Abstract
Primary central nervous system lymphoma (PCNSL) is a rare form of non-Hodgkin lymphoma, but its diagnosis is challenging in some cases. A brain biopsy is the gold standard for diagnosing PCNSL, but its invasiveness can be problematic. Thus, noninvasive imaging examinations have been developed for the pre-surgical diagnosis of PCNSL, including gadolinium-enhanced magnetic resonance imaging (MRI), 123I-N-isopropyl-p-iodoamphetamine single-photon emission computed tomography (123I-IMP SPECT), and positron emission tomography with 18F-fluorodeoxyglucose (18F-FDG PET). Here, we report the case of a 71-year-old woman with negative imaging findings for PCNSL, but who was diagnosed with PCNSL by a brain biopsy and histological analysis. Her imaging results were negative for gadolinium-enhanced cranial MRI, with low uptake in 123I-IMP SPECT and hypometabolism in 18F-FDG PET. However, a stereotactic brain biopsy from an abnormal lesion revealed that many round cells had infiltrated into the brain. Moreover, many infiltrating cells were positive for cluster of differentiation (CD)20 and CD79a, and proliferation marker protein Ki-67-positive cells accounted for nearly 80% of all cells. Based on these results, our final pathological diagnosis was PCNSL. The present case highlights the possibility of a PCNSL diagnosis even when all imaging-related examinations display negative results.
Keywords
Introduction
Primary central nervous system lymphoma (PCNSL) is a rare form of non-Hodgkin lymphoma that represents 4% of intracranial neoplasm cases, 1 , 2 although the incidence rate increases with age. 3 Unlike other primary brain tumors, PCNSL responds favorably to chemotherapy and radiation therapy, and its treatment has progressed in recent years. 4 Thus, the diagnosis of PCNSL is important, but can be challenging in some cases. Pre-surgical diagnosis of PCNSL is important for its adaptation to a brain biopsy, and gadolinium-enhanced cranial magnetic resonance imaging (MRI) is a useful modality that shows ≥95% sensitivity in the diagnosis of PCNSL. 5 , 6 However, in the current report, we present the case of an elderly woman with negative imaging for PCNSL who was nevertheless diagnosed with this disease following a brain biopsy and histological analysis. This atypical case indicates that there is a small risk that PCNSL might evade detection using current imaging techniques without a brain biopsy.
Case report
A 71-year-old woman developed subacute dementia. She had medically treated hypertension with amlodipine besylate (2.5 mg/day) but no other medical history. She had received no immunosuppression or corticosteroid therapy. Three months later, she had difficulties walking by herself. She was admitted to a local hospital 4 months after her symptoms appeared, and her fluid-attenuated inversion recovery (FLAIR) MRI images revealed subtle high intensities around the lateral ventricles (Figure 1a–c). She was admitted to our hospital for further examination 5 months after her symptoms had appeared. Upon admission, her cognitive function was in decline, with a Mini-Mental State Examination score of 17/30, a revised Hasegawa Dementia Rating Scale score of 16/30, and a Frontal Assessment Battery score of 13/18. Hematological and biochemical analyses were unremarkable, including 197 U/L lactate dehydrogenase and 170 U/mL soluble interleukin-2 receptor. A cerebrospinal fluid (CSF) analysis revealed an elevated cell count (22/μL; 100% mononuclear cells) and an elevated protein level (41 mg/dL). CSF cytology, CSF flow cytometry, a skin biopsy, and a gastric biopsy demonstrated no malignancies. The abnormally high intensity lesions, observed using cranial FLAIR MRI, expanded within 1 month (Figure 1d–f, arrows). An ophthalmologic examination indicated that the patient’s vision was preserved at 20/25 (right) and 20/25 (left), and her visual field was normal. The fundus oculi, specular microscopy, and critical fusion frequency results showed no abnormalities.

Clinical images taken before the brain biopsy. (a–c) Axial fluid-attenuated inversion recovery (FLAIR) images 4 months after the appearance of symptoms from the levels of the patient’s (a) fourth ventricle, (b) cerebral peduncle, and (c) lateral ventricle. (d–f) Axial FLAIR images 5 months after the appearance of symptoms. Abnormally high intensity areas had expanded to the left superior cerebellar peduncle, bilateral cerebral peduncles, left hippocampus, and white matter around the lateral ventricle (arrows). (g–i) Diffusion-weighted images also showed a high intensity area around the lateral ventricle (arrows). (j–l) Gadolinium-enhanced magnetic resonance imaging showed no enhancement at the lesions. (m) 123I-N-isopropyl-p-iodoamphetamine single-photon emission computed tomography showed decreased cerebral blood flow at the lesions (arrowheads). (n, o) Positron emission tomography with 18F-fluorodeoxyglucose also showed hypometabolism at the lesions (square areas)
In an attempt to diagnose the patient’s condition, we performed several imaging examinations. Although some lesions showed hyperintensity in diffusion-weighted images (Figure 1g–i), gadolinium-enhanced MRI revealed no enhancement (Figure 1j–l). 123I-N-isopropyl-p-iodoamphetamine single-photon emission computed tomography (123I-IMP SPECT) demonstrated decreased cerebral blood flow in abnormal lesions (Figure 1m, arrowheads). Furthermore, positron emission tomography with 18F-fluorodeoxyglucose (18F-FDG PET) revealed hypometabolism in the same cranial lesions (Figure 1n, o, square areas), although there was no abnormal accumulation in the extracranial cavity. Spinal cord MRI also showed no abnormalities. Based on these results, we suspected a non-tumorigenic but inflammatory disease.
At this stage, the patient’s consciousness worsened to somnolence and rapid treatment was sought. We performed a stereotactic brain biopsy from an abnormal lesion of the left portion of the corpus callosum (Figure 2a, arrow) at 37 days after admission because the treatments required for tumorigenic diseases (such as PCNSL) and non-tumorigenic diseases are very different. Hematoxylin and eosin staining of the biopsy sample showed many round cells, similar to large lymphoblasts, that had infiltrated into the brain (Figure 2b, c). Although marked demyelination is characteristic of progressive multifocal leukoencephalopathy, no demyelination lesions were observed with Klüver–Barrera staining (Figure 2d). Immunohistochemical staining and polymerase chain reaction of the human polyomavirus 2 (JC virus) from brain tissue were also negative. Immunohistochemical staining revealed the following characteristics of the infiltrating cells. A few cells were positive for the T cell markers cluster of differentiation (CD)3 (Figure 2e) and CD5 (Figure 2f). 7 No cells were positive for the precursor lymphoid cell marker CD10 (Figure 2g). In contrast, many infiltrating cells were positive for the B cell markers CD20 (Figure 2h) and CD79a (Figure 2i). 8 , 9 No cells were positive for B-cell lymphoma 6 (Bcl-6; Figure 2j), 10 a germinal center B cell marker, or for Epstein–Barr virus-encoded RNA 1 (EBER1; Figure 2k), a marker of some kinds of malignant lymphomas (such as Hodgkin, NK/T cell, and Burkitt lymphomas). 11 Proliferation marker protein Ki-67-positive cells accounted for nearly 80% of all cells (Figure 2l), suggesting malignancy. 12 Neither glial fibrillary acidic protein (GFAP)-positive cells, suggesting glioblastoma 13 (Figure 2m), nor S-100 protein-positive cells, suggesting neurogenic tumors 14 (Figure 2n), were proliferating.
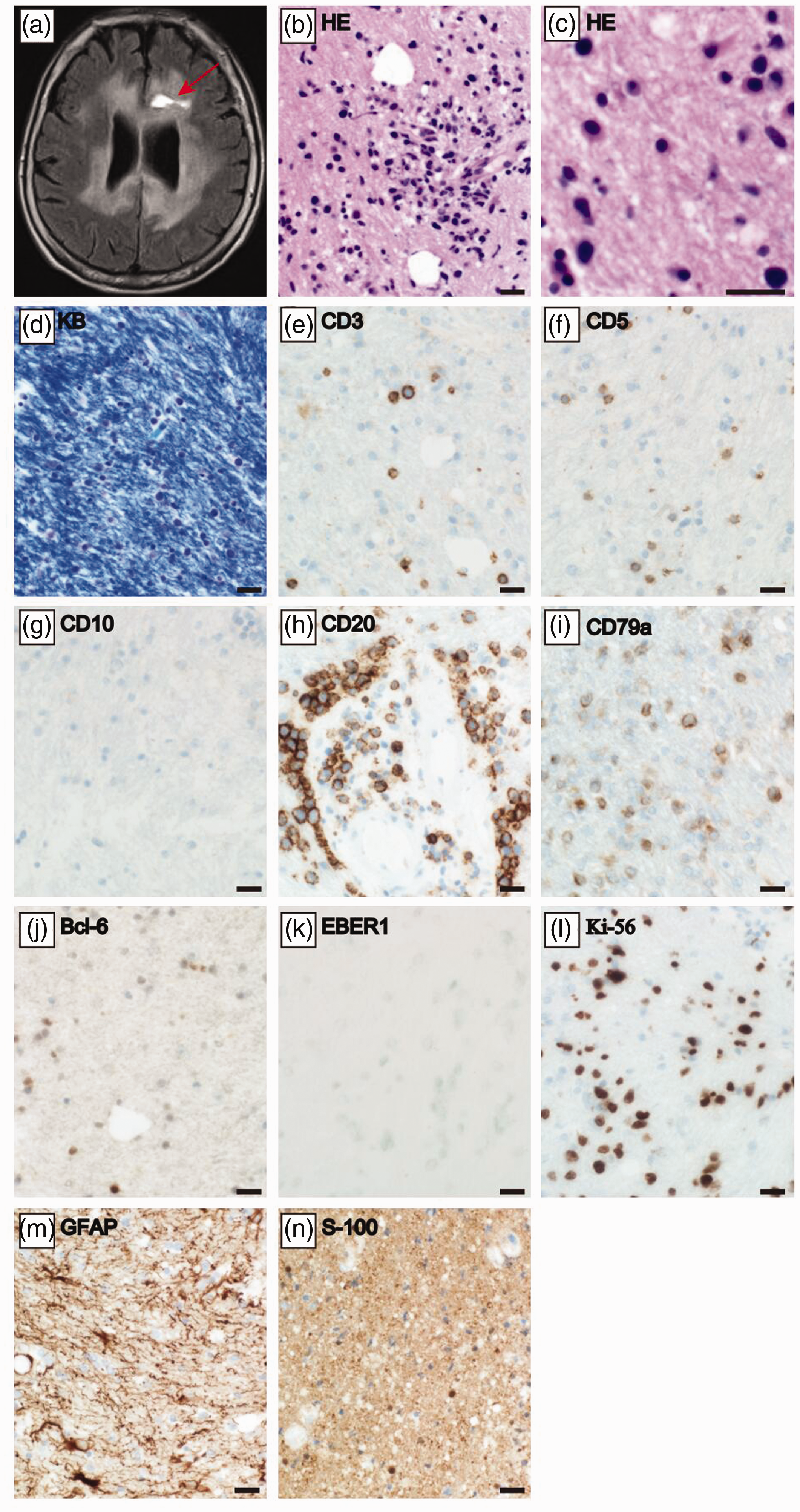
Brain biopsy examination. (a) Axial fluid-attenuated inversion recovery (FLAIR) image at the corpus callosum level immediately after the brain biopsy. The arrow indicates the brain biopsy scar. (b, c) HE staining at (b) low and (c) high magnifications. (d) KB staining. (e–n) Immunohistochemical staining for (e) CD3, (f) CD5, (g) CD10, (h) CD20, (i) CD79a, (j) Bcl-6, (k) EBER1, (l) Ki-67, (m) GFAP, and (n) S-100 protein. Scale bars = 20 μm
Based on this histological analysis following the brain biopsy from the lesion, our final pathological diagnosis was primary diffuse large B-cell lymphoma (DLBCL) of the central nervous system (CNS), also known as PCNSL. In the Karnofsky Performance Status, the patient’s score was 30 to 50. Her International Extranodal Lymphoma Study Group (IELSG) prognostic score was 4 (age, performance status, CSF protein level, and involvement of deep regions of the CNS). 15 , 16 The patient was treated with a high dose of methotrexate followed by whole brain radiotherapy (39.6 Gy in 22 fractions). Although whole-brain radiotherapy treatment has been omitted for some elderly PCNSL cases in an attempt to avoid late CNS radiation injury, 17 , 18 we performed this combination therapy of high-dose methotrexate and whole-brain radiotherapy to extend the patient’s survival time. 19 , 20 Unlike before treatment, oral intake became possible and she was able to communicate following treatment. The abnormal cranial MRI intensity areas were also reduced after treatment (Figure 3a–f). The patient returned home 13 months after symptom onset. A summary of her clinical course is shown in Figure 3g. We have de-identified all patient details in this case report and the reporting of this study conforms to CARE guidelines (for CAse REports). 21

Clinical information taken after the brain biopsy. (a–f) Axial fluid-attenuated inversion recovery (FLAIR) images (a–c) before and (d–f) after treatment. Abnormally high intensity areas were reduced within 2 months (from 6M to 8M). (g) Summary of the patient’s clinical course
Discussion
The present patient was diagnosed with PCNSL by histological analysis following a brain biopsy, even though several standard imaging techniques, including gadolinium-enhanced cranial MRI, 123I-IMP SPECT, and 18F-FDG PET, showed negative results for PCNSL. This report indicates that some cases of PCNSL—although most likely just a few—may escape detection because of negative results in imaging analyses.
PCNSL is an aggressive type of brain tumor and non-Hodgkin lymphoma that exclusively manifests in the brain. It is often difficult to differentiate PCNSL from other focal brain lesions. A brain biopsy is the gold standard method for diagnosing PCNSL, but both its invasiveness and the morbidity caused by complications can make its use challenging. A previous report 22 indicated a 3.6% incidence of major morbidity (hemiparesis, aphasia, or obtundation) and a 0.4% risk of fatality for a brain biopsy. Thus, noninvasive imaging examinations have been developed for pre-surgical PCNSL diagnosis. 23 , 24 Gadolinium-enhanced cranial MRI is the most useful imaging modality for the diagnosis of PCNSL. 25 In a study of 100 cases of PCNSL, contrast enhancement of cerebral or spinal lesions was detected in 95 cases. 5 Another report observed contrast enhancement in all 40 PCNSL cases that were investigated (i.e., 100% sensitivity). 6 Gadolinium-enhanced cranial MRI findings are also one of the standard endpoints when measuring the response to treatment. 26 In addition, 123I-IMP SPECT and 18F-FDG PET are useful imaging modalities for PCNSL diagnosis. Using 123I-IMP SPECT, PCNSL shows exceptionally high uptake, while the majority of brain tumors exhibit low uptake. 27 SPECT has a pooled sensitivity of 0.92 and a specificity of 0.84 for PCNSL. 24 In contrast, 18F-FDG PET has a pooled sensitivity of 0.88 and a specificity of 0.86 for this disease. 28 In summary, previous reports have demonstrated the high pre-surgical diagnostic accuracy of gadolinium-enhanced cranial MRI, 123I-IMP SPECT, and 18F-FDG PET for PCNSL.
According to previous studies, a negative gadolinium-enhanced cranial MRI, low uptake in 123I-IMP SPECT, and hypometabolism in 18F-FDG PET of lesions are very rare and atypical results for individuals with PCNSL. Our patient’s imaging results suggested diseases other than PCNSL. However, following a brain biopsy, the patient’s final diagnosis was PCNSL, and treatment for this disease improved the patient’s symptoms. Thus, although some reports have shown the usefulness of noninvasive imaging examinations for the diagnosis of PCNSL, 23 , 24 the present case report indicates that, in rare cases, a brain biopsy is the only method for a definitive diagnosis of PCNSL. We should therefore ensure that all patients enrolled in clinical trials for PCNSL have a histopathologically-confirmed diagnosis. 26
Lymphomatosis cerebri is a rare variant of PCNSL that is characterized by very little gadolinium contrast enhancement (only 35.6% positive). 29 Lymphomatosis cerebri is a continuous disease of PCNSL, 30 but differs from PCNSL in its histology; in lymphomatosis cerebri, the majority of tumor cells infiltrate the brain parenchyma but are limited around the vessels, while in PCNSL, many tumor cells also infiltrate around the vessels. 31 Moreover, the blood–brain barrier is preserved in lymphomatosis cerebri, and chemotherapy effectiveness in this disease is therefore more limited than in PCNSL. 32 Thus, we finally diagnosed the present case with PCNSL rather than lymphomatosis cerebri because many round cells had infiltrated around vessels (Figure 2b); however, the images were partially similar to those of lymphomatosis cerebri.
In conclusion, our patient demonstrated that it is possible to have PCNSL even though all imaging-related examinations (gadolinium-enhanced cranial MRI, 123I-IMP SPECT, and 18F-FDG PET) show negative results. Although this is likely a very rare case, it is nevertheless essential to employ supplemental histochemical analyses for the accurate diagnosis of PCNSL.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211035197 - Supplemental material for Case report of an elderly woman with atypical imaging for primary central nervous system lymphoma who needed a brain biopsy for diagnosis
Supplemental material, sj-pdf-1-imr-10.1177_03000605211035197 for Case report of an elderly woman with atypical imaging for primary central nervous system lymphoma who needed a brain biopsy for diagnosis by Kosuke Matsuzono, Tomoya Yagisawa, Keisuke Ohtani, Yohei Ishishita, Takashi Yamaguchi, Takafumi Mashiko, Tadashi Ozawa, Reiji Koide, Ryota Tanaka, Kensuke Kawai and Shigeru Fujimoto in Journal of International Medical Research
Footnotes
Acknowledgments
We appreciate the cooperation of the patient and her husband. In addition, we appreciate the assistance of Dr. Kenta Takahashi at the Department of Pathology, National Institute of Infectious Diseases, for examining the human polyomavirus 2 (JC virus) in brain tissue. We also appreciate the assistance of Dr. Yusuke Amano at the Department of Pathology, Jichi Medical University.
Ethics statement
Written informed consent for the publication of this report was obtained from the patient’s husband. The requirement for ethical approval was waived by the Ethical Committee of Jichi Medical University because this is a case report.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
