Abstract
Congenital leukemia (CL), defined as manifestations of leukemia within the first 4 weeks of life, is a rare condition with an estimated incidence of only 1 to 5 per million live births. Despite extensive research and the clinical application of new therapies, the prognosis of CL remains poor. Few large-scale studies have investigated the factors affecting the outcomes of infants with CL. Here, we conducted a retrospective study and analysis of CL cases published in the English language from 1 January 2001 to 1 May 2016. Our goal was to provide updated information about this rare disease and to investigate factors that may affect the outcomes of patients with CL.
Introduction
Congenital leukemia (CL) is defined as manifestations of leukemia within the first 4 weeks of life. CL is one of the most common cancers in neonates, after teratoma and neuroblastoma, 1 but is nevertheless rare and almost always lethal without chemotherapy. 2 The pathophysiology and prognosis of leukemia in this age group differ from those in cases occurring later in life. 3 Although the etiology is unknown, the presence of leukemia at birth suggests possible intrauterine exposure to drugs or other toxins or the presence of genetic abnormalities, and KMT2A rearrangement has accordingly been reported in 25% to 40% of cases.4,5 Despite recent multicenter clinical trials, 6 the prognosis of CL remains very poor, with a 2-year overall survival rate of only 23% prior to 2000. 7 The widespread application of immunohistochemical and cytogenetic analyses since the beginning of the century has improved our understanding of the relationships between cytogenetic abnormalities and CL, especially KMT2A translocations, thus providing more evidence to help physicians to manage patients and predict possible outcomes for neonates with CL. 5 In 2002, Bresters et al. 7 published a general review based on an analysis of cases with concise clinical data reported from 1975 to 2000. However, given that few large-scale studies have examined the factors affecting the outcomes of infants with CL since then, we conducted a review and analyzed cases of CL reported in the English language from 1 January 2001 to 1 May 2016.8–61 The aim was to provide updated information on this rare disease and to investigate factors that may affect patient outcomes.
Methods
Literature search
We defined CL as leukemia occurring in the first 4 weeks of life, together with the following criteria: (1) infiltration of extrahematopoietic tissue; (2) absence of any other disease causing leukemoid reactions, such as feto-maternal blood incompatibilities or intrauterine infections (in which dermal hematopoiesis can persist; e.g. syphilis, rubella, cytomegalovirus, toxoplasmosis, and herpes simplex infections); and (3) absence of constitutional disorders associated with ‘unstable’ hematopoiesis such as Down’s syndrome. 3 We conducted a retrospective analysis of previous studies based on a comprehensive search of the MEDLINE, Cochrane, and OVID databases for English-language papers on CL published from 1 January 2001 to 1 May 2016. We used the MeSH terms “neonate” or “newborn” or “neonatal” or “congenital” and “leukemia” to identify cases of CL confirmed by pathological diagnosis. Patients with suggested transient myeloproliferative disorder or a leukemoid reaction were excluded.
Clinical information
Clinical information was collected on the type of leukemia, sex, location of extramedullary involvement, the results of routine blood tests and bone marrow (BM) smears, prenatal abnormalities, cytogenetics, therapies, and prognoses. The duration of follow-up and outcomes at the ends of the follow-up period were also noted. The clinical characteristics of patients with and without mixed lineage leukemia (KMT2A) translocations were also described.
Statistical analysis
The results were analyzed using descriptive statistics for individual clinical data, univariate and multivariate Cox regressions, Pearson’s χ2 tests, and Mann–Whitney U tests. Factors that could affect patient outcomes were analyzed by univariate and multivariate Cox regression analyses and outcomes were compared between groups using the Kaplan–Meier method. Differences were analyzed and considered significant at P < 0.05. All statistical analyses were performed using SPSS for Windows, Version 21.0 (IBM Corp., Armonk, NY, USA).
Results and discussion
Clinical features
Fifty-nine patients diagnosed with CL according to the study criteria were analyzed. One patient was stillborn but was confirmed with leukemia at autopsy, and one patient who showed myeloid sarcoma was also included. The clinical features of the patients are shown in Table 1. As shown in the previous systematic review, 7 acute myeloid leukemia (AML) was significantly more common than acute lymphoblastic leukemia (ALL) (P<0.001). However, in contrast to previous reports3,7 indicating a male:female ratio of 2:1, the current study included 66.1% female and 33.9% male patients. A total of 67.8% of patients had skin infiltration and 47.5% had hepatosplenomegaly, hepatomegaly, or splenomegaly. Leukemia cells were detected in the cerebrospinal fluid in 25.4% of the patients. Hyperleukocytosis was detected in 62.7% of patients (37/59), with an average white cell count of 68.5 × 109/L (normal range, 4–10 × 109/L), but anemia was less common (15/42, 35.7%), and the average platelet count was 78 × 109/L (normal range, 100–300 × 109/L). Blast cells were present in most patients’ peripheral blood (PB) and BM (32/44, 72.7%).
Clinical data for patients with congenital leukemia reported in the literature from 2001 to 2016 (n = 59).

Values given as number of patients with the characteristic/number of patients in which the presence or absence of the characteristic was described, or as median (range). ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; AUL, peroxidase positivity <3% of blasts and no lymphoid or myeloid (<20% positivity) surface markers; CNS, central nervous system; WBC, white blood cells; HB, hemoglobin; PLT, platelets; PB, peripheral blood; BM, bone marrow; SR, spontaneous remission.
The median lifespan was 210 days and the 2-year survival rate was 44.2% (Figure 1), which was significantly higher than the equivalent survival rate of 23% in patients from 1975–2000 (P = 0.008). 7 A total of 57.6% patients received chemotherapy and had an average lifespan of 734 days, compared with an average lifespan of 646 days among patients without chemotherapy (P = 0.12), with 2-year overall survival rates of 47% and 41%, respectively (Figure 2). However, patients with KMT2A translocations were more likely to receive chemotherapy but their outcomes were worse. This result may thus have been caused by different distributions of KMT2A-positive patients in the two groups. Further analysis showed that more patients who received chemotherapy were KMT2A-translocation positive (56.3% vs 16.0%, P = 0.02), and we therefore calculated the survival rates and median lifespans of patients with and without chemotherapy in the KMT2A-positive and -negative groups, respectively. The 2-year survival rate of patients with KMT2A translocation was 0%, irrespective of the use of chemotherapy, but the median lifespans were 48 days with therapy and only 2 days without (P = 0.032). In contrast, the 2-year survival rates in patients without KMT2A translocation were 69.3% with therapy and 53.3% without, with respective median lifespans of more than 1460 and 1277 days, respectively, though these differences were not significant. These results suggest that chemotherapy could increase the lifespan of patients with KMT2A translocation but did not benefit patients without this translocation. Overall, chemotherapy had no effect on the 2-year survival rate or median lifespan.

Two-year overall survival rate of patients with congenital leukemia. The 2-year overall survival rate among all 59 patients was 44.2% and the median lifespan was 210 days. pOS, overall survival percent.

Two-year overall survival rates of patients with congenital leukemia with and without chemotherapy. There was no significant difference in 2-year overall survival rates between 34 patients with chemotherapy and 25 patients without chemotherapy. pOS, overall survival percent.
Initial symptoms that raised the attention of the physician and parents included cutaneous lesions (most common), hepatosplenomegaly, pallor, jaundice, epistaxis, nephromegaly, intracranial hemorrhage, fever, and abnormal PB counts.8–61 Prenatal findings suggesting potential abnormalities included hydramnios and organ masses;8–61 however, most prenatal tests showed no abnormalities. Given that CL can result in stillbirth, 58 the incidence of CL may have been underestimated.
Univariate Cox regression identified hepatosplenomegaly, genetics, and spontaneous remission (SR) as factors potentially affecting survival, but multivariate Cox regression including these three factors showed that only genetic results and SR had significant impacts on survival.
KMT2A translocations in CL
The 2-year survival rate was 0% (n = 22) for patients with a KMT2A translocation and 69.5% (n = 29 for those without a KMT2A translocation (P = 0.001) (Figure 3). No patients in the KMT2A-positive group experienced SR. Although it has been suggested that these patients should receive chemotherapy, 5 no randomized trial has proven a benefit of chemotherapy over supportive care in this population. In our study, 15 of 22 patients in the KMT2A-positive group received chemotherapy and had an average survival of 142 days, compared with only 25 days in the seven patients who only received supportive treatment (P = 0.03). This supports the suggestion that patients with KMT2A translocations should receive chemotherapy.

Two-year overall survival rates of patients with congenital leukemia with and without KMT2A translocations. The 2-year overall survival rate was significantly higher in the 29 patients without KMT2A translocations compared with the 22 patients with translocations (P = 0.001). pOS, overall survival percent.
Some previous studies3,5,7 have suggested that the type of CL (AML or ALL) may affect patient outcomes. However, the current study found no effect of lineage type on outcome. Regarding KMT2A translocations, a significantly higher proportion of patients with ALL had translocations compared with those with AML (8/11 vs 12/38) (P = 0.03). Additionally, no patients with ALL experienced SR. The different distributions of KMT2A translocations between the AML and ALL groups may explain the poorer outcomes among patients with ALL. A total of 12/38 AML patients had KMT2A translocations, with an average lifespan of 37 days, while the remaining 26 patients with AML without translocations had an average lifespan of 961 days (P<0.0001). This suggests that KMT2A translocation was associated with poorer outcomes in patients with AML. Apart from KMT2A translocation, other cytogenetic abnormalities, including recurrent cytogenetic abnormalities such as t(8; 16)(p11.2; p13.3)/KAT6A-CREBBP and t(8; 21)(p11; q22), and non-recurrent cytogenetic abnormalities such as t(5,9),14p/8p deletion were also noted, but the number of patients with these cytogenetic abnormalities was very small (16/59, 27.1%), making statistical analysis impossible. We therefore focused on KMT2A translocations in the current study. More detailed information on the other cytogenetic abnormalities in CL is provided by Bain et al. 62
Patients with and without KMT2A translocations present with distinct clinical features and may therefore be dealt with differently (Table 2). Interestingly, we noted that patients with KMT2A translocations had high WBC levels and high percentages of leukemic cells in the PB and BM.
Clinical characteristics of patients with congenital leukemia with and without KMT2A translocations reported in the literature from 2001 to 2016.
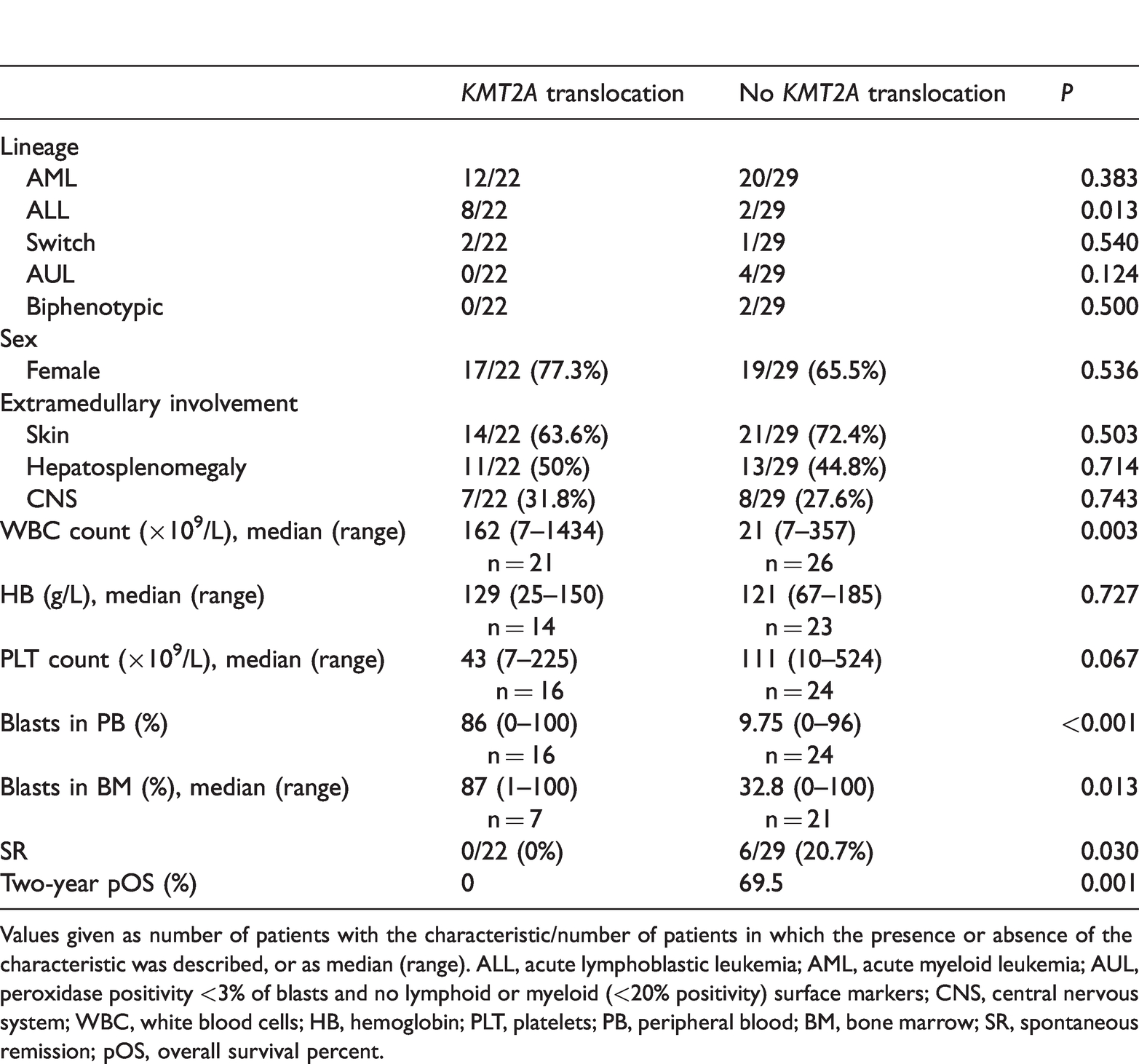
Values given as number of patients with the characteristic/number of patients in which the presence or absence of the characteristic was described, or as median (range). ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; AUL, peroxidase positivity <3% of blasts and no lymphoid or myeloid (<20% positivity) surface markers; CNS, central nervous system; WBC, white blood cells; HB, hemoglobin; PLT, platelets; PB, peripheral blood; BM, bone marrow; SR, spontaneous remission; pOS, overall survival percent.
SR in CL
The distinct outcomes and clinical features of patients with and without KMT2A translocations suggest the need to treat them differently. In this study, more patients in the KMT2A-negative group were managed conservatively, and chemotherapy had no significant effect on the outcomes of these patients (average lifespans 1094 days with chemotherapy vs 995 days without). Among those who did not receive chemotherapy, 35.3% (5/17) experienced SR. We also compared the clinical data between patients who did and did not experience SR (one SR case without genetic information was excluded) (Table 3). All cases of SR involved sustained spontaneous remissions during follow-up, with an average follow-up duration of 1825 days. Among the six patients with SR with available genetic information, two had normal cytogenetics, one had t(5,9),14p/8p deletion, and three had t(8;16)(p11.2; p13.3)/KAT6A-CREBBP; however, another two patients with t(8;16)(p11.2; p13.3)/KAT6A-CREBBP did not achieve SR. Consistent with previous studies,7,59 these results suggest that patients with neonatal leukemia associated with t(8;16)/KAT6A-CREBBP may achieve SR. Cytogenetic analysis should thus be performed in CL patients to help choose the appropriate therapies and predict the prognosis. 62 However, the use of chemotherapy in patients without KMT2A translocations remains controversial. It has been suggested that early chemotherapy should be restricted to progressing cases, defined by an increasing peripheral blast count and/or BM failure.37,59 However, there have also been reports of patients with systematic progression who subsequently experienced SR.20,56
Clinical characteristics of patients with congenital leukemia without KMT2A translocation who did and did not experience SR.

Values given as number of patients with the characteristic/number of patients in which the presence or absence of the characteristic was described. ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; AUL, peroxidase positivity <3% of blasts and no lymphoid or myeloid (<20% positivity) surface markers; CNS, central nervous system; WBC, white blood cells; HB, hemoglobin; PLT, platelets; PB, peripheral blood; BM, bone marrow; pOS, overall survival percent.
It is difficult to predict if certain patients will achieve SR. However, the current and previous studies suggest that SR may be associated with AML type, comparatively normal routine blood test results, fewer blasts in the PB and BM, and skin infiltration but no hepatosplenomegaly or CNS infiltration. 56
Conclusion
KMT2A translocation is a risk factor leading to poor outcomes among patients with CL. Chemotherapy did not affect the outcomes of patients without KMT2A translocations but could prolong the average lifespan of patients with KMT2A translocations from around 25 to 142 days. This suggests that intensive chemotherapy should only be applied in patients with KMT2A translocations, while patients without KMT2A translocations may be managed more conservatively and may subsequently achieve SR. Patients with CL should thus undergo cytogenetic analysis to help select the appropriate therapies and predict the prognosis. Nevertheless, there are currently no solid evidence-based recommendations for the treatment of CL, and further studies and new therapies are urgently required to improve outcomes.
Footnotes
Availability of data and materials
Please contact the author with any data requests.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The National Natural Science Foundation of China, 81873847; Guangzhou Technology Program, 201707010398, 201804010380.
