Abstract
Odontogenic carcinosarcoma is a very rare malignant odontogenic tumor, characterized by malignant epithelial and mesenchymal components. Studies have reported several cases of odontogenic carcinosarcoma, mainly in the upper and lower jaws, with malignant clinical manifestations. Herein, we present the case of a 58-year-old woman with odontogenic carcinosarcoma with dentinoid in the left maxilla. The invasion range was large, and the left maxillary molar was missing. Histology revealed odontogenic carcinosarcoma with bidirectional differentiation characteristics and comprising three components: malignant epithelium, malignant interstitium, and dentinoid. The patient subsequently underwent nasal endoscopic sinus tumor resection, and she recovered well after surgery. After a strict 4-year follow-up, to date, there are still no signs of disease or local recurrence. To our knowledge, this is the first reported case of odontogenic carcinosarcoma with dentinoid. Our study describes the clinical, morphological, and immunohistochemical characteristics of this case, and distinguishes it from related diseases.
Keywords
Introduction
In accordance with the 2017 World Health Organization (WHO) classification of head and neck tumors, odontogenic carcinosarcoma is a malignant biphasic tumor with malignant epithelium and mesenchyme. 1 Its etiology is not yet clear, but it is believed that odontogenic carcinosarcoma originates from primary diseases, such as ameloblastoma, ameloblast fibroma, ameloblast fibrosarcoma, and osteosarcoma. 2 Moreover, odontogenic carcinosarcoma has also been reported as primary odontogenic carcinosarcoma. Because of the nontypical clinical manifestations and the scarcity of cases, it is difficult to diagnose odontogenic carcinosarcoma. Thus, it is necessary to make a diagnosis through histopathology and to distinguish it from other related diseases.
To date, there have been only 11 cases of odontogenic carcinosarcoma reported in the literature. 3 Our case is the first case of odontogenic carcinosarcoma with dentinoid. This study aimed to explore the origin and histopathological characteristics of odontogenic carcinosarcoma and to improve the understanding of this rare disease.
Case presentation
A 58-year-old female patient was admitted to the hospital in March 2016 because of headache and tearing for more than 10 days, and a swollen left eye. The patient developed a headache after a cold more than 10 days earlier, and she experienced nausea without vomiting. One week earlier, she was seen at the neurology department of our hospital and underwent head computed tomography (CT), which showed bilateral frontal ethmoid sinus inflammation, nasal polyps, and a space-occupying lesion. Head CT also showed no abnormalities in the brain. She was referred to a nasal surgeon for three-dimensional CT examination of the sinuses, which revealed that the left paranasal sinus middle turbinate (MT) invaded the orbit. The maxilla, inner wall of the orbit, ethmoid plate, and nasal septum were involved, and the bone was damaged. There were also inflammatory signs in bilateral frontal sinuses, and in the ethmoid sinus and maxillary sinus (Figure 1a–d).

Computed tomography (CT) examination results. (a, b (arrows)) CT indicated inflammation in the maxillary sinus, bone destruction, and a space-occupying soft tissue lesion. (c, d (arrows)) The shape of the left inferior turbinate had disappeared, and the turbinate was destroyed. The tumor invaded the contralateral turbinate, the uncinate process was compressed but not damaged, and the ethmoid sinus was involved. The left lateral orbital wall was completely destroyed, and the frontal sinus was involved.
The patient suffered from a decreased sense of smell, numbness of the left side of her face, and tearing from both eyes. The left eye was swollen, and she had no fever. She reported a dry nose; occasional nosebleeds; no itchy, stuffy, or runny nose; and no paroxysmal sneezing. Furthermore, she had no dizziness and fainting, no diplopia or vision loss, no tinnitus and hearing loss, no mouth opening restriction, and normal diet and sleep, normal stools, and no obvious changes in physical strength and weight. In 2014, she underwent gingival tumor resection. On 17 March 2016, she underwent nasal endoscopic sinus tumor resection. The details of the surgical intervention are as follows: The patient was placed in the supine position. After induction of general anesthesia and oral intubation, routine disinfection and draping was performed, and the nasal mucosa was contracted with a diluted adrenaline-soaked gauze. Nasal endoscopy showed that most of the nasal septum was missing; only the front nasal columella remained, and bilateral nasal cavities merged into one cavity. The nasal cavity contained new gray-white lesions, and the surface was not smooth. Bipolar coagulation was used to coagulate the edge of the tumor, and some of the new lesions were clamped and sent for intraoperative frozen section. The coagulation system was used from front to back to remove tumor residue to the root. The scope of the tumor was as follows: the anterior boundary reached the anterior wall of the maxillary sinus, the posterior boundary reached the pterygopalatine fossa on the posterior wall of the maxillary sinus, the lateral border reached the outer wall of the maxillary sinus, and the lower boundary reached the maxillary alveolar cavity. The lesion involved the junction of the ethmoid sinus and the anterior skull base. Owing to severe bleeding, the tumor was removed to the base of the anterior skull, and bipolar coagulation was repeated to stop the bleeding. The lesions were removed satisfactorily, and iodoform gauze was placed in the left nasal cavity. The patient recovered well after surgery. After a strict follow-up to 2020, there are still no signs of disease or local recurrence. This study complied with the CARE reporting guidelines. 4
Histology/microscopic findings
The pathological examination results were as follows: hematoxylin and eosin (H&E)-stained sections showed that the tumor grew aggressively and consisted of epithelial and mesenchymal components, with a large amount of eosinophilic dentin-like substance (Figure 2a). The tumor epithelium was nested, flaky, and cord-like, with densely arranged cells. Some cells exhibited the characteristics of odontogenic cells, similar to ameloblastoma cells. Furthermore, some areas showed distribution similar to ameloblastic distribution, but the cells were malignant. The tumor was characterized by cell polymorphism, deep staining of the nucleus, obvious atypia, nucleocytoplasmic imbalance, and pathological mitoses (Figure 2a, b). Abundant dentinoid could be seen between epithelial cells, as eosinophilic dentin-like calcifications, and the cells were distributed in the form of sheets and nets. Interstitial cells were spindle-shaped, with malignant appearance. Additionally, cell polymorphism, differing cell size, large darkly-stained nuclei, nucleus-plasma ratio imbalance, nuclear atypia, and nuclear division were common, as shown in Figure 2c–f.

Hematoxylin and eosin (H&E) staining results. (a, b) The tumor was composed of epithelium, interstitium, and a mass comprising reticular and lamellar hard tissues; magnification: ×100. (c) Cells were columnar and characterized by odontogenic cells; magnification: ×100. (d) Interstitial spindle cells, dentin-like substance, and epithelial cells similar to odontogenic cells are seen; magnification: ×200. (e) Glaze-like structure and hard tissues; magnification: ×100. (f) A large number of cells with obvious atypia can been seen both in epithelial and mesenchymal cells, with deep nuclear staining and pathological mitosis (arrow); magnification: ×200.
Immunohistochemical analysis
The results of immunohistochemistry showed that androgen receptor (AR) (Figure 3a), β-catenin (Figure 3b), and B cell CLL/lymphoma-2 (Bcl-2) (Figure 3c) expression was strongly positive in both epithelial cells and interstitial spindle cells. β-catenin (Figure 3d) and Bcl-2 (Figure 3e) expression was strongly positive in cells surrounding the dentin-like substance. Bcl-2 also showed strong positivity around the papillary tissue. Cytokeratin (CK), as a specific marker of epithelial cells, showed strong positive expression in odontogenic epithelial islands and striata (Figure 3f). CK5/6 was positively expressed in the odontogenic epithelium and negatively expressed in stromal tissue (Figure 4a). CK7 (Figure 4b) and CK8/18 (Figure 4c) were expressed only in the epithelium, whereas CK7 was weakly positive, and CK8/18 was positively expressed in cells around the dentin-like substance. CK19 expression was positive in epithelial cells and negative in interstitial cells (Figure 4d). E-cadherin (Figure 4e) and vimentin (Figure 4f) were both expressed in the epithelium and stroma, showing the characteristics of the tumor’s bidirectional differentiation. Epithelial growth factor receptor (EGFR) (Figure 5a) and transforming growth factor β1 (TGF-β1) (Figure 5b) were positive around the epithelial, interstitial, and dentin-like substances. Moreover, estrogen receptor (ER) expression was positive in epithelial and interstitial cells (Figure 5c), p53 was positive in interstitial cells (Figure 5d), and p63 was positive in the epithelium (Figure 5e). Ki67 expression was positive in both epithelial and interstitial cells, with a nuclear positivity rate of 75% in epithelial cells and 10% in interstitial spindle cells, indicating that the cells were undergoing active proliferation (Figure 5f).

Immunohistochemistry surfactant protein (SP) results. (a) Positive androgen receptor (AR) expression in tumor stromal and epithelial cells; magnification: ×100. (b) Strongly positive β-catenin expression in tumor stromal and epithelial cells; magnification: ×100. (c) Strongly positive Bcl-2 expression in epithelial and interstitial spindle cells; magnification: ×200. (d) Positive β-catenin expression in cells surrounding the dentin-like substance; magnification: ×200. (e) Positive Bcl-2 expression in cells surrounding the dentin-like substance; magnification: ×400. (f) Positive expression of CK in odontogenic epithelial cells; magnification: ×200.

Immunohistochemistry surfactant protein (SP) results. (a) Positive cytokeratin (CK)5/6 expression in tumor epithelial cells and negative expression in interstitial cells; magnification: ×200. (b) CK7 expression was only weakly positive in epithelial cells and negative in interstitial cells; magnification: ×200. (c) CK8/18 was expressed in epithelial cells and in the cells around the dentin-like substance; magnification: ×200. (d) CK19 expression was positive in epithelial cells and negative in interstitial cells; magnification: ×200. (e) E-cadherin was expressed in both epithelial and stromal cells; magnification: ×200. (f) Vimentin was expressed in both epithelial and stromal cells; magnification: ×200.
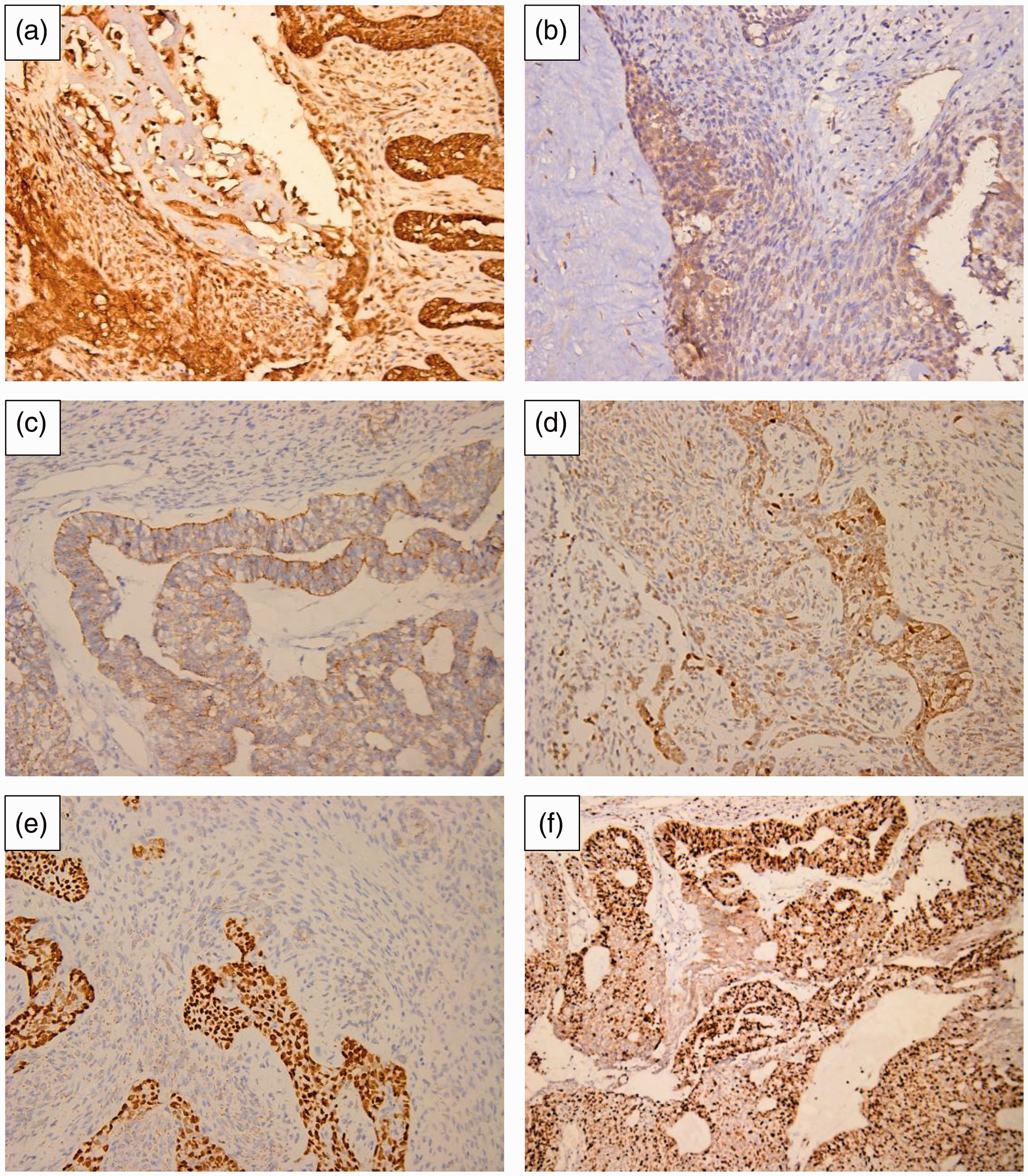
Immunohistochemistry surfactant protein (SP) results. Positive epithelial growth factor receptor (EGFR) (a) and transforming growth factor β1 (TGF-β1) (b) expression in epithelial and interstitial cells and around the dentin-like substance; magnification: ×200. (c) Estrogen receptor (ER) expression was positive in the epithelial cells and negative in the interstitial cells; magnification: ×200. (d) Weak positive expression of p53 in interstitial cells; magnification: ×200. (e) p63 expression was positive in epithelial cells and negative in interstitial cells; magnification: ×200. (f) Ki67 was positive in both epithelial and interstitial cells; magnification: ×200.
Discussion
In 1991, Tanaka and colleagues described odontogenic carcinosarcoma for the first time.4,5 To date, only 12 cases, including our case, have been reported worldwide. In this study, we reported the first case of odontogenic carcinosarcoma with dentinoid, which occurred in the left maxilla. The invasion range was large, and the left maxillary molar was missing. The odontogenic carcinosarcoma contained dentin-like substance, and the epithelium and interstitium were malignant. Previous studies have reported ameloblastoma accompanied by dentinoid formation.5–7 Currently, the etiology of odontogenic carcinosarcoma is unclear. According to previous reports, only two cases were neonatal lesions, while nine cases may have originated from primary diseases, such as ameloblastoma. However, the mechanism of transformation into malignancy is unclear; most are believed to be related to multiple relapses and the absence of adjuvant therapy after surgery. In addition, some ameloblastomas have a long disease course, which may also be a risk factor for malignant transformation to odontogenic carcinosarcoma. Therefore, we inferred that the odontogenic carcinosarcoma in this case may have derived from ameloblastoma with dentinoid.
Dentinoid is defined as a collagen tissue, generally non-mineralized and similar to dentin; however, dentinoid does not contain tubules and does not meet the dentin standard for dentin tubules. The odontogenic epithelium has a close anatomical relationship.7,8 The reason for dentinoid formation is not yet clear. Some scholars believe that dentinoid in odontogenic tumors is a hamartoma or is caused by inducible stimulation caused by proliferating epithelium on mesenchymal tissue.8,9 Papagerakis and colleagues demonstrated that mixed odontogenic tumors normally express the gene products appearing in ectodermal mesenchymal cells, resulting in the transformation and co-expression of epithelial to mesenchymal phenotypes. 10 Therefore, some tumor cells that should differentiate into enamel epithelium may produce dentinoid in some tumors.
Odontogenic carcinosarcoma should be differentiated from related tumors, namely ameloblastoma and ameloblastic fibroma. Odontogenic carcinosarcoma is a mixed tumor with malignant epithelium and mesenchyme. 2 In contrast, ameloblastoma is a benign epithelial odontogenic tumor, characterized by local invasion. The periphery of the tumor nest comprises cuboidal or columnar cells, the nuclei are arranged in a picket fence pattern and away from the basement membrane, and the center is a polygonal cell similar to star-shaped reticular cells. The epithelium of ameloblastic fibroma appears as strips or clumps, and can also appear as a structure similar to a dental plate.9–11 The peripheral layer comprises cuboidal or columnar cells, and the central cells are similar to star-shaped reticular cells. Compared with ameloblastoma, the number of astrocytes in ameloblastic fibroma is very low, and the interstitial connective tissues are naive and rich in cells, similar to tooth papilla cells of tooth germs. The fourth edition of the WHO head and neck tumor classification clearly stated that carcinosarcoma should be distinguished from ameloblastoma accompanied by malignant spindle cells because it may be related to epithelial mesenchymal transformation and sarcomatoid ameloblastoma. 1 Kawauchi and colleagues reported a case of spindle-cell ameloblastic carcinoma.10–12 The tumor contained spindle cells and had a sarcomatous appearance, but sarcomatoid spindle cells were identified as epithelial cells, and spindle cells in odontogenic carcinosarcoma were interstitial components by ultramicroscopy and immunohistochemistry. Thus, immunohistochemical methods are an effective method of identifying cell types.
The immunohistochemistry results in this case indicated that the odontogenic carcinosarcoma had bidirectional differentiation characteristics. CK, a marker of epithelial cell differentiation, was positively expressed in the tumor epithelium and negatively expressed in the mesenchyme in this case, suggesting that the spindle cells were not of epithelial origin but of interstitial origin. During the differentiation of tooth germ cells into ameloblasts, CK19 mainly expresses positively in the late stage.11–13 Moreover, CK19 is expressed in most odontogenic cysts and benign tumors, which can be used as a marker for odontogenic tumors.12–14 The positive expression of CK19 in the tumor epithelium in this study supports an odontogenic origin, and was also used to identify the nature of the spindle cells, which provides evidence for the diagnosis of odontogenic carcinosarcoma.13–15 Some studies have found that CK8 and/or CK18 expression is more common in head and neck malignant tumors, especially in poorly differentiated tumors, which is consistent with this case.14–16 The strong positive Ki-67 and Bcl-2 expression indicated that both epithelial and mesenchymal cells proliferated actively. EGFR can regulate the migration and invasion of human ameloblastoma cells and the secretion of matrix metalloproteinases, and promote the local invasion of ameloblastoma. 17 In this case, EGFR was expressed in the epithelium and mesenchyme, indicating that the tumor had a strong ability to proliferate and invade. Furthermore, the positive p53 expression in the epithelium and mesenchyme in this case indicated that the tumor had malignant cytological features and active cell proliferation. Positively p63 expression in tumor epithelial cells suggested that the epithelial cells had two malignant manifestations of low differentiation and high growth potential.15–18 The mesenchymal marker, vimentin was expressed in both mesenchymal and epithelial cells, and the positive E-cadherin expression in interstitial cells was low, indicating that the adhesion capacity of interstitial spindle cells was reduced. Additionally, the presence of epithelial cells in the tumor indicated an interstitial transformation phenomenon.16–19 In this case, β-catenin was positively expressed in epithelial and mesenchymal cells, which was mainly located in the cytoplasm. β-catenin may participate in the classical cell adhesion and Wnt signaling pathways.17–20 Studies have shown that β-catenin may be related to the invasiveness of odontogenic calcified cysts and ameloblastomas.18–21 The TGF-β/Smad signaling pathway is activated in a variety of odontogenic tumors, such as ameloblastoma.19–22 Inactivation of the TGF-β/Smad pathway may be related to aggressive biological behavior, especially in ameloblastoma, which may increase cell proliferation and reduce apoptosis and differentiation.19–22
We reported the first case of malignant odontogenic carcinosarcoma with dentinoid. The diagnosis of odontogenic carcinosarcoma was achieved through histopathology and immunohistochemistry. To date, there are few reported cases of odontogenic carcinosarcoma, and the diagnosis is difficult. More cases are needed to explore the pathogenesis and histopathological characteristics of odontogenic carcinosarcoma.
Conclusions
To our knowledge, we reported the first case of odontogenic carcinosarcoma with dentinoid. However, it is difficult to diagnose odontogenic carcinosarcoma. Therefore, it is necessary to make a diagnosis by combining histopathology and immunohistochemistry and to distinguish this carcinosarcoma from other related diseases.
Footnotes
Availability of data and material
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of the data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
This study was approved by the Ethics committee of the School and Hospital of Stomatology, China Medical University (approval number: 2016012). All procedures performed in this study involving human participants were in accordance with the Helsinki Declaration (as revised in 2013). We obtained written patient consent for treatment, and the patient provided written informed consent for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (81072197 and 81470758).
