Abstract
Objective
To present our experience, discuss the complications, and assess early vs long-term outcomes of fluoroscopy-guided caudal epidural injection for lumbar disc herniation (LDH).
Methods
This was a prospective study of 309 patients who underwent fluoroscopy-guided caudal epidural injection from 2014 to 2020. The inclusion criteria were LDH diagnosis by magnetic resonance imaging, surgical treatment not required, age >18 years, and history of low back or leg pain despite >6 weeks of treatment comprising a combination of analgesics, anti-inflammatories, and physical therapy. The epidural injection solution comprised 8 mL of 0.5% bupivacaine hydrochloride (HCL), 2 mL dexamethasone, and 10 mL saline. Each patient completed a questionnaire comprising a visual analog scale (VAS) and the Back Pain Functional Scale (BPFS) at baseline, and 1 month and 1 year after injection.
Results
The VAS and BPFS scores indicated significantly less pain at 1 month and 1 year compared with the pre-procedure baseline values. Complications developed in 11 patients (reversible paresis in 7 patients, arrhythmia in 1 patient, headache in 1 patient, seizure in 1 patient, spondylodiscitis in 1 patient).
Conclusions
Fluoroscopy-guided caudal epidural injection for LDH is safe, and the procedure reduced pain and improved functional capacity compared with baseline.
Keywords
Introduction
Lumbar disc herniation (LDH) is among the most common health problems worldwide. Low back and leg pain due to LDH can be managed with several different treatments, and a multimodal treatment strategy is often used. Caudal epidural injections are one of the most common conservative treatments for LDH.1,2
The aim of this prospective study was to assess the efficacy of fluoroscopy-guided caudal epidural injections for LDH. Pain and functional status were investigated in patients with LDH to achieve pain relief as well as to evaluate complications related to the procedure.
Material and methods
This study was approved by the Ethics Institution Committee of Tınaztepe University (approval number: 012022/16082022) and was performed in accordance with the 1964 Declaration of Helsinki and its later amendments. All enrolled patients provided written informed consent to participate in this study. This study was written in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) criteria. 3
From January 2014 to January 2020, patients with LDH who underwent fluoroscopy-guided caudal epidural injection were prospectively enrolled in this study. The inclusion criteria were LDH diagnosed by magnetic resonance imaging (MRI), surgical treatment not required, age >18 years, and a history of low back or leg pain despite >6 weeks of treatment comprising a combination of analgesics, anti-inflammatories, and physical therapy. The exclusion criteria were cauda equina syndrome, peripheral neuropathy, arterial vascular disease, degenerative spondylolisthesis, congenital or degenerative lumbar spinal stenosis, infection, tumors, pregnancy, and pain duration of less than 6 weeks.
All patients were evaluated prior to the initial injection and followed-up in the outpatient clinic or by telephone interview 1 month and 1 year after the injections. Patients who were lost to follow-up or who underwent surgery after the injections were excluded from the study. Each patient completed a questionnaire comprising a visual analog scale (VAS) 4 and the Back Pain Functional Scale (BPFS) 5 at baseline, and 1 month and 1 year after the injections. The standard VAS is usually 100 mm long. 4 When measured to 1-mm accuracy, this results in a 101-point scale, as follows: 0, 1, … , 100. In this article, VAS was reported in mm and ranged from 0 to 100. The patients were asked to show their pain intensity on the line from left to right (left: no pain, right: strongest pain). The BPFS is a self-administered questionnaire comprising 12 items (work, school, home activities, habits, bending, wearing shoes or socks, lifting an object from the ground, sitting, standing, walking, climbing stairs, and driving) that measure the patient’s ability to perform physical activities. The last question can be answered by patients who do not drive by asking the patient to think of travelling. The BPFS may be completed in 30 seconds to 5 minutes. Each item is scaled on a six-point Likert scale ranging from 0 to 5, with “0” indicating the inability to perform the action owing to back pain and “5” indicating no difficulty. The total score ranges from 0 to 60, and lower scores indicate lower functional ability. The BPFS was originally written and validated in English; only the Turkish translation and cultural adaptations were made. 6 Because the current study was performed in the Turkish population, the results of the study are valid for the Turkish translation of the questionnaire.
All caudal epidural injections were performed by the author in an operating room with a mono-planar fluoroscope with the patient in the prone position and with a standardized technique. A wedge-shaped pillow was placed under the patient’s hips to tilt the pelvis and bring the sacral hiatus into greater prominence. Sedation was provided with intravenous midazolam and fentanyl, as required. Before the epidural injection, 1 g of cefazolin was injected intravenously. All patients were monitored during and after the injection using pulse oximetry, blood pressure measurement, and electrocardiography (EKG). The epidural injection solution consisted of 8 mL 0.5% bupivacaine hydrochloride (HCL) (40 mg), 2 mL dexamethasone (4 mg/mL), and 10 mL saline. The skin overlying the sacrococcygeal area was prepared with an iodine-based antiseptic solution. After localizing the tip of the coccyx using palpation, local anesthesia was provided at the injection site with 4 mL prilocaine diluted in 10 mL saline solution. The 20-gauge, 90-mm spinal needle, which was equipped with a stylet, was then inserted quickly through the skin just below the hiatus sacralis at a 45° angle to the skin surface until it reached the bone. The needle was then withdrawn slightly and repositioned parallel to the skin surface and advanced further to enter the sacral canal. A lateral fluoroscopic view was used to confirm that the needle was in the caudal epidural space. Aspiration was routinely performed, and if the aspirate was negative, 1 mL of contrast medium (iohexol, 300 mg iodine per ml) was injected to confirm the epidural flow (Figure 1). Intravascular, intrathecal, and/or soft tissue infiltration was excluded by obtaining antero-posterior and lateral radiographic views. Once confirmation of the needle’s position was obtained, the injection solution described above was injected. The total injectate was 21 mL, including contrast. Pressure was applied to the injection site after withdrawing the needle after the site was sterilized with povidone-iodine. The patients recovered in the outpatient clinic for 60 minutes after the injection until they were ready to be discharged home.

Representative contrast distribution seen on fluoroscopic lateral and anteroposterior views during caudal epidural injection.
The Statistical Package for the Social Sciences (SPSS) 25 (IBM Corp., Armonk, NY, USA) software was used to analyze the data. In the univariate and multivariate analysis, conformation of the data to normal distribution was analyzed using the Kolmogorov–Smirnov, Shapiro–Wilk, and coefficient of variation tests. Parametric methods were used to analyze data with a normal distribution, and nonparametric methods were used to analyze variables with a non-normal distribution. After the main factors in the quantitative data were statistically analyzed under control, the partial correlation test was used to analyze correlations between the variables. Pearson’s Chi-square and Fisher’s exact tests were used to compare categorical data using the Monte Carlo simulation. Univariate and multivariate logistic regression were used to evaluate a cause and effect relationship between the categorical and explanatory variables. Variables were analyzed at a 95% confidence level, and p-values less than 0.05 were considered significant.
Results
Forty-one of the 350 eligible patients were excluded because they were lost to follow-up or underwent surgery. Of the remaining 309 patients, the mean age was 56.5 years (range, 20–91 years), and there were 100 men and 209 woman. The mean duration of symptoms was 24.5 months (range, 6–71 months), and 725 injections were administered, with a mean of 2.35 injections per patient. The most commonly affected disc level was L5–S1. This was the affected disc level in 297 of the 309 patients; the L4–5 disc level was the affected level in 252 patients, and the L3–4 disc level was the affected level in 94 patients (Table 1).
The patients’ demographic and clinical characteristics.
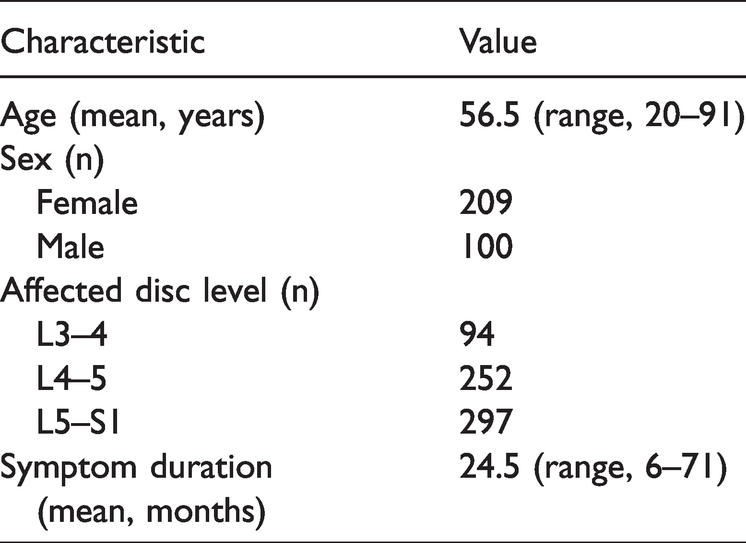
The VAS and BPFS scores indicated significantly less pain 1 month and 1 year post-procedure compared with the pre-procedure baseline values (p < 0.05; Table 2, Figure 2, Figure 3).
Comparison of VAS and BPFS scores before, and 1 month and 1 year after caudal epidural injection.

VAS, visual analog scale; BPFS, Back Pain Functional Scale; SD, standard deviation.
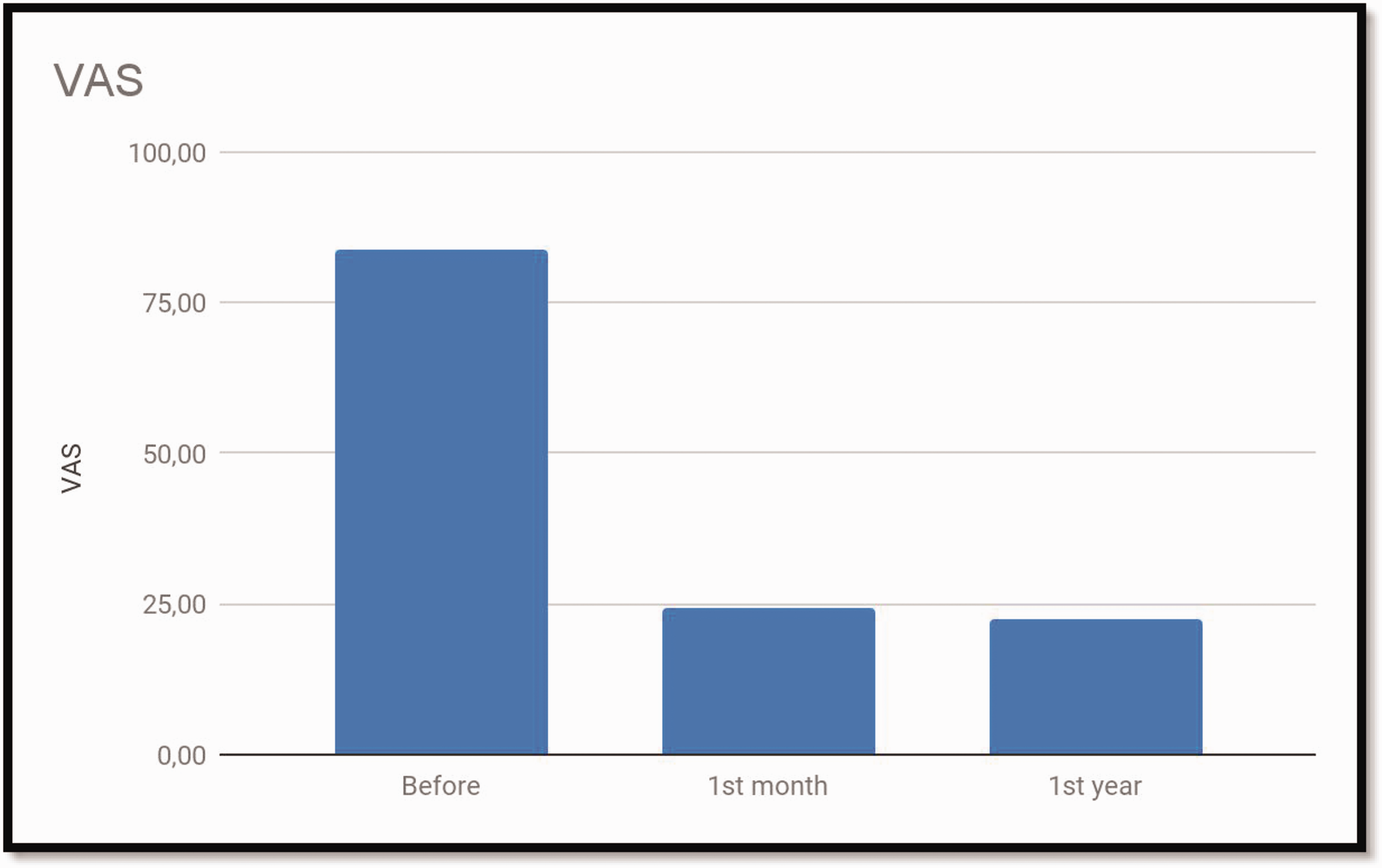
The patients’ VAS scores before, and 1 month and 1 year after undergoing caudal epidural injections for LDH.
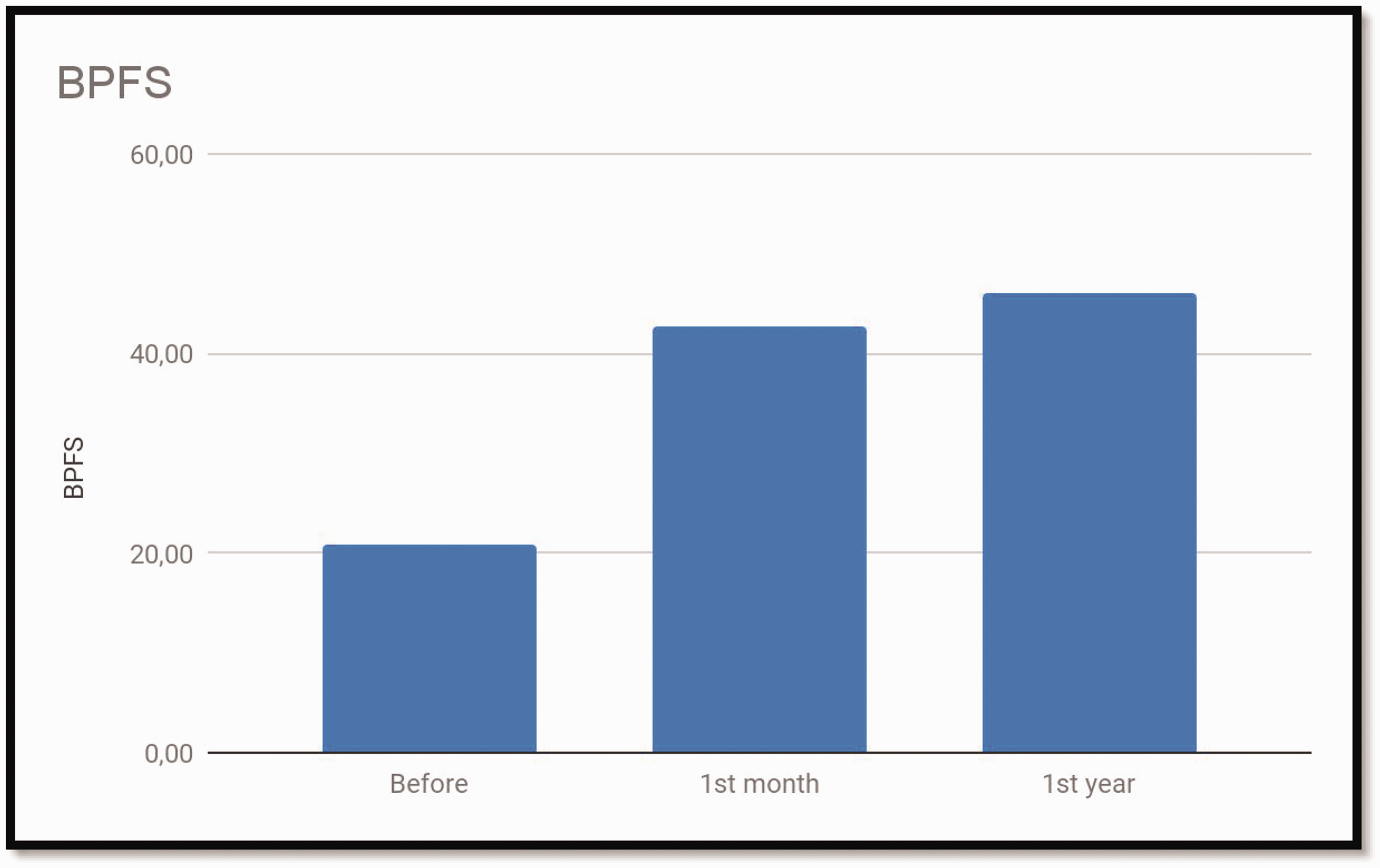
The patients’ BPFS scores before, and 1 month and 1 year after undergoing caudal epidural injections for LDH.
Complications were seen in 11 patients (3.56%) in this study. Seven patients experienced total anesthesia and paresis of the lower limbs; however, all seven patients recovered completely after 2 weeks. Cardiac arrhythmia was observed during the procedure in one patient. Cardiac examinations of this patient were performed, and no negative problem was found regarding the arrhythmia. Headache was observed during injection in one patient, and no problems were found in the neurological examination and cerebral imaging. One patient had a focal seizure during the procedure; however, no explanation was identified in further examinations of the patient. Spondylodiscitis was observed in lumbar MRI performed in one patient owing to severe low back pain that began 3 months after the procedure. Percutaneous needle biopsy was performed, and the results were negative. Intravenous antibiotic therapy was administered for 1 month followed by oral antibiotics for 2 months. The patient's low back pain complaint resolved with therapy, and the signs of spondylodiscitis observed in the initial MRI disappeared.
Discussion
The results of this study revealed a statistically significant benefit in patients with LDH and low back/leg pain 1 month and 1 year after fluoroscopy-guided caudal epidural injections. VAS and BPFS scores showed statistically significant improvement from baseline to 1 month and 1 year after the procedure.
Many questionnaires focusing on function have been developed in patients with low back and leg pain in LDH.4–7 However, despite their broad use, there is no evidence clearly suggesting that any one questionnaire is superior for assessing low back pain. The advantages and disadvantages of self-reported questionnaires evaluating activity limitations in low back pain have been studied.4–7 The VAS is one of the most commonly used scales to measure pain intensity in low back pain in the literature. 4 The BPFS is one of five Turkish-validated questionnaires evaluating activity limitation in patients with low back pain, and its validity and reliability have been proven. 6 The BPFS shows good correlation with the Roland–Morris Disability Questionnaire, Oswestry Disability Index, and Short Form 36 Health Survey.6,7
Caudal epidural injections can be performed blindly with fluoroscopic and ultrasonographic guidance. The dangerous complications of the caudal epidural technique are intravascular and intrathecal injections.1,2,8 During caudal epidural injection under fluoroscopic guidance, inadvertent intravascular injection was reported in 3% to 14% of patients, even after negative aspiration.1,2 Additionally, the incidence of closed sacral hiatus ranges from 2% to 3%. 8 However, caudal epidural injection is still considered the gold standard therapy for disc-related low back/leg pain, despite its disadvantages, namely high cost, difficulty performing the procedure outside the operating room, and radiation exposure. 2 Complications were seen in 11 patients (3.56%) in this study; 7 patients developed temporary paresis, which was considered to be caused by intravascular or intrathecal injection. Regarding the patients with cardiac arrhythmia, headache, and seizure (one each), these complications may have been related to the rate of injection.
Manchikanti et al. 2 performed a comparative systematic review and meta-analysis in 2021. The study assessed the efficacy of three routes of administration of epidural injections for LDH. The study analyzed the results of seven randomized controlled trials for caudal epidural injection. The authors reported that caudal epidural steroid injections were performed in three studies with a 6-month follow-up and two studies with a 12-month follow-up. The results demonstrated significant efficacy of steroids in caudal epidural injection. Caudal epidural injections showed level II evidence with local anesthetic and with steroids, or with local anesthetic alone for both short- and long-term pain relief. 2 Oliveira et al. 9 published a review comparing epidural injections of any steroid drug with placebo injections in patients with lumbosacral radicular pain in 2020. According to the authors, epidural steroid injections slightly reduced leg pain and disability at the short-term follow-up compared with baseline. No adverse events were reported during the short-term follow-up after epidural steroid or placebo injection in the reviewed studies. 9
Caudal epidural injection and transforaminal epidural injection are similarly effective in managing LDH. 10 The outcomes of fluoroscopy-guided caudal epidural injection and transforaminal epidural injection for LDH are similar regarding pain reduction and functional improvement. 10 However, both methods have advantages and disadvantages, especially regarding their use in L3–4 and L4–5 disc herniations. 10 The major disadvantage of caudal epidural injection in L3–4 and L4–5 disc herniations compared with transforaminal epidural injection is its low rate of anterior spread and that the procedure requires a much higher volume to achieve efficacy. 10 Additionally, the risk of vessel, nerve, and dural injury is higher with transforaminal epidural injections compared with caudal epidural injections. 10
There are some limitations in this study. First, there was no control group; neither the patients nor the doctor were blinded. Second, this was not a double-blind, placebo-controlled trial. This design was not chosen because the patients had severe pain, and it was thought that placebo administration would be unethical.
Conclusion
The results of LDH treatment with caudal epidural injection were satisfactory in the current study. This study showed that fluoroscopy-guided caudal epidural injection was safe, and that both early and late clinical outcomes were favorable.
