Abstract
Although the incidence of epidural hematoma formation after lumbar fusion surgery is low, it is a severe complication. If not managed promptly, it may cause compression of the spinal cord or nerve roots, leading to neurological dysfunctions such as limb numbness, weakness, and abnormal bowel or bladder function. We conducted a comprehensive retrospective review of the medical records, test results, and imaging data of a patient with a rare “spreading-type” symptomatic epidural hematoma occurring in the nonsurgical segment after lumbar fusion surgery. The patient was a 66-year-old male who, due to lumbar disc herniation with spinal stenosis, underwent transforaminal lumbar interbody fusion at L3/4–L4/5 levels under general anesthesia. On the second day after surgery, pain, numbness, weakness, and poor movement of both lower limbs—especially ankles and feet—suddenly appeared. Emergency computed tomography three-dimensional reconstruction of the lumbar spine showed that the implants of the lumbar spine were in good position, and an epidural hematoma was detected in the lumbar operative area. Exploration, hematoma removal, and enlarged decompression of the spinal canal were performed under emergency settings. Postoperative magnetic resonance imaging T2-weighted images revealed hypointensity in the spinal canal between the 12th thoracic and 2nd lumbar vertebra, suggesting the presence of a “spreading-type” epidural hematoma at the nonoperative level. A spreading-type epidural hematoma has been rarely reported in the literature. This type of hematoma mainly spreads longitudinally within the spinal canal and spreads till a distance of more than one vertebral body. A thorough understanding of the occurrence, development, and treatment process of symptomatic epidural hematoma after lumbar spine surgery (especially nonsurgical segment spreading-type hematoma) as well as the implementation of the “prevention–early detection–early surgery” process are crucial for preventing the formation of symptomatic epidural hematoma after spinal surgery and reducing the risk of nerve injury.
Keywords
Introduction
The formation of epidural hematoma after posterior lumbar decompression and internal fixation is often difficult to avoid, and this complication is closely monitored by spine surgeons. Postoperative intraspinal hematoma generally presents in two forms: the more common limited hematoma and the rarer spreading-type hematoma. Several factors can increase the incidence of postoperative epidural hematoma, such as concomitant medical conditions (e.g. hypertension, multiple myeloma, end-stage renal disease, and liver cirrhosis), the presence of highly vascularized tumors, and coagulation dysfunction. Once a symptomatic hematoma develops after surgery, early and timely identification, surgical removal of the hematoma, and complete decompression are essential for achieving a favorable prognosis. Here, we report the case of a 66-year-old patient who underwent posterior lumbar decompression, fusion, and internal fixation. The patient developed sudden, rapid, and progressive deterioration of lower limb neurological function after surgery. Importantly, we emphasize a relatively rare and special type of symptomatic epidural hematoma (SEH), referred to as the “spreading type,” which may help distinguish it from the more common “limited hematoma” at the surgical site. This case may serve as a warning to other spine surgeons when managing acute deterioration of lower limb neurological function after lumbar spine surgery and provides valuable reference for spreading-type SEH. This report conforms to the Case Report (CARE) guidelines, and patient information has been de-identified. 1
Case presentation
A 66-year-old man was admitted with complaints of lower back and left lower limb pain accompanied with numbness for more than 10 days, with no apparent cause. He reported that the pain was primarily localized to the lumbosacral region, left buttock, and anterior side of the left calf. The numbness was mainly present on the anterior side of the left calf and the medial side of the left foot. The symptoms worsened with standing or walking, and intermittent claudication in the left lower limb occurred after a few minutes of walking. The patient did not experience urinary or fecal dysfunction. Magnetic resonance imaging (MRI) at a local hospital revealed lumbar spinal stenosis at L3/4 and L4/5 levels. Nonsurgical treatments, including oral medication and physical therapy, failed to provide satisfactory symptom relief. The patient was therefore referred to the Department of Orthopaedics, Southwest Hospital, Third Military Medical University (Army Medical University), China, in July 2022 for further treatment, including surgical intervention. The patient had a history of smoking and alcohol consumption for >40 and >30 years, respectively. Physical examination revealed reduced superficial sensation of the skin on the posterolateral side of the left thigh, anteromedial side of the calf, and medial side of the left foot compared with that on the contralateral side. The muscle strength of the bilateral iliopsoas, quadriceps femoris, and extensor hallucis longus was grade 4, while that of the remaining lower limb muscles was grade 5. The Laseque sign was positive at 50°, and the strengthening test was also positive. Periurethral and perianal sensation as well as anal sphincter muscle strength were normal. MRI revealed disc herniation at L3/4 (Figure 1(a)) and L4/5 (Figure 1(b)) levels, with significant narrowing of the central canal and bilateral lateral recess at the corresponding levels. No significant abnormalities were observed in hematology or biochemical test results (Table 1).

(a–b) Sagittal and axial magnetic resonance image of the lumbar spine showing disc herniation at L3/4 and L4/5 levels, with the central canal and bilateral lateral recess significantly narrowed at the corresponding level. (c) The first postoperative anteroposterior and lateral lumbar X-ray showed that the pedicle screw length was appropriate, the screw and cage were well-positioned, and the screw did not enter the central canal or foramen intervertebrale and (d) the first postoperative urgent lumbar CT three-dimensional reconstruction examination also showed that the implants were in good position, and an epidural hematoma (red dotted circle) was formed in the lumbar surgery area. CT: computed tomography.
Coagulation function, HGB, and PLT were examined at each time point before and after the operation. The results showed no significant abnormality in hematology and biochemistry.

PT: prothrombin time; PT-INR: prothrombin time–international normalized ratio; PT%: prothrombin time percentage; APTT: activated partial thromboplastin time; Fib: fibrinogen; TT: thrombin time; HGB: hemoglobin; PLT: platelet count.
Based on the patient’s medical history, physical findings, and imaging results, the diagnosis of lumbar disc herniation with spinal stenosis was confirmed, and the patient fully met the surgical indications. After obtaining the patient’s written informed consent, L3/4 and L4/5 dual-segment transforaminal lumbar interbody fusion (TLIF) surgery was performed under general anesthesia, including L3/4 and L4/5 left laminectomy. The total intraoperative blood loss was approximately 200 mL. Before the end of the procedure, the dural sac and the left L3, L4, and L5 nerve roots were observed to be intact, relaxed, and thoroughly decompressed. A drainage tube was placed on each side of the incision. After recovery from anesthesia, the patient’s lower limb mobility was normal, and muscle strength was consistent with preoperative findings.
On the first postoperative day, the patient reported reduced numbness in the left calf and foot and demonstrated good voluntary movement of both lower limbs. The total drained volume of bloody fluid was 10 mL on the left and 23 mL on the right. On the second postoperative day, the patient experienced pain, numbness, and weakness in both lower limbs, accompanied with impaired mobility, particularly in the ankles and feet. Physical examination revealed decreased sensation on the posterolateral aspects of both thighs, anteromedial and anterolateral aspects of both calves, and the dorsum and soles of both feet. The muscle strength of the bilateral iliopsoas and quadriceps femoris was grade 4. The strength of the left tibialis anterior muscle and extensor hallucis longus was significantly reduced to grade 2, and that of the right tibialis anterior muscle and extensor hallucis longus significantly decreased to grades 1–2. The strength of the plantar flexor muscles, such as the gastrocnemius in both feet, was grade 4. The total drainage volume of bloody fluid on the left and right was 5 and 17 mL, respectively. Emergency lumbar X-ray showed that the implants were in good position (Figure 1(c)), and computed tomography (CT) three-dimensional reconstruction suggested the presence of an epidural hematoma in the lumbar surgical area (Figure 1(d)).
Emergency surgery was performed, consisting of exploration, hematoma removal, and spinal canal decompression. Intraoperative exploration revealed that the positions of the pedicle screws and titanium rods were normal. A hematoma measuring approximately 3 × 3 × 4 cm was detected at the fenestration site of the L3/4 and L4/5 laminae. The hematoma was solidified with slightly increased elasticity and extended into the spinal canal. The left L4 and L5 nerve roots were compressed. The hematoma was carefully separated from the nerve roots and thoroughly aspirated. The four walls of the pedicles implanted with screws were intact. The left L4 and L5 nerve roots were congested and edematous but structurally intact. To achieve complete nerve root decompression, the left L4 and L5 nerve root outlets were re-expanded. The right L3/4 and L4/5 laminae as well as ligamentum flavum were excised. The right lateral recess and nerve root outlet were expanded, achieving thorough decompression. A drainage tube was again placed on each side of the incision.
After the emergency surgery, physical examination revealed decreased sensation on the lateral aspect of both calves and the dorsum of the right foot. The strength of the left ankle dorsum extensor and extensor hallucis longus was approximately grade 4, while the strength of the right tibialis anterior muscle and extensor hallucis longus was grade 1. Postoperatively, the patient received antibiotic therapy to prevent infection, mannitol to reduce swelling, hormones (dexamethasone sodium phosphate injection) to alleviate inflammation, and cimetidine to prevent gastrointestinal stress reactions and provide pain relief. Neurotrophic therapy was also administered. In addition, the patient was given short-term methylprednisolone as pulse therapy. The incision drainage tubes were kept unobstructed and open. The postoperative recovery status at various time points is shown in Table 2 and Figure 2.
Changes in the patient’s condition and recovery status at different time points after the second operation.
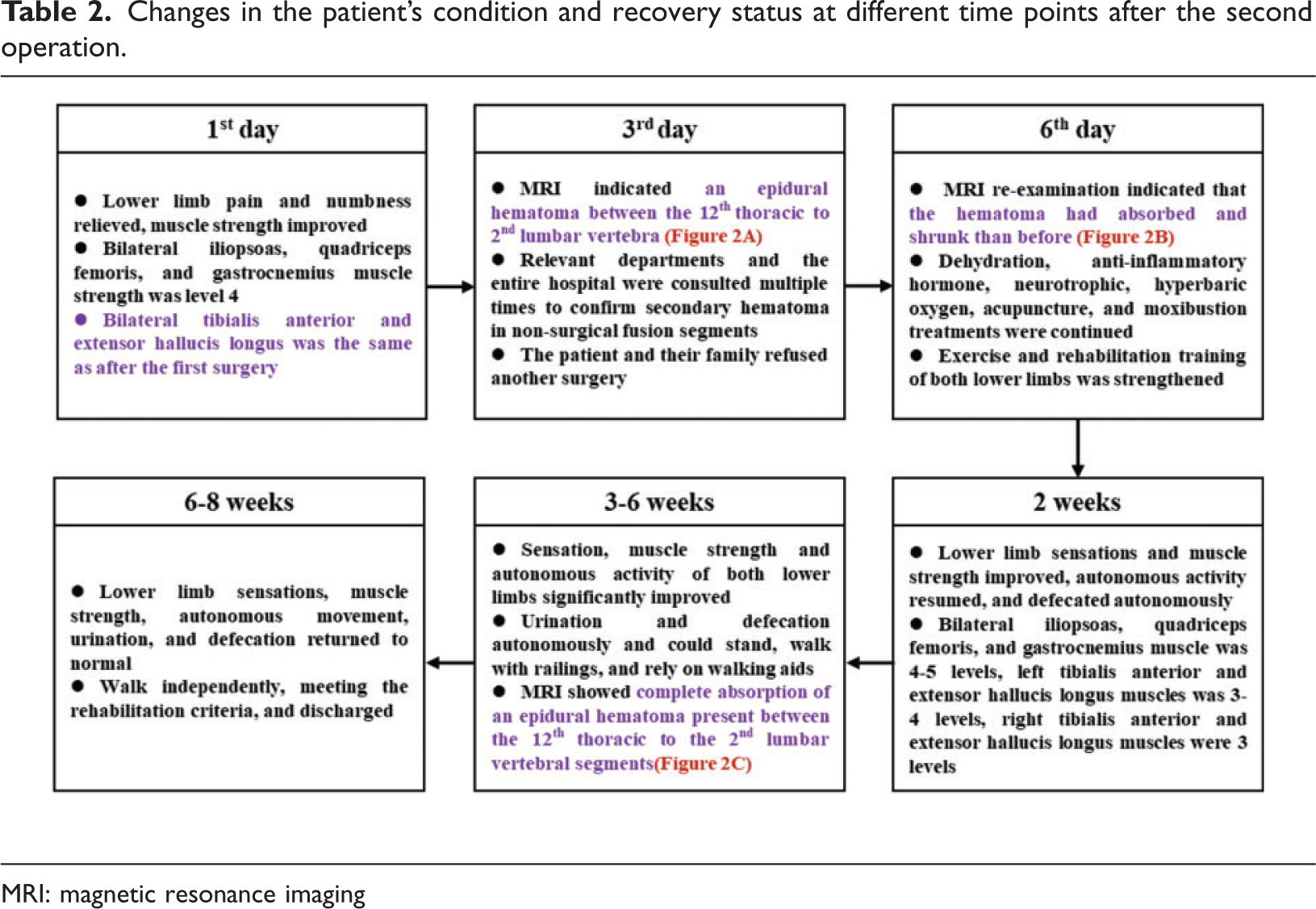
MRI: magnetic resonance imaging

(a) Lumbar MRI was re-examined on the third day after the second surgery, and sagittal plane and axial T2-weighted MRI images indicated hypointensity signals in the spinal canal between the 12th thoracic and 2nd lumbar vertebra, suggesting a nonsurgical segment spreading epidural hematoma (yellow dotted ellipse). (b) On the sixth day after the second surgery, an MRI re-examination was conducted, which indicated that the spreading epidural hematoma had absorbed and shrunk than that before the examination (yellow dotted ellipse) and (c) re-examination of sagittal plane and axial T2-weighted MRI (fifth week after the second surgery) showed complete absorption of the epidural hematoma present between the 12th thoracic and 2nd lumbar vertebral segments. MRI: magnetic resonance imaging.
Discussion
Most spinal surgical procedures result in the formation of a small, clinically inconspicuous epidural hematoma. SEH is characterized by the postoperative accumulation of hematoma in the spinal canal, leading to compression of the cauda equina, nerve roots, or spinal cord. This compression can be severe enough to cause neurological symptoms and often requires reoperation. 2 Although postoperative SEH is rare, it is a typical complication of spinal surgery. 3 The reported incidence of SEH after lumbar spine surgery is approximately 0.24%. 4 However, with the increasing complexity and frequency of lumbar procedures, the incidence of complications has also risen. 5 For instance, a systematic review of 1508 patients who underwent open posterior short-segment fixation reported 48 complications, including 5 cases of hematoma formation (incidence of approximately 0.33%). 6 SEH typically originates from the abundant venous plexus in the epidural space and is most frequently found in the thoracic vertebrae, where the epidural space is most prominent. 4 Clinically, SEH manifests as lower back pain and nerve root pain, with progressive neurological deterioration. The severity of symptoms depends on the level of compression. 7 Patients with new postoperative or immediate neurological deficits consistent with spinal cord injury or cauda equina syndrome should be evaluated for postoperative spinal epidural hematoma. 8
There are two common types of intraspinal hematoma. The first type is known as the “limited hematoma,” which primarily develops in the Kambin triangle on the side of the laminectomy and decompression, displacing the dural membrane to the opposite side. This hematoma spreads posteriorly from the vertebral space and can extend into the muscular space outside the laminae. Typically, it spreads cranially and caudally no more than the height of one vertebral body. The second type is the “spreading-type hematoma,” which has not been comprehensively studied. This type mainly extends longitudinally within the spinal canal over a distance of >1 vertebral body. 9 The present study reports a rare case of a spreading-type hematoma. The literature suggests that patients with preoperative hypocalcemia have a higher incidence of spreading-type hematomas and >2 segments of decompression fusion. 9 Repeated intraoperative irrigation may also create a loose epidural gap, allowing water pressure to open the space and facilitate hematoma migration. One possible explanation for the spreading epidural hematoma in this case is a postoperative rise in local pressure following hemorrhage, causing the hematoma to extend along the epidural space. Because this type of hematoma occurs in nonsurgical segments and develops suddenly and severely, it is usually challenging to manage. A better understanding of the characteristics of spreading SEH may help reduce the incidence of complications associated with postoperative epidural hematoma.
If not treated in the early stages, SEH can lead to neurological dysfunction, such as lower limb weakness, sensory disorders in the perineal region, and bladder detrusor and anal sphincter dysfunction. Therefore, early identification of SEH risk factors is crucial for prevention. Although various risk factors for SEH have been reported in the literature, 10 the risk factors specific to lumbar SEH remain unclear. This is largely because most studies have focused on cervical, thoracic, and lumbar spine surgeries collectively, and the presence of multiple confounding variables has resulted in inconsistent findings. Previous reports suggest that age ≥65 years is a preoperative risk factor for the development of lumbar SEH. In older patients, increased vascular fragility of the epidural venous plexus elevates the risk of bleeding. However, no significant association has been reported between the occurrence of lumbar SEH and sex.8,11–15 In the present case, the patient was a 66-year-old man, and his advanced age may have contributed to the postoperative spreading epidural hematoma in nonsurgical segments. These findings underscore the need for particular caution during spinal procedures in older patients, given their higher risk of developing epidural hematoma. Furthermore, several internal diseases have been associated with the incidence of SEH, including hypertension,11–14,16,17 multiple myeloma, end-stage renal disease, viral hepatitis, and cirrhosis. Epidural hematoma is most frequently observed in patients with coagulation dysfunction or highly vascularized tumors. A relationship between hypertension and blood viscosity has been established, with diastolic blood pressure identified as an independent predictor of whole blood viscosity. 18 Elevated diastolic blood pressure may increase blood viscosity, predisposing patients to blood clot formation and drainage dysfunction. 11 Intraoperative blood pressure elevations may lead to incomplete hemostasis, and progressive postoperative hematoma can compress the dural sac, resulting in SEH. Consequently, greater fluctuations in blood pressure are associated with a higher likelihood of hematoma formation. In contrast, diabetes and coronary heart disease have not been identified as risk factors for SEH.12,13,16,17,19 Similarly, adverse lifestyle habits such as smoking and alcohol consumption were not found to be significant risk factors for lumbar SEH.13,14,16,17 Laboratory findings have also indicated that preoperative platelet count abnormalities and coagulation dysfunction are risk factors for SEH.13–15,17 All coagulation function test results performed preoperatively and on postoperative days 1 and 3 were within the normal range. On postoperative day 9, the fibrinogen (Fib) level was slightly reduced (1.63 g/L), which may prolong coagulation time, impair hemostatic function, and relatively increase the risk of bleeding. However, because this abnormal value was detected on postoperative day 9—outside the typical time frame for hematoma formation—its clinical significance is limited.
Meta-analyses and other studies have indicated that intraoperative risk factors for lumbar SEH include revision surgery, operative time of > 2 h, intraoperative blood loss of ≥ 600 mL, intraoperative frozen plasma infusion, use of hemostatic materials, and multisegment surgery.8,11–14,16,17,19,20 Revision surgery has been identified as a risk factor for SEH after lumbar spine surgery, primarily because of the severity of local scarring in patients undergoing the surgery. 3 Prolonged operative time is often associated with increased intraoperative blood loss, which can lead to coagulopathy. In addition, extended incision exposure exacerbates paravertebral muscle injury, predisposing tissues to ischemic necrosis and further increasing the risk of SEH formation. In this case, the duration of the initial surgery was 177 min (approximately 3 h), and two lumbar segments were operated on, which might have increased the risk of the spreading type of SEH. Kita et al. reported that incomplete intraoperative hemostasis or improper techniques are key contributors to excessive intraoperative blood loss, thereby increasing the risk of postoperative vascular bleeding and SEH formation. 21 In addition, frozen plasma infusion reflects significant intraoperative blood loss. After infusion, abnormal coagulation factors and alterations in the internal environment—such as decreased body temperature and platelet concentration—can impair the normal coagulation process. 22 Meta-analyses have revealed that postoperative risk factors for lumbar SEH include the use of nonsteroidal anti-inflammatory drugs and anticoagulant therapy, whereas postoperative total protein levels are not risk factors for lumbar SEH.11,16,17,23
The following recommendations may help prevent the incidence of spreading SEH and improve patient outcomes. Preoperative anticoagulant therapy should be discontinued at least 1 week before surgery, and bridging therapy should be closely monitored. Preoperative blood calcium levels should be optimized. Bone wax should be applied to control bleeding at osteoporotic bone surfaces, and injury to the end plate should be avoided as much as possible during intervertebral disc treatment. During multisegment decompression, the operative time should be minimized, and tranexamic acid may be administered. Intraoperative hemostasis should be carefully maintained through methods such as electrocoagulation, compression, and the use of hemostatic materials. If postoperative pain in the operative area or neurological symptoms in the lower extremity occur, CT reconstruction should be performed promptly to assess screw positioning, and continuous MRI examination is essential to identify the underlying cause. Once an epidural hematoma is confirmed and accompanied by lower limb neurological symptoms or cauda equina syndrome, emergency hematoma evacuation and decompression should be performed immediately to preserve nerve function. Postoperatively, treatments should include infection prevention, dehydration therapy, hormonal anti-inflammatory therapy, methylprednisolone pulse therapy, and administration of neurotrophic factors for nerve support. In addition, to facilitate postoperative rehabilitation, adjuvant therapies such as hyperbaric oxygen therapy, traditional Chinese medicine acupuncture, and rehabilitation training may be considered.
Conclusion
Nonsurgical segment “spreading-type” SEH after lumbar fusion surgery is a rare but serious complication. This type of hematoma extends vertically within the spinal canal, potentially causing extensive nerve compression and severe neurological dysfunction, with its reported incidence varying across studies. Risk factors may include advanced age, low bone mineral density, coagulation dysfunction, concurrent systemic diseases, and surgical factors. Early recognition and timely intervention are essential to improve patient outcomes. For patients presenting with postoperative neurological deficits that do not correspond to the operative segment, MRI is the preferred diagnostic modality because it can accurately delineate the location and extent of the hematoma and the degree of neural compression. CT can provide complementary information regarding bony structures and hematoma density. In terms of treatment, once the diagnosis is confirmed and the patient presents with significant neurological deterioration, immediate surgical evacuation of the hematoma and extended decompression should be performed to maximize neurological function preservation. For patients with small hematomas, mild neurological symptoms, and no disease progression, conservative management may be cautiously considered, accompanied by close monitoring. This case report also has certain limitations. Multiple intraoperative photographs were originally taken to document the surgical findings, including the hematoma in the operative area and its compression on the nerves. Unfortunately, because of the passage of time and accidental damage to the camera, the memory card containing these images was lost, making it impossible to provide intraoperative photographs as supporting evidence. Nevertheless, relevant imaging scans have been provided as the basis for this report.
Footnotes
Acknowledgments
This manuscript has undergone professional editing and proofreading by MJ Language Editing Services.
Authors’ contributions
Zhigang Rong conceptualized and designed the study, acquired and analyzed the data, and drafted the manuscript. Lei Song and Chengmin Zhang contributed equally to this work by consulting relevant literature, organizing and analyzing data, and participating in manuscript preparation. Tianyong Hou is the guarantor of the integrity of the entire study, provided guidance and quality supervision, coordinated the work, and assisted in drafting the manuscript.
Availability of data and materials
All data and materials in this article are accurate, reliable, and available.
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Consent for publication
Written informed consent was obtained from the patient included in this study. The patient agreed to the publication of the research findings. To ensure anonymity, all identifiable details have been de-identified. Patient-specific information (e.g. name, date of birth, and appointment details) has been removed from all figures and tables.
All authors have read and approved the final version of the manuscript and consented to its publication in the Journal of International Medical Research.
Declaration of conflicting interests
The authors declare that they have no conflicts of interests.
Ethical approval
All procedures involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This study was reviewed and approved by the Ethics Review Committee of the Southwest Hospital (Project number: BIIT2024202), which authorized its implementation and publication.
Funding
The study was supported by a grant from the Chongqing Young and Middle-aged Medical High-end Talent Project (YXGD202401).

