Abstract
The case was a 17-year-old young woman with a one-year history of recurrent abdominal pain and discomfort. B-scan ultrasonography identified intussusception and contrast-enhanced computed tomography of the pelvis revealed volvulus. A laparoscopic procedure was planned to identify the reason for the intussusception and obstruction. Intraoperatively, the intussusception was found to be caused by a cauliflower-shaped polypoid tumor measuring approximately 4 × 3 cm. Postoperative pathological examination identified the tumor to be a traditional serrated adenoma of the small intestine, which is rare and has atypical clinical manifestations. If unexplained abdominal pain or gastrointestinal bleeding occurs and an abdominal mass cannot be accurately located, laparoscopic or open surgery should be performed immediately. Early surgery is the most effective and reliable way of securing a prompt diagnosis and a favorable prognosis.
Keywords
Introduction
The clinical symptoms and signs of serrated adenoma are non-specific and diagnostic methods are relatively limited, which may lead to delayed diagnosis or misdiagnosis. In recent years, there has been a gradual increase in the incidence of gastrointestinal tumors. However, tumors in the small intestine are rare, accounting for only 1% to 3.6% of all gastrointestinal tumors. 1 Serrated adenomas are particularly rare in the small intestine and are more likely to occur in the colon or rectum than in other parts of the gastrointestinal tract. Therefore, there are no guidelines or consensus recommendations with regard to the natural course of serrated adenoma in the small intestine or its diagnosis, treatment, and prognosis. However, a previous report suggests that serrated adenoma in the small intestine may be more malignant and aggressive than that in the large intestine. 2 This report describes a case of intussusception caused by a traditional serrated adenoma (TSA) of the small intestine in an adolescent female patient admitted to the Department of Gastrointestinal Surgery at the Affiliated Hospital of Zunyi Medical University. In reporting this case, we hope to improve the understanding of TSA in the small intestine and its diagnosis and treatment.
Case report
A 17-year-old unmarried female patient was admitted to our hospital with a 1-year history of intermittent abdominal pain that had worsened in the previous week. She had been diagnosed to have “incomplete intestinal obstruction” at another hospital and discharged after conservative treatment. However, her abdominal pain and discomfort continued and did not respond to self-medication with traditional Chinese or Western medicine. One week before presenting to our hospital, the patient developed severe abdominal pain, which was mainly in the lower abdomen and more pronounced after eating. She also complained of mushy diarrhea that occurred approximately four to five times a day. The patient was previously healthy with an unremarkable medical and family history. On physical examination, her vital signs were normal and findings otherwise were unremarkable. The abdomen was soft and flat, no abdominal mass was palpated, and bowel sounds were normal. The lower abdomen was tender but there was no rebound tenderness. Routine blood tests revealed a white blood cell count of 5.92 × 109/L, hemoglobin of 126.0/L, a platelet count of 246 × 109/L, and a neutrophil count of 0.7. There were no significant abnormalities in her liver or kidney function tests or electrolyte levels. Tests for gastrointestinal tumor markers were negative. Contrast-enhanced computed tomography (CT) of the pelvis revealed volvulus in the lower abdomen and ileum (Figure 1), a small amount of fluid in the pelvic and uterine cavities, a small amount of gas in the intestines, and multiple lymph nodes in the abdominal cavity, including some that were slightly enlarged. Abdominal B-ultrasonographic examination (Figure 2) revealed a concentric ring sign in the right lower abdomen, suggestive of intussusception.
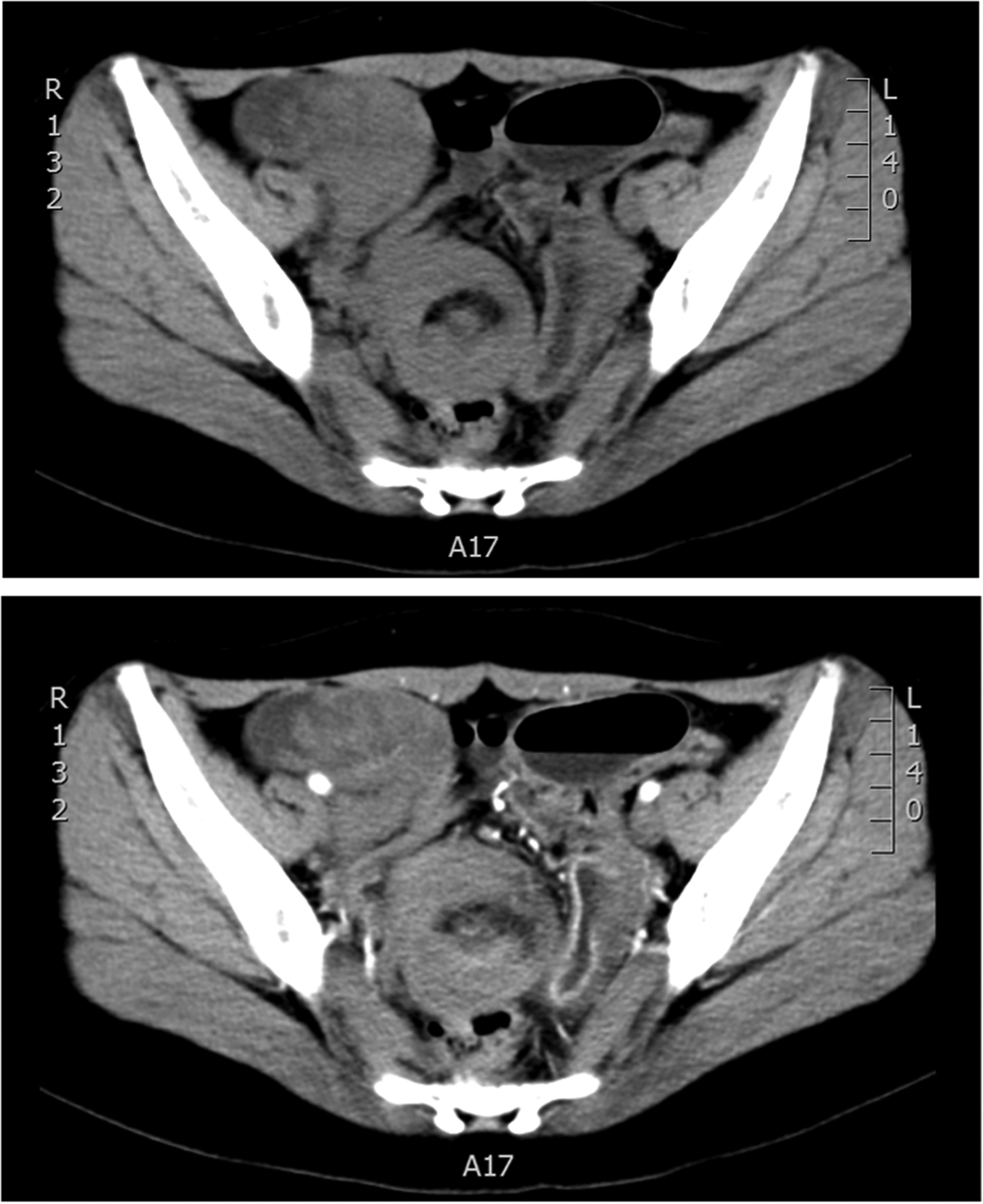
Computed tomography scan showing volvulus of the lower abdomen and intestine. The length of the twisted intestine was approximately 62 mm.

B-ultrasonography scan of the abdomen showing the concentric ring sign. The cross-sectional size is about 37 × 27 mm.
The patient’s abdominal pain and distension gradually worsened despite conservative measures, which included mild enemas, antibiotic therapy, and rehydration. Therefore, we proceeded to diagnostic laparoscopy. Intussusception was considered as a cause of the patient’s abdominal pain, localized peritonitis, and pelvic volvulus preoperatively. Ultrasonography suggested intussusception and CT had suggested torsion. To identify the cause of this female adolescent patient’s abdominal pain and exclude nesting and obstruction, emergency laparoscopic exploration was performed in the general surgery department on the day following admission. Laparoscopic examination revealed telescoping about 50 cm into the ileum, which formed an obstruction. Severe intestinal adhesions were found in an area where the mesangium and surrounding omentum overlapped. We then switched from laparoscopic surgery to laparotomy, which revealed a cauliflower-shaped hard polypoid tumor measuring approximately 4 × 3 cm (Figure 3). Intraoperatively, there was no evidence of regional lymph node enlargement, seeding in the liver, spleen, or other abdominal organs, or metastasis. The tumor and the part of the small intestine that was involved were resected, a peristaltic anastomosis was performed, and a pelvic drainage tube was placed.

Slightly hard cauliflower-shaped polypoid tumor measuring approximately 4 × 3 cm was found in the mid ileum at the intussusception site.
After surgery, the need for sexual abstinence, fasting, prevention of infection, nutritional support, regulation of intestinal function, water and electrolyte intake, and maintenance of acid–base balance was discussed with the patient. Passage of gas and intestinal peristalsis resumed spontaneously on postoperative day 1; intake of water was gradually resumed with introduction of a liquid diet. Postoperative pathological analysis revealed a TSA of the small intestine (Figure 4). Immunohistochemistry revealed membranous expression of β-catenin, suggesting adenocarcinoma. Genetic testing was also positive for the mismatch repair genes associated with adenocarcinoma (MLH1, MSH2, MSH6, and PMS2). The Ki-67 index was approximately 80% and P53 status was positive. On postoperative day 7, when fewer than 50 mL of clear abdominal fluid were drained, the drainage tube was removed and the patient was discharged. Her condition improved significantly after surgery, with resolution of her abdominal pain and diarrhea. The reporting of this study conforms to the CARE guidelines. 3

(a) Histopathological examination of tumor cells revealed irregular serrated structures, including serrated glands and epithelial dysplasia (HE staining, ×100). (b) Histopathological examination of the lumen demonstrated serrated changes (HE staining, ×40) and (c) Mature goblet cells were observed at the bottom of the crypts, with abnormal nuclei, flattened nuclei on the basal side, and abundant cytoplasmic secretion (HE staining, ×40).
Discussion
Tumors of the small intestine are insidious, accounting for 1.0% to 3.6% of all gastrointestinal tumors and 0.2% to 0.3% of all systemic tumors. 1 The majority of tumors in the small intestine are malignant, with only approximately 25% being benign. The most common site is the ileum followed by the duodenum and jejunum, and 50% to 70% of benign tumors at these sites are adenomas or leiomyomas. 4 Approximately 30% to 40% of small bowel tumors are adenocarcinoma. 5 Most small bowel adenocarcinomas are found in the duodenum (54.1%–57.0%) followed by the jejunum (25.8%–29.0%) and ileum (10.0%–20.0%).6,7 There are very few reports on small bowel adenomas, and reports on serrated adenoma in the ileum are extremely rare, reflecting the very low incidence of adenoma in the small intestine.
In recent years, various researchers have investigated the mechanism of progression of serrated lesions. Serrated polyps are heterogeneous lesions that can be divided into hyperplastic polyps, TSAs, and sessile serrated lesions. 8 Serrated adenoma is a precursor lesion of colorectal cancer.9,10 It has been suggested that serrated adenoma in the small intestine is an invasive lesion with high malignant potential. Like in the colon or rectum, adenoma in the small intestine is considered to be a precursor of adenocarcinoma, and villous adenoma is the most common morphology. 3 There are few clinical studies and reports on serrated adenoma of the small intestine, which most commonly occurs in the duodenum and is similar to TSA in the colorectal region. Serrated adenomas are a rare subtype of the serrated lesions that occur in the colon or rectum, accounting for only 5% of all these lesions. 11 On pathological examination, these lesions are morphologically characterized by a serrated structure, granular eosinophilic cytoplasm, a serrated lumen, ectopic crypts, and slender pencil-shaped nuclei. 11 The mechanism via which intestinal tumors develop is not fully understood, and most of the current studies are in colorectal cancer. The conventional pathway is thought to start with inactivation of the adenomatous polyposis coli tumor suppressor gene, which leads to activation of several oncogenes, including KRAS, TP53, and SMAD4. 11 However, BRAF mutations have been identified as key drivers in serrated lesions (including sessile serrated adenomas/polyps, TSAs, and hyperplastic polyps). Another known driver of serrated neogenesis is oncogenic mutations in KRAS (commonly codon 12/13), which, like BRAF mutations, also lead to activation of components of the MAPK signaling cascade. 12 Moreover, the serrated morphology is retained in only about one-third of colorectal cancers that develop through the serrated pathway, while the remaining two-thirds lose their serrated histological characteristics when they progress to advanced cancer.13,14 β-catenin is an important component of the Wnt signal transduction pathway and is mainly involved in gene expression and mediating cell adhesion. In this case, immunostaining for β-catenin was positive for membranous protein and was not nuclear. An increasing body of evidence shows that dysregulation of the Wnt/β-catenin signaling pathway can lead to development of malignant tumors by affecting apoptosis. Abnormal regulation of β-catenin gene expression leads to abnormal activation of this pathway and increases the expression level of β-catenin protein, which leads to tumorigenesis. Nuclear expression of β-catenin may suggest the possibility of malignancy. 15 We compared the pathological findings and molecular changes in the 13 serrated adenomas in the small intestine reported by Rosty et al. 3 (Table 1). Six of these lesions showed focal highly atypical hyperplasia. In one case, an adenoma at the duodenojejunal junction transformed into serrated adenocarcinoma; P53 expression was positive in this case, suggesting that the risk of malignant transformation may be higher for serrated adenomas of the small intestine than for conventional adenomas with less highly atypical hyperplasia. There was no significant relationship between pathological findings and patient age or sex. Only 1 of the 13 cases was located in the terminal ileum, and the remaining 12 cases were located in the duodenum, which further underscores the rarity of our case of serrated adenoma in the ileum. The adenoma in our patient was relatively large and indicated for surgery.
Clinical and pathological characteristics of 13 patients with serrated neoplasm of the small intestine.3

SA, serrated adenoma; SAHG, serrated adenoma, high-grade dysplasia; SAC, serrated adenocarcinoma.
The molecular mechanisms of colorectal tumorigenesis include three main pathways: chromosomal instability, microsatellite instability, and epigenetic instability. The main cause of microsatellite instability is dysfunction of mismatch repair proteins (MLH1, MSH2, PMS2, and MSH6). The mismatch repair genes were all positive without deletions in our patient, consistent with the 13 cases shown in Table 1. The phosphatase and tensin homolog (PTEN) gene is frequently mutated in the serrated pathway. 16 This mutation pattern is unique to microsatellite-unstable malignancies. Ninety percent of the TSAs and microsatellite-stable CRCs in Table 1 were serrated, and the majority (65%) were diagnosed as serrated adenocarcinoma. TSA and microsatellite-stable CRC share similar unique modes of carcinogenesis. These two entities show a common histological pattern with similar molecular alterations and most likely constitute the TSA pathway.
It should be noted that, in our case, we only obtained immunohistochemistry results, and did not conduct other tests. Significantly, the Ki67 index, one marker of tumor malignancy, was 80%, which is higher than that in the previous cases. The data published by Rosty et al. 3 indicate that the Ki67 index value is significantly correlated with high dysplasia. Morphologically, adenoma in the small bowel is similar to adenoma in the colon but the villus component is higher. At the molecular level, the mechanism of carcinogenesis in the small intestine is still unclear, although studies have shown that small bowel adenocarcinoma and colorectal adenocarcinoma follow different paths. 17
Tumors of the small intestine are rare in adolescents and have an insidious onset. Serrated adenoma in the ileum, as in this case, is even rarer. In the early stages, these tumors are usually asymptomatic with atypical clinical manifestations and complications. Therefore, there is a high likelihood of delayed diagnosis or misdiagnosis and the period during which treatment is likely to be most effective is often missed. Delayed diagnosis and missed treatment rates are high, and the misdiagnosis rate is 70% to 90%. 18 As the tumor progresses, compression or obstruction and other serious complications occur, and most patients have advanced disease by the time they consult a doctor. Clinical manifestations include abdominal pain, intestinal bleeding, intussusception, intestinal obstruction, and an abdominal mass. Tumors in the small intestine can induce chronic or recurrent intussusception. Intussusception of the small intestine is a specific type of intussusception with a low incidence and can occur in both adults and children. 19 Therefore, these tumors can be difficult to diagnose preoperatively. Our patient presented with abdominal pain as a result of intussusception caused by a tumor in the small intestine and had been diagnosed without a detailed examination to have incomplete intestinal obstruction at another hospital, which resulted in inappropriate treatment and a delayed diagnosis. Unlike traditional adenomas, serrated adenomas of the small intestine may have a higher risk of malignant transformation. Although reports of serrated adenomas of the small intestine are relatively rare, they may represent a unique morphological and biological subtype with malignant potential. 20 Patients with malignancy of the small intestine have a poor prognosis, with a 5-year survival rate of only 21% to 42%. 21 Early diagnosis and surgery are key to treating malignant intestinal tumors. Although many factors affect the prognosis, recurrence and metastasis are the main causes of death. Therefore, the best standard chemotherapy and comprehensive treatment are crucial and need further investigation.
Small bowel tumors require immediate surgery. Laparoscopy or laparotomy should be performed urgently in patients with unexplained abdominal pain, gastrointestinal bleeding, or an abdominal mass that cannot be accurately located but is suspected clinically to be in the small bowel. Early surgery is the most effective and reliable way of avoiding delayed diagnosis and a poor prognosis. In our case, when abdominal CT and B-ultrasonography indicated intussusception, emergency laparoscopic exploration was performed. After a tumor was identified as the cause of intussusception, the tumor, diseased bowel, and corresponding mesentery were removed. Postoperative infection should be prevented and nutritional support, pelvic drainage, and other symptomatic treatments provided. Our patient has recovered with no evidence of tumor recurrence. Improving the rate of early diagnosis based on clinical symptoms and auxiliary investigations could improve the clinical cure rate and the prognosis. If clinical symptoms such as intestinal intussusception and obstruction are observed, it is important to consider other diseases of the small intestine, including a tumor, and whether laparotomy is needed for diagnosis and treatment. When a tumor of the small intestine is suspected, further imaging is helpful for diagnosis. The diagnosis and exclusion rates for abdominal B-ultrasonography and CT have been reported to be higher than 90%. 22
Conclusion
Serrated adenomas of the small intestine, whether benign or malignant, are rare. Surgery was the main treatment option in this patient, who presented with abdominal pain and was found to have intussusception caused by a serrated adenoma in the small intestine with an insidious onset. An early benign tumor of the small intestine was diagnosed postoperatively. There were few postoperative complications and the prognosis was good.
Research Data
Research Data for Small bowel intussusception caused by a serrated adenoma: a case report
Research Data for Small bowel intussusception caused by a serrated adenoma: a case report by Yi Chen, Jiwei Wang, Maijian Wang, Xinbing Zheng and Jianguo Li in Journal of International Medical Research
Footnotes
Acknowledgement
The authors appreciate the patient’s willingness to participate in this research.
Author contributions
JW and JL managed the patient’s care. YC, MW, and XZ contributed to data collection and analysis. YC drafted the manuscript. JL revised the manuscript for intellectual content. All authors critically revised the manuscript and agree to be fully accountable for the integrity and accuracy of the work. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
This research was approved by the Ethical Committee of the Affiliated Hospital of Zunyi Medical University, People’s Republic of China, on January 21, 2021. Written informed consent was obtained from the patient to undergo the procedures described in this report and for its publication. The report was compiled in accordance with the CARE guidelines (![]() ).
).
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Regional Project of the National Natural Science Foundation of China (grant number 81969105), the Doctoral Research Foundation of Zunyi Medical University (grant number ZC220200604), and the Science and Technology Program of Guizhou Province (grant number [2020]1Y301).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
