Abstract
Despite the considerable efforts made to increase the prevalence of autogenous fistula in patients on hemodialysis, tunneled cuffed catheters are still an important access modality and used in a high percentage of the hemodialysis population. However, because of the conundrum posed by tunneled cuffed catheters, patients can develop a multitude of complications, including thrombosis, infections, formation of a fibrin sheath, and central vein stenosis, resulting in increased morbidity and mortality as well as placing a heavy burden on the healthcare system. However, with an increasing number of studies now focusing on how to manage these catheter-related complications, there has been less translational research on the pathology of these complications. This review of the most recent literature provides an update on the pathological aspects of catheter-related complications, highlighting what we need to know and what is yet to be discovered. The future research strategies and innovations needed to prevent these complications are also addressed.
Keywords
Introduction
The importance of vascular access in hemodialysis is becoming increasingly apparent. Placement of a tunneled cuffed catheter (TCC) is not an uncommon type of vascular access. 1 It is estimated that more than 70% of patients on hemodialysis in the United States choose a TCC as their primary access device at the start of long-term hemodialysis. Therefore, the performance of the catheter is vital in terms of the adequacy of hemodialysis and complications for patients. 2
In recent years, the increasing number of patients on hemodialysis has become a substantial economic burden for the public healthcare system because of the extra resources needed to manage TCC-related complications, which frequently arise in patients with end-stage renal disease. Catheter-related access complications include catheter dysfunction, catheter-related infections, and central vein stenosis (CVS). In the United States, more than 30% of all hospital admissions for patients on hemodialysis are for catheter-related complications, resulting in an annual length of hospital stay of 17 days per patient with end-stage renal disease.3,4
The National Kidney Foundation Kidney Disease Outcomes Quality Initiative (KDOQI) has provided evidence-based guidelines for vascular access in patients on hemodialysis since 1996. The recommendations for central venous catheters (CVCs) focus mainly on catheter dysfunction and catheter-related infection.
In the 2019 KDOQI guidelines, CVC dysfunction is defined as the inability to maintain the prescribed extracorporeal blood flow required to ensure adequacy of hemodialysis without increasing the duration of the hemodialysis session. The strategy for catheter recovery usually includes pharmacological prevention by use of a locking solution, thrombolytic therapy, and endovascular mechanical disruption of a fibrin sheath. Catheter exchange is the last step used to treat catheter dysfunction. 5
For catheter-related infections, the KDOQI guidelines recommend an infection control program that includes an infection surveillance team to monitor, track, and help prevent vascular access infections as well as evaluate the outcomes. Appropriate cultures must be obtained before starting empiric antibiotic therapy for the treatment of an infection suspected of having a connection with CVC because any change in antibiotics should consider the type of bacterial culture.
The KDOQI believes that it is reasonable to set up an individualized strategy to manage an infected catheter with detailed instructions according to the patient’s comorbidities, dialysis, and vascular access circumstances. The approach usually includes catheter exchange via a guidewire, removal and reinsertion of the catheter, salvage of a CVC, and antibiotic lock therapy. However, disruption of the fibrin sheath in patients with catheter-related bloodstream infections still needs to be studied. Furthermore, asymptomatic CVS should not be treated based only on the views of experts.
To better utilize catheters, knowledge of the pathology of catheter-related complications should be integrated with the guidelines to reduce morbidity and mortality in patients on hemodialysis.
Unlike for other types of vascular access, the ideal attributes of a TCC are multiple and necessary, 6 including a need to provide high blood flow rates with a moderate decrease in pressure, and with little outflow failure and pressure alarms, despite the patient’s fluid volume and catheter location inside the vascular lumen. Furthermore, an ideal TCC should prevent thrombosis and CVS by reducing trauma at the vein site, have a low risk of formation of a fibrous sheath, and prevent migration of bacteria from the exit site and subsequent formation of biofilm inside the lumen of the catheter. However, this has been impossible to achieve thus far. Each type of TCC is at risk of failing one or more of the above requirements, leading to serious complications. Therefore, it is imperative to understand the pathology of TCC-related complications, separating what is known and what is yet to be discovered, in order to develop successful therapies and devices and to help prevent these complications. 7 This review article summarizes the latest knowledge about the pathology of catheter-related complications, as well as future research strategies and innovations in this field.
Catheter dysfunction
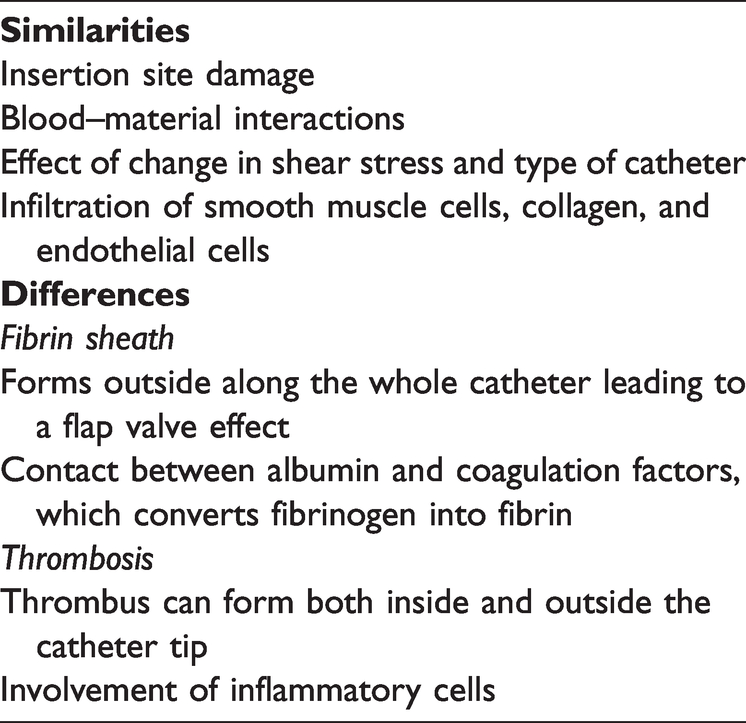
Catheter dysfunction is a very common complication, which can be classified as early or late. In general, technical problems are associated with early catheter malfunction, whereas late catheter malfunction refers to thrombosis and formation of a fibrin sheath. 8 Both have some similarities and differences (Table 1). These and other catheter-related complications are discussed below.
Differences and similarities between formation of the fibrin sheath and development of thrombosis.
Thrombosis
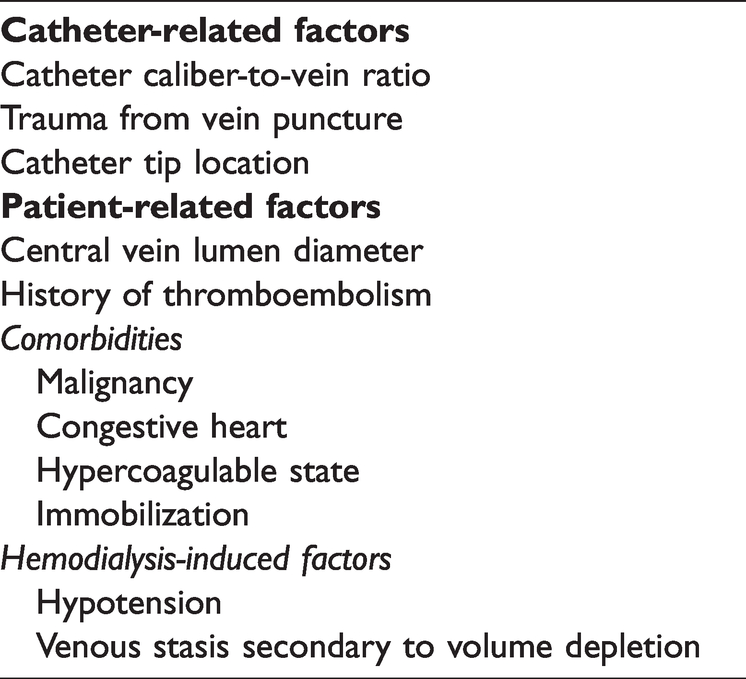
Thrombotic complications associated with use of a TCC are common in patients on hemodialysis, significantly increasing the cost of care and resulting in serious clinical outcomes, including catheter dysfunction, infections, and CVS. 9 Many factors can contribute to the development of catheter-related thrombosis, including the catheter caliber-to-vein ratio, 10 trauma from vein puncture, and location of the catheter tip. 11 Additional factors are patient-related characteristics, including the diameter of the central vein lumen, malignancy, history of thromboembolism, venous stasis following volume reduction, congestive heart failure, immobilization, hypotension, and a hypercoagulable state12,13 (Table 2). The exact mechanism underlying catheter-associated thrombosis is still unclear. However, some contributing factors have been suggested, including recurrent creation of vascular access, dysfunction of platelets and endothelium, inflammation, and clotting abnormalities. 14
Risk of thrombosis in tunneled cuffed catheters.
The pathophysiology of thrombus formation in patients with a TCC may involve multiple processes. First, the vessel walls at the access site are damaged after initial insertion of the catheter. Second, the coagulation and inflammatory response cascades come into play, activating leukocytes that release myeloperoxidase, leading to formation of platelet aggregates. 15 A soft thrombus then forms at or within the catheter tip. 16 Meanwhile, clots might develop throughout the lumen of the intravascular catheter. Thrombosis might also occur at the venous entry site. Sometimes clots may develop unnoticed, allowing for further deposition into a sheath-like structure encircling the catheter. Finally, fibrin forms on the surface of the thrombogenic catheter; thus, infiltration of smooth muscle starts within hours of insertion, with development of vascularized connective tissue that includes smooth muscle cells, collagen, and endothelial cells. 17 Inflammatory cells may also have a role in venous thrombosis 18 (Figure 1).

Pathology of thrombus formation in a tunneled cuffed catheter. The vessel wall at the access site is damaged after initial insertion of the catheter with activation of the coagulation and inflammatory response cascades. A soft thrombus then forms at or within the catheter tip. Finally, fibrin forms on the surface of the thrombogenic catheter. Infiltration of smooth muscle starts within hours of insertion, with development of vascularized connective tissue that includes smooth muscle cells, collagen, and endothelial cells.
After catheterization, the contact between the tip of the catheter and the venous wall becomes permanent, resulting in continuous formation of pericatheter thrombus in these areas, namely, where the catheter tip comes into contact with the vascular endothelium and at normal anatomic turning points.19–21 After catheters were put into the jugular vein in a swine model, a partial or circumferential mixed cellular and noncellular covering composed of smooth muscle cells, thrombus, and regions of endothelial cell populations was seen to develop. 9
In another study, researchers found that fibrin plaque was the most common component of the thrombi and was densely structured, even in central venous catheters removed after a short catheterization time (5 days). 22 This finding suggests that a solid-structured thrombus can form in the catheter lumen even after a short period of time.
Given the blood flow in the catheter, changes in fluid shear stress also have an influence on the development of thrombosis. When a catheter is inserted into a vessel, the physiological flow of blood inside the vessel may be disturbed, leading to non-physiological pressure and changes in shear stress.23,24 In terms of the importance of shear stress, coagulation may be influenced by hemodynamic parameters after catheterization. 25 In our study in a dog model, formation of thrombosis was also associated with high wall shear stress, and the likelihood of thrombosis increased over time. Wall shear stress at different sites in the venous wall varied according to the average circulation time and temporal fluctuation in pump blood flow. 26 There has also been a study demonstrating that platelet activation is generated mainly by the thrombogenicity of the device. The mechanical effects of shear forces on red blood cells are generally insignificant in comparison with those on platelets, which are harder than blood cells, and can be activated by shear stress at an order of magnitude less than that leading to hemolysis. 27 For example, platelets cannot be activated by a short exposure to a shear force of 6.0 Pa (0.1 s every 90 s) but can be activated if the exposure time is longer (>30 minutes), resulting in development of a significant amount of thrombin. 28 Thrombus can even start to form under low shear stress (<0.41 Pa) and a low shear strain rate (<54/s). 29 Furthermore, it has been shown that under extracorporeal flow, microaggregates can cause microvascular occlusion and tissue morbidity, which can contribute to platelet activation as a result of changes in shear stress. Additionally, minor concentrations of adenosine diphosphate are released when platelets are under the effect of shear stress in the extracorporeal flow. 25
Side holes in the TCC also contribute to formation of thrombus. Using computational fluid dynamics (CFD), Lucas et al. investigated the deposition of fibrin and formation of thrombus under the influence of shear stress in non-tunneled hemodialysis catheters with and without the obstruction of side holes. Development of thrombus in catheters removed from human patients undergoing hemodialysis has been investigated by scanning electron microscopy and two-photon laser-scanning microscopy. 24 The results indicate that there is more flow disturbance in the venous hole regions than in the arterial hole regions and more rapid development of thrombosis, which suggests that side holes play an important role in development of thrombosis in a TCC. It has been established that a larger side hole and a nozzle-shaped distal tip increase the effectiveness of catheters used for hemodialysis. 30 Using CFD in our dog model, we found that the distribution of wall shear stress on a vessel near side holes in a catheter is very uneven. As a result of the structure of the catheter, there was laminar blood flow in the area of the catheter without side holes and turbulent flow inside the area with side holes. Meanwhile, focal areas of intimal thickening were also seen in the venous wall adjacent to the sites of high wall shear stress. Smooth muscle cell migration is influenced by fluid shear stress; moreover, high shear stress stimulated migration of smooth muscle cells in a dynamic way for the entire length of the study, which suggests that shear stress and side holes have a role in intimal thickening after catheter insertion. 26
Fricker and Rockwell also investigated the effect of side holes on catheters. At the arterial inlet port, blood enters the catheter at an increasing rate, resulting in high shear stress inside the catheter lumen. As the flow rate increases, parabolic flow will develop after a longer distance within the catheter lumen, after which the hydraulic resistance of the catheter will also increase. 31 In this case, if the tip of the catheter is blocked, the entire flow will pass through the side holes, regardless of their size. Tal et al. performed a prospective study in which they compared Mahurkar-type single-body tunneled catheters with or without side holes. 32 They found that the catheters with side holes had more adherent clots than those without. There was also a high catheter-related bloodstream infection rate in the group with side holes. These findings suggest that clots attached to the tip of the catheter may operate as a nidus for infection caused by systemic bacteremia or contamination of the lumen.
It is also thought that different types of catheters have different characteristics in terms of formation of thrombi. Using CFD simulations and post-processing, Clark et al. investigated various types of catheters with regard to platelet activation induced by shear stress. They found that different types of catheters had different flow patterns, both at the side holes and the tip, which resulted in a different potential for thrombosis. Their findings may have consequences in terms of variation in the thrombogenic risk associated with the clinical performance of these catheters. 23
Another possible cause of thrombus formation is the origin of the artificial surfaces that come into contact with blood. These surfaces are thought to cause platelet adhesion; therefore, the reactivity of blood with this material needs to be evaluated because it is the first stage in the cascade responsible for development of thrombus. Platelets can adhere to certain altered plasma proteins adsorbed onto the surface of the material, which is central to initiation of the inflammatory and immune responses. 33
Fibrin sheath
In patients undergoing hemodialysis, the catheter dysfunction rate caused by formation of a fibrin sheath has been reported to be between 13% and 57%. 21 A fibrin sheath is the basic structural protein of the thrombus and leads to occlusion of the catheter, catheter exchange, and, finally, loss of the access site.34–36 Therefore, fibrin sheath development will determine the long-term performance of the catheter. 22
Formation of a fibrin sheath typically starts at the venous insertion site and continues distally along the catheter, 14 leading to a dynamic and continuous reaction between the venous wall and the catheter, as well as accompanying thrombus. 9 A fibrin sheath can develop within 24 hours of catheter implantation and is found around the majority of central venous catheters during autopsy, 37 and may eventually enclose the catheter completely during a period of 5 to 7 days.38–40 Furthermore, formation of the fibrin sheath can be worsened by continuous movement and mechanical irritation of the vein wall by a catheter made of biocompatible materials.16,41
From a biological point of view, the process is more complicated. When exposed to blood, the foreign surfaces of the catheter rapidly acquire a protein layer of approximately 100 nm, which is composed of fibrinogen, albumin, gamma-globulin, lipoproteins, and coagulation factors, which may contribute to subsequent blood-material interactions. 42 Platelets and coagulation factors are attracted to the fibrin sheath, which increases leukocyte adhesion.
Meanwhile, albumin reduces adhesion of platelets and leukocytes and counteracts these coagulation effects. Specific coagulation proteins, such as prekallikrein, high molecular weight kininogen, Factor XI, and Factor XII are engaged in the “contact phase” process, activating the intrinsic clotting pathway that converts fibrinogen into fibrin. 42 Within a period of weeks to months, smooth muscle cells from the venous wall move towards the intimal layer, resulting in deposition of collagen. We have previously reported that shear stress-regulated proliferation and migration of vascular smooth muscle cells is an endothelial cell-dependent process and that low shear stress modulates proliferation and migration of these cells through the caveolin-1-ERK1/2 pathway.26,43 The rate at which this process takes place varies between patients because of inherited or acquired factors 44 (Figure 2).

Pathology of formation of the fibrin sheath around a tunneled cuffed catheter. When exposed to blood, the foreign surface of the catheter rapidly acquires a protein layer of approximately 100 nm that is composed of fibrinogen, albumin, gamma-globulin, lipoproteins, and coagulation factors. Meanwhile, by reducing adhesion of platelets and leukocytes, albumin counteracts these coagulation effects, activating the intrinsic clotting pathway that converts fibrinogen into fibrin. Within a period of weeks to months, smooth muscle cells from the venous wall move towards the intimal layer, resulting in deposition of collagen.
The distal end of the fibrin sheath subsequently moves forward, progressively organizes, and eventually converts into fibrous connective tissue. Between the proximal and distal ends of the fibrin sheath is an area known as the transitional zone, which consists of a combination of fibrin and connective tissue. This zone finally covers the catheter tip as the sheath extends distally, where it can cause catheter dysfunction by creating a flap valve effect, allowing injection but preventing withdrawal of blood flow. 45 Furthermore, it has been shown that, unlike the collagen content, the cellularity of the fibrin sheath becomes less prominent as the indwelling time increases. 9
Shear stress also plays an important role in formation of the fibrin sheath. As already mentioned, high shear stress can cause structural changes in the fibrinogen absorbed onto a polymer, thereby increasing the binding surface area between fibrinogen and platelet receptors.46,47 Furthermore, in a study that compared the flow parameters between unobstructed and obstructed devices using CFD, it was found that the blood flow could be separated downstream into a high-velocity circulation, which is ejected from the venous holes of different types of catheters, and a low-velocity circulation, which occurs along the catheter surface wall. These conditions are thought to contribute to formation of fibrin plaques on the wall of the vein. 24 Moreover, in a clinical study, we found that the TCC dysfunction rate was higher in the iliac vein than in the internal jugular vein, 48 which suggests that different directions of blood flow may also influence formation of a fibrin sheath and thrombosis, but this awaits further investigation.
Regarding the catheter design, some authors have suggested that a catheter like the Split Cath, which has a “self-cleaning” function with high-velocity flow along the side holes created by the “step-down” tip, might reduce the likelihood of formation of a fibrin sheath, thereby decreasing the risk of bacterial infection. 7 However, this suggestion has yet to be tested in large trials.
Catheter infection
TCC-related infections are the primary cause of death in patients on hemodialysis but are preventable. 41 It is estimated that between 250,000 and 500,000 catheter-related infections occur annually in the United States.49,50 These infections have a substantial economic impact in terms of antibiotics, catheters, and hospitalization costs. According to the US Renal Data System, infections are a leading cause of death in patients on dialysis; in 2019, the total death rate in patients with chronic kidney disease on dialysis was 176/1000 patients, and that for septicemia was 26/1000 patients.51,52
There are many risk factors for infections in patients on hemodialysis. Some studies 53 suggest that these infections may result from a combination of three factors, namely, reduced human immunity, bacterial aggressiveness, and the intrinsic dangers of hemodialysis. Other factors related to the patient or catheter can also increase the risk of infection, 54 including diabetes mellitus, skin and nasal colonization, colonization of the catheter hub, long-term catheter use, history of catheter-related bacteremia, frequent manipulation of the catheter, the general health status of the patient at the time of catheter placement, 55 and formation of a bacterial biofilm (Table 3).
Risk factors for catheter-related infections.

The mechanism of catheter-related infection is even more complicated in patients with chronic kidney disease. In these cases, decreased neutrophil activity,56–58 abnormalities in cell-mediated and humoral immunity, cytokine production,59,60 and impairments in the function of the Fc receptor all contribute to depression of host immunity. Exposure to a foreign surface, such as a TCC, may cause bacteria to acquire virulence properties by formation of a biofilm layer, resulting from a matrix that consists of extracellular polysaccharides. 61 These molecules are secreted by the bacterial colony, leading to an increase in virulence gene expression through transcriptional activators. Virulence factors enable organisms to evade destruction by neutrophils. Furthermore, bacteria may be insensitive to antimicrobial agents as a result of the protection afforded by the biofilm. 14
Colonization of bacteria from the catheter tip may cause catheter-related bloodstream infections in three steps: first, bacteria from the skin migrate from the hub and along the lumen; second, bacteria outside the catheter migrate along the external surface of the catheter through the skin tunnel; and finally, bacteria can seed from other sites in the body via the blood. 62
The adherence properties of bacteria also play a role in catheter-related infections. Staphylococcus aureus usually adheres to fibronectin, which is a common host protein on catheters. Coagulase-negative Staphylococci usually adhere to polymer surfaces. Furthermore, there are many procedure-related risk factors for catheter-related infection, including damage to the skin barrier during catheterization, exposure to dialysis water, and the practice of dialyzer reuse. 63 Some pathogenetic mechanisms can also cause catheter-related bloodstream infections, and in extreme cases, intraluminal contamination of the catheter from the infusate liquid. Finally, irregularities on the surface of the catheter and its thrombogenicity contribute to microbial adhesion and the likelihood of colonization of the catheter and infection.
Bacterial biofilms can also cause catheter-related infections. They can develop from day 1 to day 14 and are a microbial-derived sessile colony, represented by cells that are permanently attached to a substrate or to each other and surrounded by a self-secreted exopolysaccharide matrix. 64 The exact reason for formation of biofilm in a TCC is unknown. However, hypothetically, the initial interaction of free bacteria with the surface of the hemodialysis catheter could trigger development of biofilm, resulting in generation of an exopolysaccharide layer that protects the bacteria from an inhospitable environment. Furthermore, the phenotype of the bacteria present in a biofilm is subject to change, resulting in resistance to antibiotics and disinfectants. Finally, data from an in vivo study suggest that formation of biofilm can be promoted by a low dose of heparin. 65
Jones et al. investigated formation of biofilm in different types of catheters used by patients on hemodialysis. They found that biofilm was present in all TCCs. However, the content of the biofilm varied according to whether or not bacteremia was present or a catheter locking solution was used. Meanwhile, there was a decreasing trend in bacterial density, biofilm thickness, percentage of surface coverage, and presence of an exopolysaccharide layer along the length of the catheter from the tip to the hub, which suggests that the original source of the infection may have been bacteria that migrated along the outside of the catheter to the tip or bacteria in the bloodstream. 61
Current studies that are investigating methods for prevention of catheter-related infections are focusing on catheter materials, chemical impregnation methods, and catheter locks that can kill bacteria in the biofilm layers both outside and inside the TCC.30,66,67
Central vein stenosis
CVS usually goes undetected in patients on hemodialysis because venograms are not routinely performed following placement or removal of a TCC. Therefore, the prevalence of CVS cannot be accurately determined and is generally underestimated. Its high prevalence in patients on dialysis probably reflects the fact that CVS manifests clinically only when blood flow via the maturing dialysis access increases, resulting in venous engorgement as a result of inadequate outflow. Therefore, most currently available data were obtained from imaging studies of symptomatic patients on dialysis. 68
Previous or concurrent use of a central venous device, such as a TCC or cardiac rhythm device, is the most common reason for development of CVS. CVS not related to a device is rare but may be caused by external compression or may even be idiopathic. 69 The mechanism of CVS is complicated and most likely related to catheter placement, which is complicated by heightened inflammation, increased oxidative stress, activation of leukocytes, release of myeloperoxidase, and activation of the coagulation cascade. 15
From a biological point of view, development of CVS usually starts with endothelial damage caused by venous cannulation and sustained by an indwelling biocompatible foreign body. Our research in a pig model identified cells in the venous neointima that were positive for α-smooth muscle actin, CD68, Ki67, smoothelin, and vimentin. 70 Movement of the catheter with respiration, head movements, changes in position, increased flow and turbulence during maturation of the arteriovenous access, and changes in shear stress lead to deposition of platelets and venous wall remodeling. 71 Meanwhile, trauma to the vessel wall contributes to generation of thrombin, activation of platelets, and expression of P-selectin with an inflammatory response and activation of leukocytes, all of which cause release of myeloperoxidase and development of platelet aggregates, which deposit into thrombi inside the vascular lumen.72,73 Catheters usually trigger formation of thrombus combined with venous stenosis at the same site. The relationship between thrombus and stenosis is still unclear 74 (Figure 3).

Pathology of central vein stenosis in a tunneled cuffed catheter. Development of central vein stenosis usually starts with endothelial damage caused by venous cannulation and sustained by an indwelling biocompatible foreign body. Movement of the catheter with respiration, head movements, changes in position, increased flow and turbulence during maturation of the arteriovenous access, and changes in shear stress lead to deposition of platelets and venous wall remodeling.
Activation of coagulation factors could also impact the development of CVS. In animal models, after catheterization, platelet microthrombi developed within 24 hours of endothelial denudation, which induced structural changes in the vein. 75 After a short period of time, smooth muscle cells migrated into the injured areas, leading to a localized proliferative change, but no obvious hyperplasia was found in other areas. Less direct evidence of histopathological changes is available for humans. A study in patients who had undergone directional atherectomy found that although they had no symptoms of stenosis or occlusion, their subclavian vein specimens showed intimal hyperplasia as well as fibrous tissue. 76 Another study in patients in whom a catheter was used for less than 14 days found adherent clots with intimal injury. Furthermore, in patients who had a catheter in place for more than 90 days, proliferation of smooth muscle cells and thickening of the venous wall could be observed. 77 Importantly, the catheters were focally adhered to the wall of the vein alongside organized thrombus, endothelial cells, and collagen, suggesting the possibility of formation of a fibrin sheath or central CVS.
Finally, lack of biocompatibility of the intravascular catheter is likely to be a factor contributing to venous injury and inflammation. Catheter materials may vary in their antigenicity, which can trigger tissue growth and fibrogenesis and should be investigated further. 78 Furthermore, TCC-related infection may aggravate inflammation and predispose to development of CVS. Interestingly, CVS is also thought to be a predisposing factor for infection, possibly leading to perpetual cycles. 15
Conclusion
Despite investment over decades in the development of dialysis catheters, the long-term problem of catheter-related complications have not been conquered and needs to be researched further. Although the ‘‘ideal’’ dialysis catheter has yet to be invented, catheters undeniably have an important role in immediate vascular access and as a bridge to an arteriovenous fistula or arteriovenous graft. 3
The common misconception that catheter-related complications are “par for the course” is still prevalent among patients. 20 Therefore, it is time for us to introduce some innovations to change the lives of patients with catheters. Before we start, there are still some questions to answer. For example, how long can a catheter be left in patients? What is the best location for the catheter tip? How does uremia influence catheter-related infection and thrombus? Is there a role for TCC as a preferred option?
Each of these questions will someday have an answer that improves the technology and benefits of TCCs used for hemodialysis. Future innovations should be focused on catheter-related infections, thrombosis, formation of a fibrous sheath around the catheter, CVS, and catheter material. We believe that a TCC would be an effective and safe long-term access option for most patients on dialysis and a good alternative when vascular resources are limited or difficulty is encountered with creation of an arteriovenous fistula or arteriovenous graft.
Footnotes
Author contributions
Lihua Wang and Lan Jia drafted the manuscript. Aili Jiang reviewed and edited the manuscript. All the authors read through the manuscript and agreed to the submission of the final version.
Declaration of conflicting interests
All the authors declare that they have no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the National Natural Science Foundation of China (No: 82170763).
