Abstract
Objective
To perform a single-centre, detailed analysis of the preparations for the introduction of the first pressurized intraperitoneal aerosol chemotherapy (PIPAC) programme in the eastern part of Central Europe.
Methods
The study analysed the 14-month preparation period prior to the performance of the first PIPAC procedure with respect to: (i) general preparations; (ii) patient referral and qualification; (iii) the first PIPAC procedure; (iv) the 2 weeks following PIPAC programme establishment; and (v) general problematic issues that arose.
Results
The length of time needed to prepare our institution for the first PIPAC procedure was extremely long compared with other European Union PIPAC centres: 14 months versus a standard 3–6 months of preparation. The longest amount of time (12 months) was required to prepare the required paperwork.
Conclusions
A new PIPAC programme was successfully established in the eastern part of Central Europe. The length of time to implement this method was significantly longer because of lengthy bureaucratic processes. These current findings should help new centres, especially in this part of Europe, to establish a PIPAC programme more quickly.
Introduction
Peritoneal carcinomatosis (PC) is a devastating diagnosis characterized by a rapidly progressing metastatic process derived from different types of primary neoplasms involving the peritoneal cavity, which is commonly classified and treated as stage IV.1–3 The most common primary tumours that metastasize to the peritoneal cavity include ovarian and digestive-tract cancers.4,5 For many years, gastroenterologists and surgical oncologists considered PC to be a considerable challenge. As PC was considered to be the terminal stage of the disease, patients were referred for palliative treatment with only a modest portfolio of therapeutic options.2,6,7 This situation had arisen because patients with PC and a gastrointestinal primary tumour have a poor prognosis with a relatively short median survival time compared with other types of metastasis. 8 Notably, the median time of survival for patients receiving only supportive care and standard palliative treatment has been estimated as 1–3 months for patients suffering from PC of gastric cancer origin, 16–17 months for patients with PC of colorectal cancer origin receiving only systemic chemotherapy and 14–24 months for patients with PC of ovarian origin.9–12
The approach to PC has changed since the 1980s, after the implementation of cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy (CRS + HIPEC), which was introduced experimentally by Spratt, introduced clinically by Sugarbaker, and promoted by a number of clinicians belonging mostly to the Peritoneal Surface Oncology Group International over the last 15 years.13,14 The CRS + HIPEC method is now a well-established treatment worldwide for PC with a mean per capita number of procedures of 15 patients/1 million per year. 15 Nevertheless, despite the excellent success of the CRS + HIPEC method, there are still patients with PC who cannot be treated with CRS + HIPEC due to a high PC index or because their physical condition only allows for a minimally invasive procedure without aggressive debulking. Another problem is that some patients initially require more than one cycle of specific intraperitoneal chemotherapy.16–18
On the 5th of November 2011, Reymond first performed and introduced a new PC treatment technique to the clinic, known as pressurized intraperitoneal aerosol chemotherapy (PIPAC), which became a promising therapeutic method for this group of patients that offered the potential to solve many of the previously unresolved problems associated with CRS + HIPEC. 19 To date, many HIPEC centres have added PIPAC as a therapeutic option. Currently, the PIPAC procedure is performed in nearly 30 countries, mostly in Western Europe, in a total of 100 highly specialized treatment centres. 20 For many countries, such as Italy, France and Denmark, implementation of this novel drug delivery technique occurred only within the last 2 years, in a smooth implementation period lasting 3–6 months. Many institutions worldwide are now preparing to undertake their first PIPAC procedure.21,22 To the best of our knowledge, based on a careful review of the literature and documentation through to May 2017 pertaining to the supply of specialized medical equipment necessary for PIPAC, no PIPAC procedures have been performed in the eastern part of Central Europe. This region includes the Visegrád countries such as the Czech Republic, Hungary, Poland and Slovakia. We are also not aware of any publications that have addressed the preparations involved in establishing a new PIPAC centre in this region.
The aim of this current study was to perform a retrospective, single-centre, detailed analysis of the preparations for performing the first PIPAC procedure at a surgical oncology centre in Bydgoszcz, Poland. This current report describes the major delays and other limitations that were encountered during the preparations and how these were resolved. It is our expectation that this current report will enable other institutions to implement PIPAC programmes in a timely manner without any unnecessary delays, possibly by eliminating some of the problematic aspects that are discussed herein.
Methods
This retrospective study analysed the period of 14 months (n = 14) prior to 10 May 2017 when the first patients underwent a PIPAC procedure in the Department of Surgical Oncology, Ludwik Rydygier’s Collegium Medicum, Nicolaus Copernicus University in Torun, Bydgoszcz, Poland. In addition, the study analysed the day of the first PIPAC surgery and the first 2 weeks after establishment of the PIPAC programme. The findings and analyses have been divided into five sections: (i) general preparations for the first PIPAC procedure; (ii) patient referral and qualification; (iii) the first PIPAC procedure; (iv) the 2 weeks following establishment of the PIPAC programme; and (v) general problematic issues that arose during preparation for the PIPAC procedures and how these were resolved. In these five sections, the following issues were analysed: practical aspects (including the surgical learning curve for PIPAC); creation of the multidisciplinary (MDT)-PIPAC team; obtaining the required paperwork, such as approvals and required permits; organization of the operating theatre and acquisition of the required equipment; patient recruitment and selection; and any other additional steps deemed essential to the establishment of the PIPAC programme at our institution. The fifth section, listed as general problematic issues, describes the most problematic issues and critical points that arose during preparation for the PIPAC procedures, of preparations that occurred in our institution for the procedure, including important turning points and the solutions used to resolve the problems that occurred.
All patients gave voluntary and informed consent to the planned treatment and the study was performed in accordance with the Declaration of Helsinki. Consent for all steps in this study were obtained from the Oncology Review Board of Ludwik Rydygier’s Collegium Medicum.
Results
General preparations for the first PIPAC procedure
This study evaluated the distribution of the amount of time spent on the key steps during the preparation for the first PIPAC procedure to be undertaken in the Department of Surgical Oncology, Ludwik Rydygier’s Collegium Medicum, Nicolaus Copernicus University in Torun, Bydgoszcz, Poland. These time periods are presented in Figure 1 and include the following: (i) the process of making a decision to establish the PIPAC procedure (2 months); (ii) an extensive literature review (7 months); (iii) the participation in international PIPAC meetings and workshops (4 months); (iv) visits to professional PIPAC centres (1 month); (v) the establishment of the MDT-PIPAC team (5 months); (vi) the completion of the required documents, approvals and certificates (12 months); (vii) the acquisition of the required instruments and equipment (7 months); (viii) the preparation of information, materials, tutorials and collection of patient consents, including their approval (4 months); (ix) the recruitment of candidate patients for the procedure and selection of the patients (3 months); (x) the oncological MDT evaluation of qualified patients with detailed analysis of all of their possible therapeutic options (1 month); and (xi) the preparation of patient specimens with histological and immunohistochemical evaluation by the Department of Anatomical Pathology, Ludwik Rydygier’s Collegium Medicum (1 month). As a result, a checklist based on the key preparation steps is presented in Table 1.
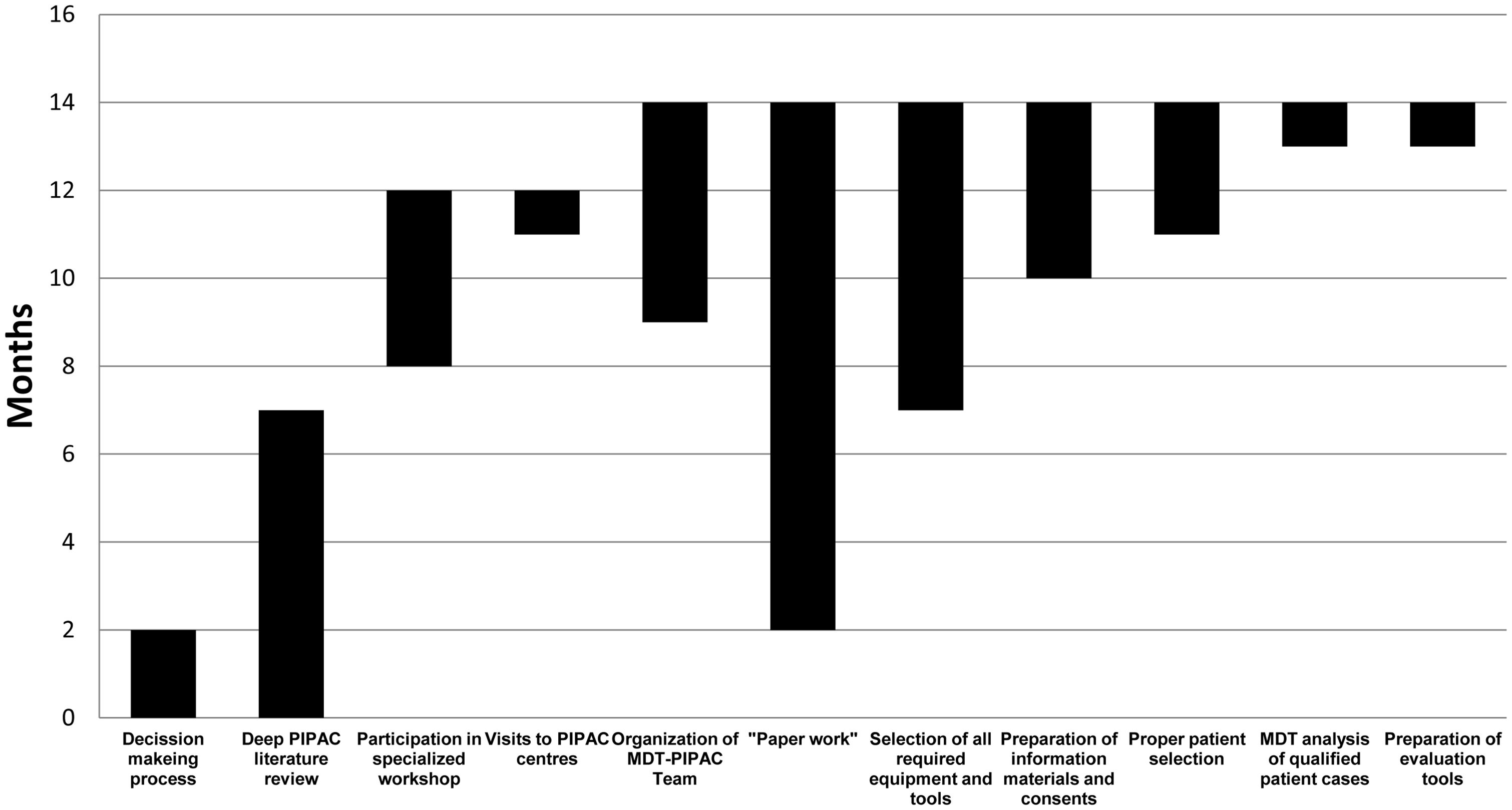
Time periods for each step during the 14-month preparation for the establishment of the first pressurized intraperitoneal aerosol chemotherapy (PIPAC) programme in the eastern part of Central Europe. MDT, multidisciplinary.
The most important steps of the preparation for the establishment of the first pressurized intraperitoneal aerosol chemotherapy (PIPAC) programme in the eastern part of Central Europe presented in the recommended sequence.

The authors attended one international PIPAC meeting held in Baltimore, Maryland, USA and one PIPAC workshop held in Lausanne, Switzerland. The authors visited two different PIPAC centres: (i) CHUV Lausanne University Hospital in Switzerland, while they attended the workshop; and (ii) Centre Hospitalier Lyon Sud in France, which supplied the possibility for active surgery assistance.
Formation of the MDT-PIPAC team started 9 months before the first PIPAC procedure and comprised of a team of 11 people with the skills and/or responsibilities that are detailed in Table 2. The longest step, which lasted approximately 1 year, involved completing the required paperwork, which entailed the collection and analysis of documents related to occupational health and safety, medical permits, certificates for all of the required equipment, protocol/procedure description materials, patient consents and legal approvals. The selection and ordering of required materials and tools were possible after attending the PIPAC workshop and meeting; four independent suppliers of medical equipment were needed.
Composition of the multidisciplinary pressurized intraperitoneal aerosol chemotherapy (PIPAC) programme team responsible for implementation of the first PIPAC procedure undertaken in the eastern part of Central Europe.

Patient referral and qualification
To determine which patients qualified for the PIPAC procedure, a thorough review of the scientific literature was undertaken in conjunction with the existing knowledge base and experience gained from being a HIPEC centre. Two qualification/recommendation criteria lists were developed (Tables 3 and 4) 23 and shared with medical oncologists and surgical oncologists at the Ludwik Rydygier’s Collegium Medicum as part of a call for patient referrals. The two lists called for patients in good clinical condition who qualified for a minimally invasive surgical procedure and who had a confirmed PC status with the PC originating from colorectal cancer, gastric cancer, ovarian cancer, appendiceal cancer, gallbladder cancer, pancreatic cancer, pseudomyxoma peritonei and peritoneal mesothelioma. After a 3-month evaluation period, two patients (male: 72 years; female: 49 years) with a PC of gastric cancer origin who met all the inclusion criteria were selected.
Qualification criteria for the pressurized intraperitoneal aerosol chemotherapy (PIPAC) procedure according to the clinical status and presentation of peritoneal carcinomatosis (PC) based on the current PIPAC procedure standards. 23

The list of recommended tumours of origin of the peritoneal carcinomatosis (PC) for patients being referred for the pressurized intraperitoneal aerosol chemotherapy (PIPAC) procedure.

The first PIPAC procedure
A single laparoscopic operating room equipped with laminar airflow was prepared for the first PIPAC procedure. One day before the procedure, all the required instruments and devices were tested and an inventory of all the required tools was thoroughly double-checked. One day before the procedure, a final briefing with all MDT-PIPAC members was organized. For each PIPAC procedure (one surgery for each patient), two different laparoscopic (LAP) sets were sterilized and prepared, including a 5 mm (30°) Olympus Autoclavable Laparoscope (Olympus, Tokyo, Japan). Thus, four LAP sets were prepared; two were designated for use in surgery and the other two were designated reserve LAP sets. Each LAP set additionally included one 5 mm and one 12 mm Kii blunt tip double balloon secured access trocar (Applied Medical, Rancho Santa Margarita, CA, USA), one CapnoPen laparoscopic nebulizer (Capnomed, Villingendorf, Germany) and one standard suction device. A double syringe Accutron HP-D Injector (Medtron AG, Saarbrücken, Germany) was used as the injection device during the PIPAC surgery.
During surgery, the patient was placed in the supine-basic position with extended and maintained legs. At the beginning of the surgery, the Veress-needle technique was used to establish a 12 mmHg capnoperitoneum. Four biopsies were performed for each patient with an additional local peritonectomy. The chemotherapy agents, 7.5 mg/m2 cisplatin and 1.5 mg/m2 doxorubicin, were used as suggested by several PIPAC centres, for 30 min at 37°C. 24 The total operating time was 108 min for the first patient and 96 min for the second patient. The toxic aerosol was released safely via a closed aerosol waste system.
The 2 weeks following establishment of the PIPAC programme
Regional oncologists were informed about the preparations for the first procedure at the beginning of the programme implementation. During this period, the authors started to look for potential patients that could be referred for the PIPAC procedure. During the 2 weeks following the successful performance of the first PIPAC procedure, the number of patient referrals increased. The number of regional medical oncologists referring their patients for the PIPAC procedure increased from one who was interested initially in the procedure to 24 who were interested in referring their patients for the PIPAC procedure by the end of the first 2 weeks. There were also new groups of medical specialists referring their patients for the procedure including general surgeons, general practitioners, gastroenterologists and gynaecologists. There were also some return referrals from palliative care specialists. Before the first PIPAC procedure, oncological surgeons (33 of 34; 97%) were the primary referrers.
General problematic issues
The time that it took the Ludwik Rydygier’s Collegium Medicum to prepare for the first PIPAC procedure was extremely long compared with other European Union PIPAC centres: 14 months versus a standard 3–6 months of preparation. During general preparations for the first PIPAC procedure, three key problems arose. The first occurred at the time that the institution decided to establish a PIPAC programme. Establishing this procedure for the first time in the eastern part of Central Europe had some inherent difficulties, most notably a lack of regional scientific and technical expertise in the PIPAC procedure and familiarity/experience with the efficient and timely establishment of the technique. This issue was resolved by our active participation in a PIPAC meeting and workshop and by visiting PIPAC centres. A second major issue involved the acquisition of the required devices and tools for the procedure. The primary contacts and vendors for the required equipment were not represented in the eastern part of Central Europe. In addition, due to Polish medical safety and anticorruption regulations, it was not possible to simply order all of the required equipment via another country such as Germany or Switzerland. This problem was resolved in two ways. First, the main company headquarters of the equipment were contacted to obtain information about any possible Polish representatives. Secondly, we appealed for representation in Poland by these companies and for assistance in obtaining any needed permits, approvals and certificates. This particular issue was a rate-limiting factor in the preparations because it took 7 months to acquire the necessary equipment and associated paperwork. The last major issue entailed patient recruitment and the number of patients qualifying for PIPAC as well as the relatively small initial response to our call for patients from medical oncologists (one of 34; 3%) compared with the very high response from surgical oncologists (33 of 34; 97%). This problem was resolved with several informational campaigns and the creation of rigorous and clear qualification criteria.
Discussion
The PIPAC technique has become an important, innovative method of drug delivery that offers hope to patients suffering from PC, who due to some limitation(s) cannot be treated using other methods, such as CRS + HIPEC. 25 This hope has been supported by a growing number of published clinical and preclinical studies in the field of PC treatment indicating that PIPAC is highly efficient.23,26
The implementation of any new and especially innovative technique is not simple.27,28 This situation is well observed in specific medical specialties such as oncology and surgery.29,30 On 10 May 2017, two patients underwent PIPAC procedures performed for the first time in the Department of Surgical Oncology, Ludwik Rydygier’s Collegium Medicum, Nicolaus Copernicus University in Torun, Bydgoszcz, Poland. The intensive preparations for this procedure lasted for over 1 year (14 months) and consisted of several important steps. The time of implementation was significantly longer than in other centres because of bureaucratic problems related mainly to the organization of paperwork and the first use of specific medical devices and surgical tools in Poland.
At the beginning of the process of establishing PIPAC at our institution, as in many cases wherein a new treatment technique is implemented, our first contact with PIPAC occurred through an extensive analysis of the literature using the standard public bibliographic database PubMed. 27 The preparation stage required the most time (12 months) during the establishment of PIPAC and involved completing the required paperwork, such as obtaining approvals and certificates. Indeed, increased administrative and bureaucratic requirements appear to be a limitation experienced around the globe for implementing new therapeutic procedures. 31 An important preparation step, which accelerated the learning curve, was the authors’ participation in a PIPAC workshop (4 months) and visits to other PIPAC centres (1 month), which increased the probability of success. While the importance of carefully planned preparation steps for establishing PIPAC is self-evident, the literature supports that methodical preparation in the implementation of new therapies positively impacts the safety and feasibility of the planned procedure, obtains positive therapeutic effects, improves the learning curve for the procedure and facilitates the acquisition of a strong knowledge base and understanding of the principles of the technique, all of which are critical for good medical practice. 32 The final number of individuals in the MDT-PIPAC team was 11, which contrasts with the standard number of individuals involved in a PIPAC procedure (usually only two surgeons, two scrub nurses and one anaesthetist). 33 The increased number of personnel on our team was due to the need to create a unified team responsible for the assessment of patient qualification, the performance of the procedure and the assessment of the outcome. Overall, in our opinion, the early formation of the interdisciplinary team during the preparation phase was essential to the success of the procedure and appears to be in line with current trends in surgical oncology. 34 Our two main qualification criteria for PC patients were related to factors associated with the condition of the patient and to the aetiology of the cancer. This biphasic qualification methodology was formulated based on the literature and was consistent with the qualification criteria used by other PIPAC centres that aim to precisely determine eligibility for the procedure.35,36 We recommend strict adherence to published standard PIPAC protocols, as we did during the performance of our first PIPAC procedure. We believe that this adherence will not only satisfy important legal and safety considerations but also enable the acquisition of uniform data from among PIPAC centres, as demonstrated by historical examples, such as the introduction of robotic techniques in surgery.37,38
The 2 weeks following the successful PIPAC procedure were particularly interesting. Starting the day after the surgery, there was a marked increase in the number of inquiries for PIPAC surgery. Our general call centre was unprepared for the high number of calls by patients with cancer metastases other than PC seeking information about PIPAC. We remedied this by creating PIPAC and PC instructional tutorials and by introducing broad media campaigns using the internet and social media to widen our reach of patients and their families. 39 There were also a significantly higher percentage of patients referred for PIPAC by medical oncologists, which is a group of specialists who initially responded poorly to our call for potential patients for PIPAC surgery. We speculate this change may have been due to an effect described by Kleinrock termed ‘challenging choices’ in oncology innovation, wherein healthcare professionals opt to wait for the positive effects or success of an innovative treatment to be realized before introducing it to their patients because of a fear of responsibility and/or funding concerns. 40 Another factor driving the change may be related to the curiosity sparked by the success of our first PIPAC procedure, coupled with the underlying hope and desire to find a novel PC treatment possibility. 41
In conclusion, this current report has described the successful establishment of a new PIPAC programme. The time taken to implement this new methodology was significantly longer than in some other PIPAC centres, probably due to the long bureaucratic processes involved. We anticipate that our findings will help new centres, especially in this part of Europe, to more quickly implement PIPAC programmes, through the elimination of the major problems that we experienced and discussed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
