Abstract
Single-photon emission computed tomography (SPECT)/computed tomography (CT) imaging of the gouty spine is rare. We describe a 66-year-old man who presented with pain and numbness in the right lower leg; he reported a 2-month history of intermittent low back pain. Imaging revealed neoplastic lesions of the spine, which were initially regarded as tumors. Thus, the patient underwent surgical removal of the lumbar lesion. However, the postoperative pathological diagnosis was gout spondylitis. In this report, we show multimodal images of advanced gout spondylitis. The metabolic information provided by SPECT/CT, combined with the microscopic changes in bone structure revealed by dual-source thin-layer CT and the anatomical localization information provided by magnetic resonance imaging, can help clinicians to more fully understand the pathophysiological mechanisms and imaging manifestations of gout from multiple perspectives, thereby reducing the rate of misdiagnosis.
Keywords
Introduction
Metabolism-related gouty arthritis results from the deposition of monosodium urate crystals in articular and surrounding tissues because of renal and intestinal under-excretion. Current diagnostic guidelines for gout1,2 recommend the use of ultrasound and dual-energy computed tomography (CT) imaging for the assessment of patients with gout arthritis. To our knowledge, there have been few reports concerning the use of [99mTc]-methylene diphosphonate (MDP) single-photon emission computed tomography (SPECT)/CT imaging to assess spinal tophi.3–6 Here, we discuss the usefulness of [99mTc]-MDP SPECT/CT imaging in the diagnosis of gout spondylitis.
Case report
A 66-year-old man presented with pain and numbness in the right lower leg. He reported a 2-month history of intermittent low back pain that worsened with activity, and which was accompanied by fatigue and walking instability. Twenty-one years prior to this visit, the patient had been diagnosed with gouty arthritis, for which he had received occasional treatment (e.g., resection of tophi on the right first toe 4 years prior to this visit). Physical examination revealed tenderness in the L4 to L5 spinous process and paraspinal region, which radiated to the right lower limb; pain was observed in the right foot. Skin sensation was reduced in the medial region of the right thigh and below the popliteal fossa. The patient was admitted to the hospital to undergo further assessment of a suspected lumbar disc herniation.
Laboratory tests revealed a serum uric acid level of 663 μmol/L (normal: 210–420 μmol/L), a serum creatinine level of 162 μmol/L (normal: 57–111 μmol/L), a serum C-reactive protein level of 79.91 mg/L (normal: 0–5 mg/L), and an erythrocyte sedimentation rate of 81 mm/hour (normal: 0–15 mm/hour).
The patient also underwent imaging examinations, including X-ray, CT, magnetic resonance imaging (MRI), and [99mTc]-MDP SPECT/CT. X-ray examination revealed degenerative changes in the lumbar vertebra (Figure 1b) and bilateral soft tissue swelling of the knee joint (Figure 1a), along with bone destruction of the articular surface. CT revealed punched-out bone erosion with cystiform changes in the bilateral knee joints, as well as marginal sclerosis and multiple nodules of increased density in the joint cavity and surrounding soft tissue (Figure 1d); these features supported a diagnosis of gouty arthritis.
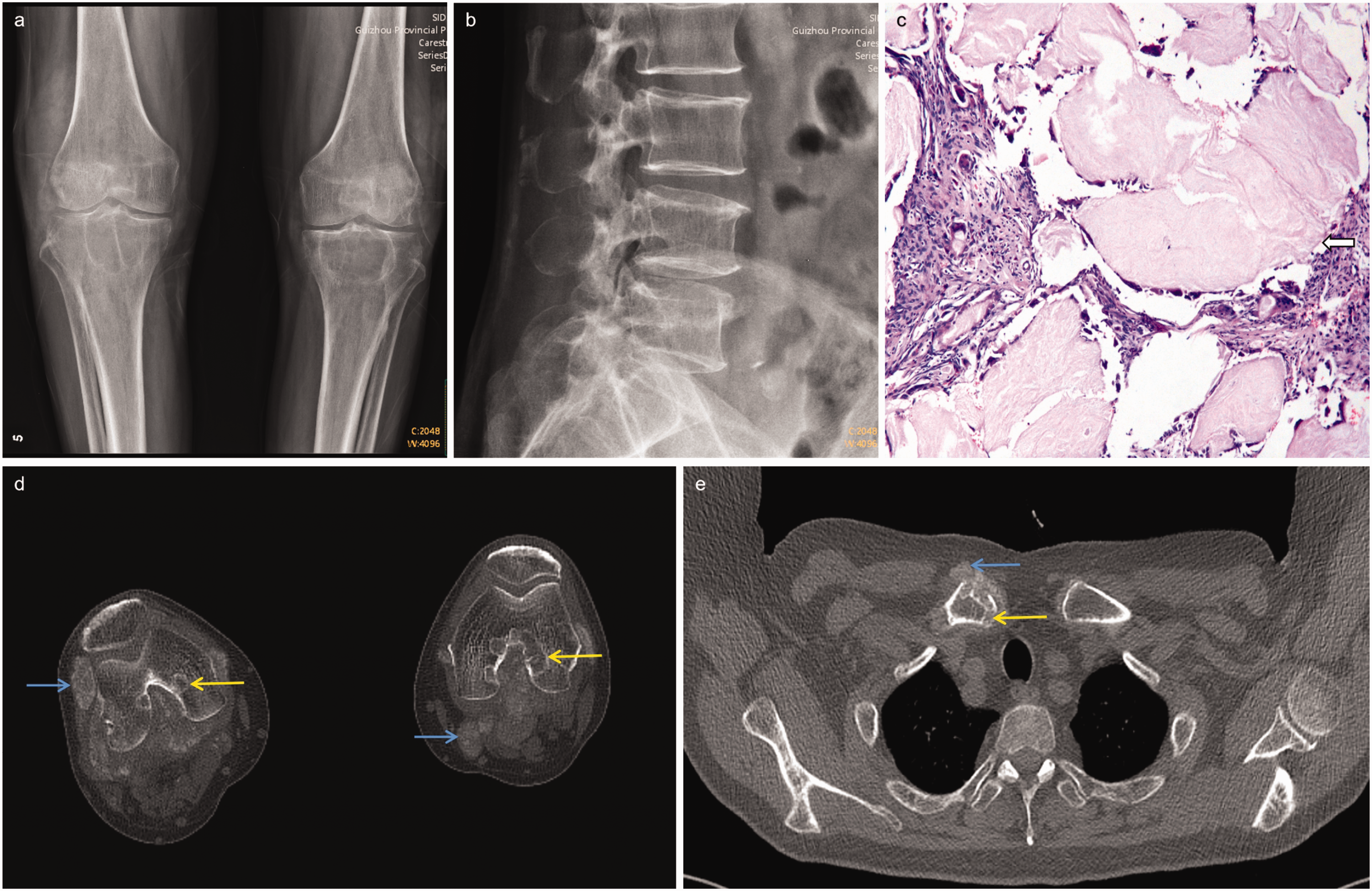
Findings of X-ray imaging and histological examinations. X-ray images showing oncotic, multiple density reduction shadows of the joint surface in the bilateral knee (a) and osteophytes at the edge of the lumbar spine (b). Microscopic histological examination (c) of a sample shows in panels a and b revealed the presence of gouty material (white arrow) (hematoxylin and eosin, ×200). Computed tomography image of the knee joint (d) with window settings appropriate for bone shows multiple punched-out erosion in the bilateral upper tibia (yellow arrows), and multiple nodules of increased density in the surrounding soft tissue (blue arrows) and Computed tomography of the chest (e) with window settings appropriate for bone shows multiple sites of punched-out erosion in the sternoclavicular joint (yellow arrow) and multiple nodules of increased density in the surrounding soft tissue (blue arrow).
MRI revealed several neoplastic lesions, including a nodule in the L1 vertebra (Figure 2), an epidural tumor at L1/L2 causing coni medullaris compression of the spinal canal, and hypointensity of the zygapophyseal joints on the right side of the L1 to L3 vertebra. All of these features were significantly enhanced after the administration of gadolinium contrast agent.

Findings of magnetic resonance imaging examination. Magnetic resonance images of the lumbar spine (a and b) show an intra-vertebral nodule at L1 (white arrow in a) and an intraspinal canal nodule located posterior to the spinal cord (white arrow in b), resulting in lumbar spinal stenosis at L1/L2 (white triangles in a and b). Both nodules show low signal intensity on T1 (a)- and T2 (b)-weighted images and Contrast-enhanced magnetic resonance images (c–g) show a significant increase in signal intensity of inter-vertebral nodule at L1 (white arrow in c), of the intraspinal canal nodule at L1/L2 (white triangle in d), and of bone erosion in the right interapophyseal joints at L1 to L3 (e–g, yellow arrows).
The patient exhibited multiple regions of anomalous uptake of [99mTc]-MDP, including the right acromioclavicular joint, lumbar spine, knees, and ankles (Figure 3a). In the lumbar region, there was evidence of a neoplastic lesion in the L1 vertebra, an intraspinal canal nodule at L1/L2, and anomalous uptake in the facet joints of the right L1 to L3 vertebra (Figure 3b–e).

Findings of 99mTc-methylene diphosphonate single-photon emission computed tomography/computed tomography examination. Anterior and posterior whole-body views of 99mTc-methylene diphosphonate skeletal scintigraphy (a). Images in (b) through (e) depict the lumbar spine region: columns (1) and (2) show computed tomography and single-photon emission computed tomography images, respectively, whereas column (3) shows merged images. Focal anomalous uptake of 99mTc-methylene diphosphonate is evident in an intraspinal canal nodule at L1/L2 (white triangles in d) and bone erosions are evident in the right interapophyseal joints at L1/L2 (white arrows in b and e) and L2/L3 (blue arrows in c and e).
Based on the imaging findings, the neoplastic lesions of the spine were initially regarded as tumors. Thus, the patient underwent surgical removal of the lumbar lesion. Postoperative pathological diagnosis of zygapophyseal joints on the right side of the L1 to L3 vertebra (Figure 1c) revealed additional gout nodules in fiber, fat, and bone tissue. The patient exhibited considerable symptomatic improvement after surgery.
This case report was written in accordance with the CARE guidelines. 7
Discussion
Gout is a systemic disorder of uric acid metabolism, and a high serum urate concentration is the most important risk factor for the development of gout. The course of gout has been classified into four stages: asymptomatic hyperuricemia, acute gouty attack, inter-critical period, and chronic tophaceous gout. 8
The main symptoms of gout spondylitis are pain caused by spinal cord or nerve root compression; these types of pain may be easily misdiagnosed or ignored because of their similarity to pain caused by disc herniation. Additionally, gout is associated with conditions such as hypertension, obesity, cardiovascular disease, diabetes mellitus, dyslipidemia, chronic kidney disease, and kidney stones; these associations can complicate the treatment of gout and lead to premature death.9–11
The diagnosis of gout is usually based on the results of clinical examination, laboratory tests, and imaging. With regard to the standard for gout diagnosis, the American College of Rheumatology and the European League Against Rheumatism recommend microscopic confirmation of monosodium urate crystals in synovial fluid or tophi as the gold standard for gout diagnosis. 1 Furthermore, ultrasound imaging and dual-energy CT are the main imaging techniques recommended for evaluation of gout.2,8
In most patients with gout, punched-out erosion is evident when a bone and its surrounding joints are eroded by tophi. Another characteristic feature is the presence of a so-called “overhanging edge.” In an X-ray of the patient’s lumbar spine, there was no characteristic sign of bone destruction; only degeneration of the lumbar spine was observed. However, an X-ray of the patient’s knee joint showed a more typical form of tophi-related erosion (Figure 1d). CT images are superior to X-rays in terms of evaluating the spine and sacroiliac joints; CT images can also be used to measure tophi density. In CT images of the patient’s lumbar region, erosion of the interapophyseal joints was evident, along with increased density in a portion of the vertebra (Figure 2).
MRI is the preferred imaging modality for evaluation of the spinal canal. Gout in the spinal canal is generally rare and difficult to diagnose because its clinical presentation and imaging findings are similar to the characteristics of tumors, abscesses, tuberculosis, and degenerative spinal diseases; moreover, gouty tophi show various patterns. It has been reported 12 that most spinal tophi are hypointense on both T1- and T2-weighted images; additionally, gadolinium enhancement occurs because of vascularized reactive granulation. Although the MRI appearance of tophi is non-specific, the diagnosis of gout should be considered when a mass exhibits heterogeneous low to intermediate signal intensity on T2-weighted images, particularly if the mass is linked to erosion of adjacent bones. In the patient, the tophi were hypointense on both T1- and T2-weighted magnetic resonance images; administration of gadolinium contrast agent revealed the enhancement of tophi and soft tissues adjacent to the facet joints.
Nuclear medicine is rarely used to evaluate patients with gout. However, skeletal scintigraphy using [99mTc] phosphate compounds may be helpful in determining the extent, severity, and distribution of gouty arthritis,13,14 particularly when assessing injury to osteogenic joint structures and visualizing the behavior of inflammatory factors. The amount of [99mTc]-MDP uptake provides information concerning local blood flow and bone metabolism in the joint; corresponding CT images allow the assessment of bone destruction in the gouty lesion area. To our knowledge, only a few case reports have described the use of SPECT/CT to evaluate gout tophi.3–6 In these reports, anomalous increases in [99mTc]-MDP were reported in tophi; these increases were presumably the result of inflammation, which led to greater blood supply in the region. Furthermore, tophi can cause bone erosion and remodeling. Hydroxyapatite crystal deposition can also occur, and the crystal surface can adsorb a large amount of [99mTc]-MDP. This increase is non-specific because it can also occur in patients with infections or neoplastic lesions. However, these signs provide clinical indications of an early inflammatory response to gout.
The gout classification criteria established by the American College of Rheumatology and the European League Against Rheumatism 1 initially did not consider the use of imaging. However, after a systematic review of the literature, imaging was included as an evaluation parameter for those criteria. Ultrasound and color-coded post-processed dual-energy CT images can be used to provide imaging evidence of urate deposition. Furthermore, X-ray imaging of the hands and/or feet that reveals at least one region of erosion is regarded as imaging evidence of potential gout-related joint damage. Subsequently, current classification criteria do not consider the use of advanced molecular imaging techniques. SPECT/CT combines the advantages of two imaging modalities: functional information from SPECT and precise morphological information from CT. Advances in hardware, algorithms for image reconstruction, and techniques for correcting photon attenuation and scattering have led to very accurate quantification using SPECT/CT.15,16 Furthermore, quantitative bone SPECT/CT can potentially be used for prognostic assessment and differential diagnosis of patients with various bone and joint disorders. 17 When patients receive a confirmatory diagnosis of gout, SPECT/CT is recommended to monitor the extent of bone and joint structural damage, as well as local inflammation; this monitoring approach allows clinicians to develop personalized treatment strategies for patients. Furthermore, because urate is filtered and excreted by the glomerulus, an evaluation of glomerular filtration function could potentially be added to nuclear medicine imaging protocols for the management of patients with gout. 18
In this report, we have described the multimodal imaging of advanced gout spondylitis. We recommend SPECT/CT to monitor the extent of bone and joint structural damage, as well as local inflammation. This approach will enable clinicians to evaluate treatment efficacy and develop personalized treatment strategies for their patients.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221129557 - Supplemental material for 99mTc-methylene diphosphonate SPECT/CT imaging of gout spondylitis: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221129557 for 99mTc-methylene diphosphonate SPECT/CT imaging of gout spondylitis: a case report by Yan Wang, Yan Zha, Rongfeng She, Gaofeng Sun, Shuang Li, Lin Liu, Neil Roberts, Yaying Li in Journal of International Medical Research
Footnotes
Author contributions
YW reviewed the literature and wrote the manuscript; YZ provided professional guidance as an expert in renal rheumatology immunology; RS provided this case; GS provided professional guidance concerning nuclear medicine; SL provided professional guidance regarding pathology; LL provided professional guidance; NR provided professional guidance regarding Imaging and revised the language; and YL reviewed and edited the manuscript. All authors have reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
Ethical approval was not required for this case report because no intervention or changes were made to the clinical course of events. However, the patient provided written informed consent for the use of his data for scientific purposes.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Guizhou Provincial Science and Technology Projects, Qian Ke He Foundation-ZK [2022] General 253; Doctor Foundation of Guizhou Provincial People’s Hospital (GZSYBS[2019]02); and the Science and Technology Fund Projects of Guizhou Health Commission (gzwkj2023-210).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
