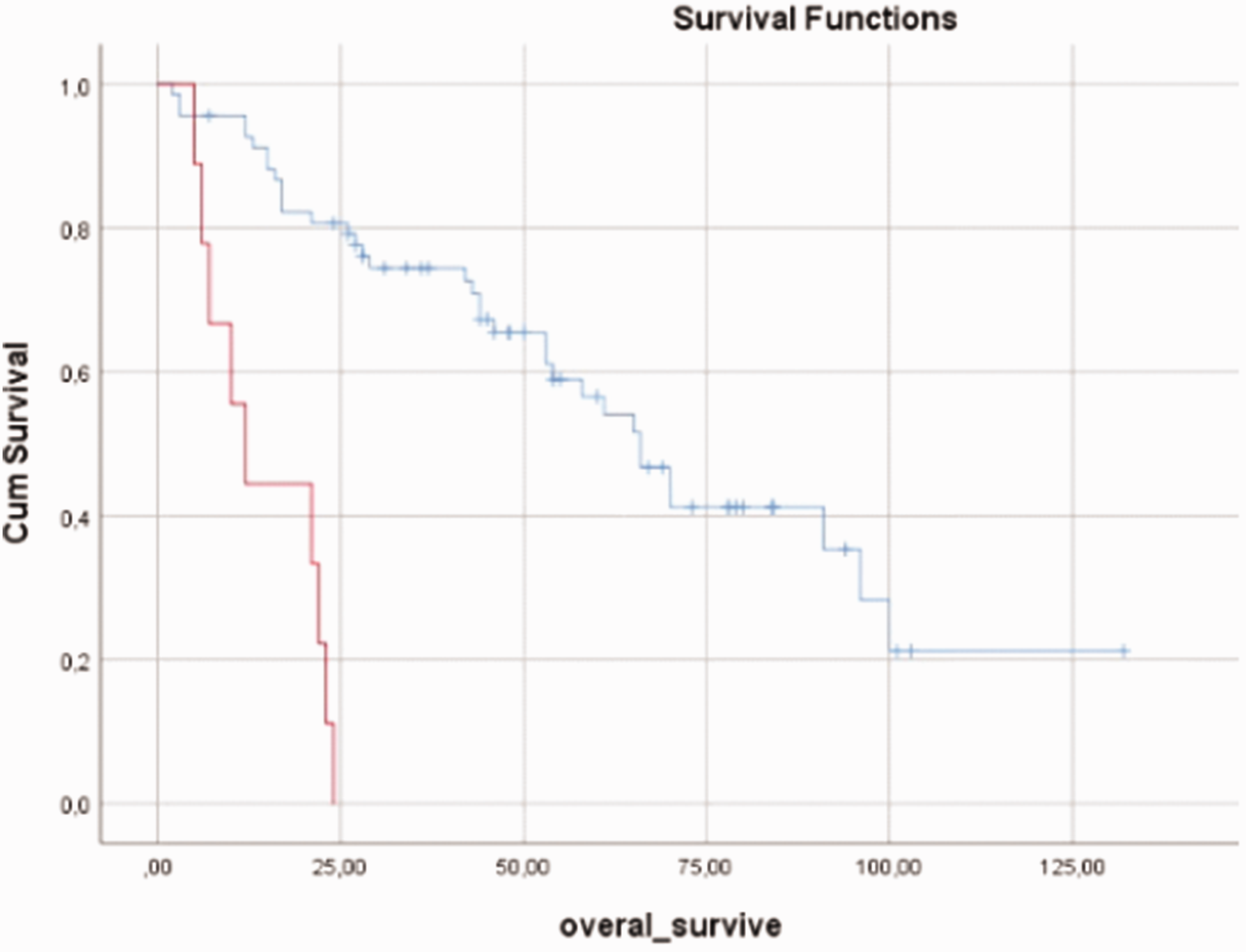
Graph of overall survival according to surgical resection. Blue, operated; red, not operated.
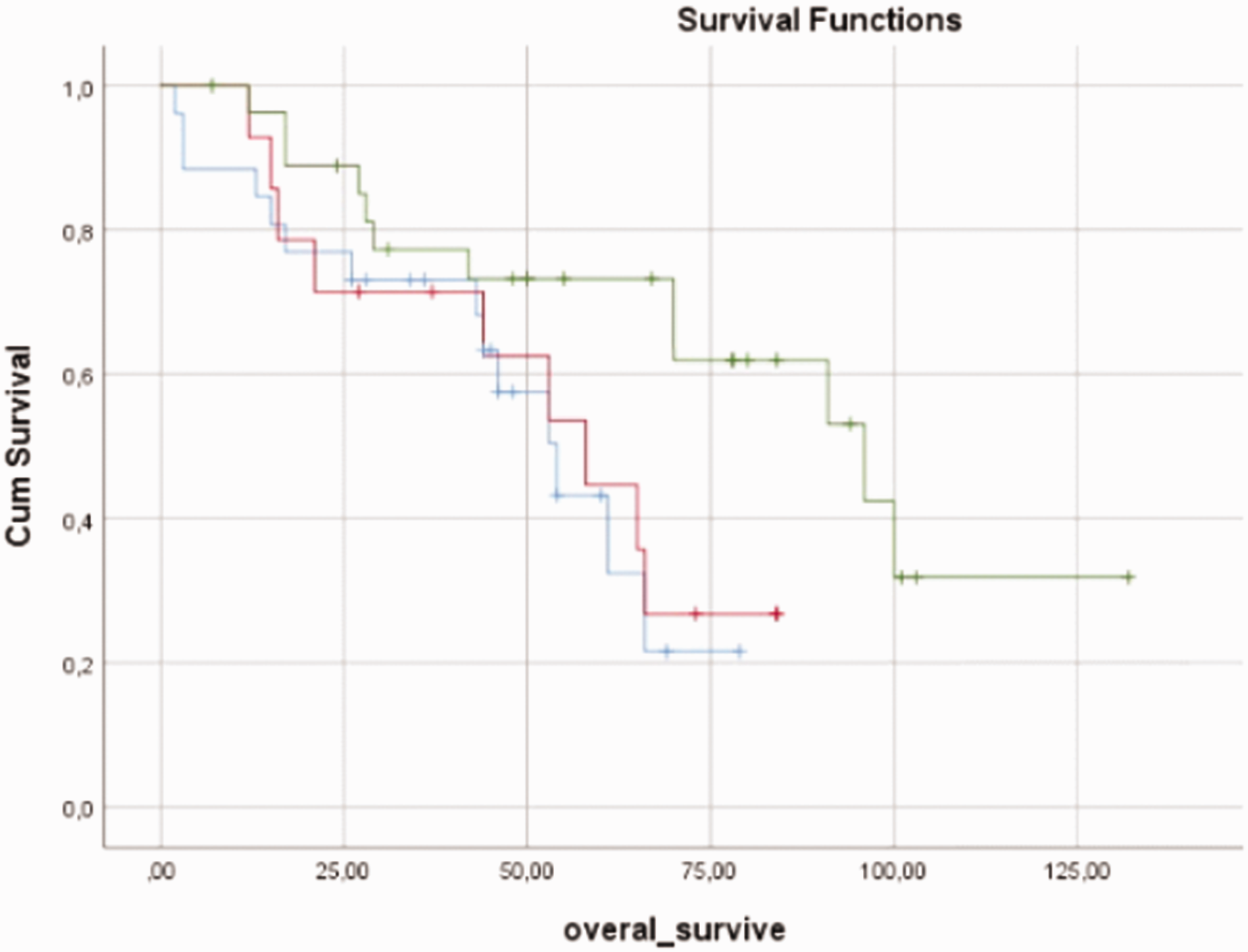
Graph of overall survival according to surgical margin. Blue, unknown; red, positive; green, negative.

Graph of overall survival according to stage. Blue, stages 1–2; red, stages 3–4.
Overall survival according to various patient characteristics.

OS, overall survival; CI, confidence interval.
Factors affecting mortality were also examined. Age at diagnosis and development of recurrence/metastasis significantly affected mortality (p < 0.001 and p = 0.018, respectively). A 1-year increase in the age at diagnosis increased mortality by 1.147 times. The risk of mortality in patients with recurrence/metastasis was 3.3 times higher than that in patients without recurrence/metastasis (Table 3).
Factors affecting mortality.

SE, standard error; df, degrees of freedom; CI, confidence interval.
The final status of the patients was examined. Forty-six (57.5%) patients died and 34 (42.5%) survived. The 1-, 2-, 3-, and 5-year survival rates were 87.2%, 67.9%, 55.1%, and 24.4%, respectively.
Discussion
Soft tissue sarcomas have a poor prognosis, a relapse rate of approximately 50%, and a survival duration of 12 to 18 months in the metastatic stage. 6 In one study, 54% of malignancies were observed in geriatric patients. 3 Comorbid diseases are common in geriatric patients, and geriatric patients are more fragile than younger patients. Therefore, the desired treatment is not always able to be administered, and treatment complications are common. One study involving a geriatric assessment showed no findings of concern in the routine physical examination or anamnesis, but 36.6% of the patients had fatigue and 37.6% had difficulty eating. 7 Polypharmacy is also common in geriatric patients. In a study by Sharma et al., 8 polypharmacy was detected in 84% of geriatric patients with cancer. Because of their fragile nature and frequent drug interactions, geriatric patients with cancer require great care in the planning of their treatment approach.
In the present study, the patients’ median age was 69 years (range, 65–88 years). Fifty-eight (72.5%) patients were 65 to 74 years of age, and 22 (27.5%) patients were ≥75 years of age. The median overall survival of the 58 patients diagnosed between the ages of 65 and 74 years was 70 months, and the median survival of the 22 patients diagnosed at the age of ≥75 years was 46 months, with no significant difference. Advanced age is a poor prognostic factor. 9 Interestingly, in a study of 202 geriatric patients with sarcoma in Japan, 10 there was no statistically significant difference in 5-year survival between patients aged <75 and ≥75 years. In the present study, the median overall survival significantly differed between patients aged <75 and >75 years. In this respect, there is a difference between our study and the above-mentioned Japanese study. This may have occurred because most patients in the Japanese study had poor prognostic factors.
Soft tissue sarcomas originate from the lower extremities in 28% of cases, the upper extremities in 12%, the retroperitoneum in 16%, 11 and the head and neck in 5% to 15%. 12 In our study, soft tissue sarcomas originating from the extremities and abdomen were more common. Our patient group differs from the literature in this respect. A survival analysis was performed by tumor location, and patients with head and neck soft tissue sarcoma showed significantly lower median survival than patients with other localizations (p = 0.019). This is an important result of our study. In a study of 11,481 patients in which SEER data were analyzed, 13 head and neck localization did not cause a worse prognosis, especially in patients with malignant fibrous histiocytoma histology.
Approximately half of the patients in this study had comorbid disease. The median overall survival of those without comorbid disease was better than that of those with comorbid disease, although the difference was not statistically significant. In a recent study conducted in 2021, comorbid disease was found in 45% of 563 geriatric patients with cancer, but the presence of comorbidity did not pose an additional risk for mortality. 14 The lack of a soft tissue sarcoma subgroup in this study can be considered a limitation; in general, however, our study yielded results consistent with the literature in this respect.
The histological subgroup of the tumor was also examined. Thirty-seven (46.25%) patients had fusiform cell sarcoma (no subtyping), 17 (21.25%) had liposarcoma, and 7 (8.75%) had myxofibrosarcoma; however, 12% of soft tissue sarcomas were in the histological subgroup of liposarcoma, 12% in leiomyosarcoma, and 11% in undifferentiated pleomorphic sarcoma. 15 A limitation of our study is that the histological subgroup could not be determined in approximately half of the patients. A survival analysis was also performed according to histological subgroups. There was no statistically significant difference in survival between patients with fusiform cell sarcoma (not subtyped), liposarcoma, and other histological subgroups. Twenty-nine (36.25%) patients had grade 1 tumors and 45 (56.25%) had grade 2 and 3 tumors. No grading was performed in six patients. The median overall survival in patients with grade 1 tumors was 96 months and that in patients with grade 2 and 3 tumors was 61 months, with no significant difference. The lack of statistical significance despite the 35-month difference may be related to the low number of patients included in the study. High-grade tumors are known to have a poor prognosis.16,17
Sixty-nine (86.25%) patients in the present study underwent operations. The surgical margin was positive in 14 patients and negative in 29 patients. The surgical margin information could not be obtained for 26 patients. The median survival of the 69 patients who underwent surgical resection was 66 months, and that of 11 patients who did not was 11 months (p < 0.001). The median survival of patients without surgical margin information was 54 months, that of patients with positive surgical margins was 58 months, and that of patients with negative surgical margins was 96 months (p = 0.044). The main treatment for soft tissue sarcoma is surgery. Many studies have shown that surgical margin positivity causes more local recurrence. 18 We thus expected to observe a significantly better survival rate in patients who underwent R0 resection. A meta-analysis of 33 studies showed that surgical margin negativity was associated with a good prognosis. 19 Consistent with the literature, surgical margin negativity was found to be associated with a good prognosis in the geriatric population of the present study.
Twenty-five (31.5%) patients developed disease relapse. Eighteen (22.5%) patients had metastasis (nine at the first visit and nine at follow-up). In a previous study of patients with soft tissue sarcomas, the local recurrence rate was 20% to 30% and the distant metastasis rate was 30% to 50%. 20 In our study, fewer patients developed distant metastasis than reported in the literature. This is a remarkable result. Only 11 (61.1%) of these 18 patients were able to receive metastatic regimen chemotherapy, and 4 patients were able to receive adriamycin and ifosfamide. The low number of patients receiving chemotherapy may be explained by the fact that the patients were fragile and not in condition to receive chemotherapy.
The median survival of patients with stage 1 and 2 tumors was 66 months, and the median survival of patients with stage 3 and 4 tumors was 44 months; the difference was not statistically significant. Patients with advanced stages of disease are expected to have worse disease courses. The lack of statistical significance in survival may have been due to the small number of patients.
The most important finding of our study is that the age at diagnosis and the development of recurrence/metastasis affected mortality. A 1-year increase in the age at diagnosis increased mortality by 1.147 times. The mortality risk was 3.3 times higher in patients with than without recurrence/metastasis.
Our study had some limitations due to its retrospective nature: tumor grading was not performed in some patients, special evaluation according to the patients’ geriatric status was not performed, and tests that would specifically show fragility in geriatric patients were not performed.
The world population is becoming older. Clinicians are certain to see more geriatric patients with cancer in the near future. According to the National Cancer Institute data, 25% of patients included in clinical trials are aged 65 to 74 years, and 10% are aged >75 years. 21 The number of geriatric patients enrolled in clinical trials needs to increase. Studies with higher numbers of patients would serve as a guide for the treatment of geriatric patients with cancer.
Conclusion
Forty percent of soft tissue sarcomas occur in the geriatric population. Age of >75 years, inability to undergo surgery, positive surgical margins, and head and neck location may be associated with a poor prognosis. A 1-year increase in the age at diagnosis increases mortality by 1.147 times, and recurrence or metastasis increases mortality by 3.3 times. Studies involving more patients are needed.
Research Data
Research Data for Clinical course and features of soft tissue sarcomas in geriatric patients: a single-center experience
Research Data for Clinical course and features of soft tissue sarcomas in geriatric patients: a single-center experience by Metin Pehlivan, Ayça İribaş, Bilge Bilgiç, Mert Başaran and Meltem Ekenel in Journal of International Medical Research
Footnotes
Acknowledgements
The authors thank all who participated in and contributed to the study.
Author contributions
Metin Pehlivan: Study design, data collection, data interpretation, writing
Ayca Iribas Celik: Data interpretation
Bilge Bilgic: Data interpretation
Mert Basaran: Study design, data interpretation
Meltem Ekenel: Study design, data interpretation
Data availability statement
The data of the study can be shared upon request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethical approval
The study was approved by the academic board at Istanbul University Oncology Institute and the Istanbul Medical Faculty Clinical Research Ethics Committee (decision number 364259). The study was conducted in accordance with the latest version of the World Medical Association Declaration of Helsinki and the newly published Good Clinical Practices Guide/Good Laboratory Practices Guide of the Ministry of Health. The reporting of this study conforms to the STROBE guidelines. 5 Informed consent was not required because of the retrospective nature of the study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
