Abstract
Chronic myelocytic leukemia (CML) can occasionally occur after long-term chemotherapy for solid tumors; solid tumors secondary to chemotherapy and biotherapy for CML have also been reported. However, concurrence of these two phenomena in an untreated patient has seldom been reported. Herein, we describe the case of a female patient in her early 60 s who was transferred to the liver surgery department after the discovery of a large liver mass and elevated plasma alpha-fetoprotein levels. She was initially diagnosed with liver cancer. Blood tests indicated an increased platelet count (2464 × 109/L). Chromosomal examination from a bone marrow biopsy indicated the presence of the t(9;22) translocation, and subsequent fluorescence in situ hybridization and PCR were positive for the BCR-ABL rearrangement. A diagnosis of CML was made. The patient received hydroxyurea and imatinib to treat CML and underwent subsequent platelet-lowering therapy and a liver biopsy, which suggested moderately poorly differentiated adenocarcinoma or potentially hepatic metastatic carcinoma. However, the patient refused further pathological examination or screening for the site of the primary tumor. She died 6.5 months after discharge. The exact relationship between the two tumors remains unclear, and more patients need to be evaluated.
Introduction
Chronic myeloid leukemia (CML) is a myeloproliferative disease caused by the reciprocal translocation of chromosomes 9 and 22, which leads to a chimeric gene product known as BCR-ABL. 1 The Bcr-Abl fusion protein possesses constitutively activated Abl tyrosine kinase activity and activates downstream signaling pathways including Ras/MAPK, JAK/STAT, and PI3K/AKT, which are responsible for the pathogenesis of CML. 2 Clinically, CML is considered a triphasic disease, with an initial chronic phase, an intermediate accelerated phase, and a final, fatal blastic phase. 3 CML patients are usually characterized by extreme leukocytosis, myelocyte bulge, basophilia, and eosinophilia in peripheral blood. 4 Some CML patients can have mild to moderate thrombocytosis, and there are even individual reports of platelet counts in CML patients reaching over 10000 × 109/L. 5
Concurrent CML and solid tumors have been infrequently reported in four conditions. The first condition is myeloid sarcoma, commonly known as extramedullary acute myeloid leukemia and granulocytic sarcoma or chloroma, a tumor composed of myeloid cells occurring at an extramedullary site. 6 The most common sites of involvement for these include the small intestine, bone, skin, and lymph nodes. They can occur in different clinical settings such as in acute myeloid leukemia (AML) patients as a localized tissue mass, as blast crisis in CML patients, in leukemic transformation in myelodysplastic syndrome, before AML onset, and as an isolated neoplasm without evidence of AML. 7 The second condition is cases of secondary solid tumors reported after CML treatment including hepatocellular carcinoma (HCC), pulmonary carcinoid, and thyroid adenocarcinoma.8–10 The underlying mechanisms of these cases are unclear but may be related to chemotherapy-induced DNA damage, multiple genetic alterations, and increasing host susceptibility to oncogenic viruses due to immunosuppression. 8 The third condition involves the formation of secondary CML after treatment for solid tumors, which has been reported in non-small cell lung cancer, thyroid carcinoma, breast cancer, and gastric adenocarcinoma.11–13 To date, evidence is lacking regarding the frequency of therapy-related CML as a complication of cytotoxic therapy. The fourth condition is that CML can occur after organ transplant, with more than 30 cases of CML associated with organ transplants having been reported to date, most of which developed after kidney transplants.14,15 The development of CML in this situation may be associated with the post-organ transplant immunosuppressed state. However, it remains controversial whether the incidence of CML in such patients is truly higher than that of the general population owing to the limited number of reported cases.
In this case report, we report concurrent liver cancer with CML and extreme thrombocytosis in an untreated elderly woman.
Case report
An female patient in her early 60s was transferred to our hospital with complaints of upper abdominal pain accompanied by anorexia for 4 months. Doctors at a local hospital found a large mass in the patient’s right hepatic lobe by magnetic resonance imaging (MRI) and elevated plasma alpha-fetoprotein (AFP) levels (216.5 ng/mL). She was initially diagnosed with liver cancer and admitted to our liver surgery department. The patient had a 5-year history of hepatitis B (HBV) infection but had never been treated; she also had a 5-year history of hypertension and was treated with indapamide tablets. The patient neither smoked tobacco nor consumed alcohol. There was no notable family medical history, such as cancer. Physical examination upon admission showed no jaundice, ascites, pedal edema, or spider nevi, and the liver and spleen were not involved below the costal margin. There was no obvious enlargement of superficial lymph nodes, and the remaining physical examination findings were unremarkable. Laboratory tests at admission (Table 1) showed a hemoglobin level of 111 g/L, white blood cell count of 7.01 × 109/L, and platelet count of 2464 × 109/L. Liver function results showed serum alanine aminotransferase levels of 55 U/L, serum aspartate aminotransferase levels of 82 U/L, and globulin levels of 69 g/L. HBV-DNA levels were 2.11 × 107 IU/mL, and AFP levels were 308.8 ng/mL. Abdominal color Doppler ultrasonography showed a large hypoechoic area of approximately 9.5 × 5.7 cm in the right lobe of the liver, which was irregular in shape, had an unclear boundary, an uneven internal echo, and the distal part of the right hepatic vein was not clearly displayed as it was compressed by the mass (Figure 1a). Abdominal contrast-enhanced computed tomography (CT) scans showed a slightly low-density lesion in the right lobe of the liver with a maximum diameter of approximately 9.1 × 5.5 cm, slightly uneven enhancement, and delayed enhancement (Figure 1b, c). The hilar and retroperitoneal lymph nodes were increased and enlarged. These imaging and biochemical examination results suggested a neoplastic lesion in the liver, specifically, HCC.
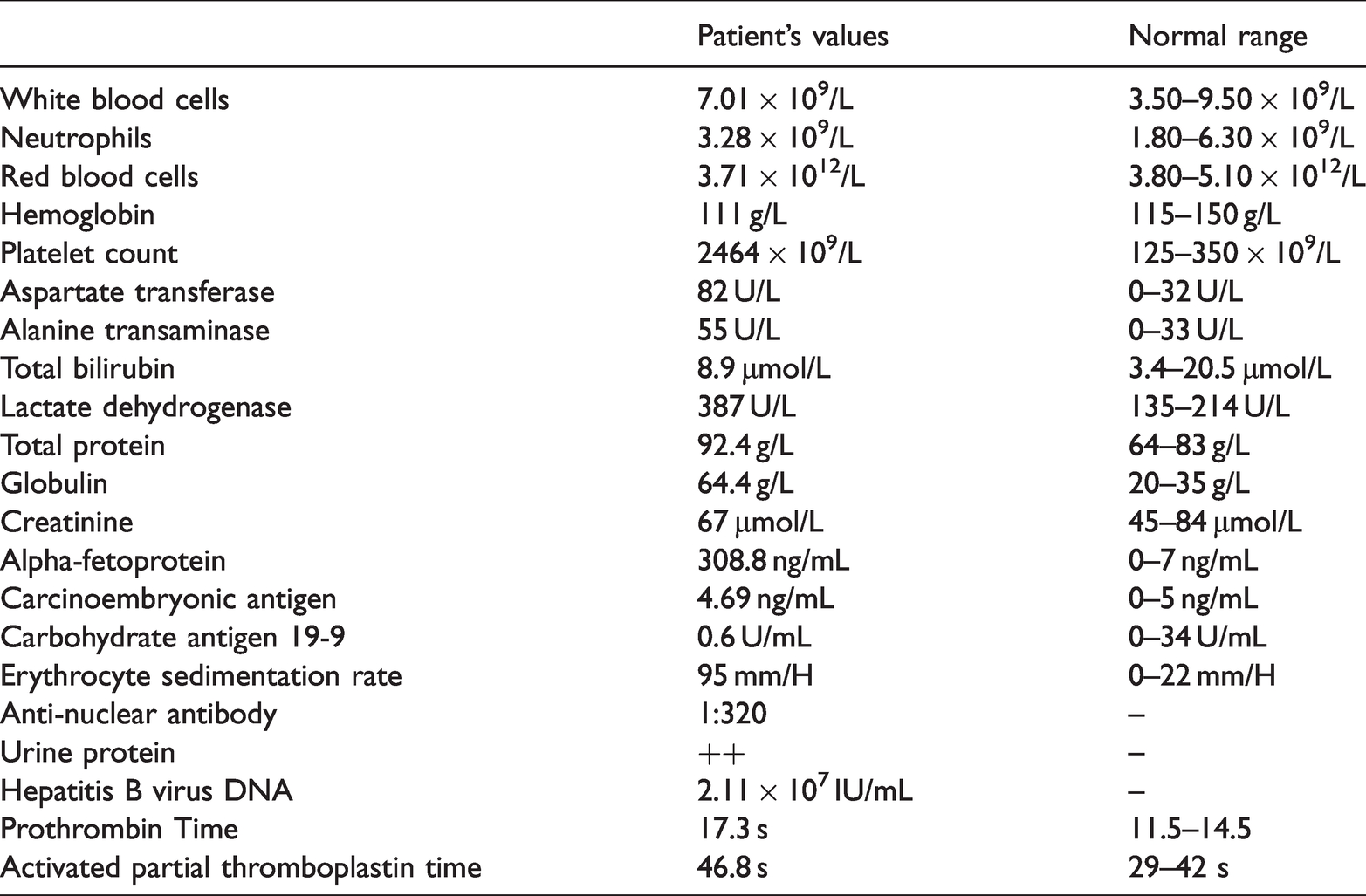
The main laboratory test results at admission.

Imaging features of the liver tumor in the patient. Abdominal color Doppler ultrasonography (a) showed a large hypoechoic area in the right lobe of the liver that was irregular in shape, had unclear boundaries, an uneven internal echo, and the distal part of the right hepatic vein was compressed. Abdominal contrast-enhanced computed tomography and (b) showed a 9.1 × 5.5-cm slightly low-density lesion in the right lobe of the liver that had slightly uneven enhancement and delayed enhancement.
The patient was transferred to our hematology department, and further biochemical examination results showed that the erythrocyte sedimentation rate was 95 mm/H, beta-2 microglobulin concentration was 4.62 mg/L, and anti-nuclear antibody was 1:320. No M protein was found in the monoclonal gamma globulin disease test. Bone marrow aspiration and biopsy were performed to examine bone marrow, and the results showed a hypercellular marrow with marked granulocytic and megakaryocytic hyperplasia (Figure 2a, b). Fluorescence in situ hybridization (FISH) analysis with the BCR-ABL dual fusion probe was performed to detect the BCR and Abl loci. When 200 cells in interphase were counted, approximately 20% of the cells expressed two fusion signals, one red signal, and one green signal (Figure 3a), suggesting that the sample was positive for the BCR-ABL fusion. Chromosome examination also indicated the existence of the t(9;22) translocation (Figure 3b). Quantitative PCR for the BCR-ABL (p210) fusion transcript showed that the copy number of the BCR-ABL transcript was 421,662, the ABL transcript copy number was 384,566, with a ratio of BCR-ABL to ABL transcripts of 109.65%; thus, the international standardization (IS) value was 109.65%. A diagnosis of chronic-phase CML was then made. The patient received hydroxyurea (1 g po tid) and imatinib (0.4 g po qd) to treat CML and platelet-lowering therapy with anagrelide (0.5 mg po bid), bayaspirin (100 mg po qd), and sodium bicarbonate (1 g po tid). She also received anti-HBV therapy with entecavir (0.5 mg po qd). She developed agranulocytosis on the seventh day of treatment with hydroxyurea (Figure 4a); thus, we reduced the hydroxyurea dose and gradually stopped it. She was also treated with an anti-infection and recombinant human granulocyte stimulating factor injection. Subsequently, her hemoglobin and platelets gradually decreased (Figure 4b, c). After 14 days of hydroxyurea treatment, the patient’s platelets returned to within the normal range (317 × 109/L), and then 3 days later, continued to drop to 21 × 109/L and gradually returned to normal. The main therapeutic drugs provided during the course of treatment are shown in Figure 4d. The second bone marrow aspiration and biopsy results both suggested that the erythrocyte and granulocyte series of bone marrow were suppressed (Figure 2c, d). Quantitative PCR for BCR-ABL showed that the copy number of the BCR-ABL transcript was 79,692, the ABL transcript copy was 1,282,604, with a ratio of BCR-ABL to ABL transcript of 6.21%; thus, the IS value was 2.36%. Flow cytology of the bone marrow suggested an increased proportion of lymphocytes and no abnormal expression of T lymphocytes, natural killer cells, or monoclonal B lymphocytes. The proportion of myeloid primordial cells was significantly decreased, and neutrophils and monocytes were mainly in the mature stage (complete results are available in Additional file 1).

Bone marrow aspiration and biopsy during hospitalization. Cytology of bone marrow at admission (a) showed a large number of platelet aggregations on bone marrow cytology slide. Megakaryocytes were easily found, and there were 16 granular megakaryocytes and four thromocytogenic megakaryocytes in 20 megakaryocytes. Excessive nuclear lobulation and abnormal nuclear lobulations were also found. The bone marrow biopsy at admission (b) suggested that megakaryocytes were clustered or scattered in different cell sizes, with hypolobation (mononuclear) or multinucleation (two or more round separated nuclei). The second bone marrow cytology (c) and biopsy (d) after treatment both showed severely inhibited bone marrow proliferation, megakaryocytes were easily seen, and hypolobations of megakaryocytes were also observed.

Bone marrow genetic testing results. A representative image of fluorescence in situ hybridization (FISH) analysis with a BCR-ABL dual fusion probe (a). The red probe represents ABL (chromosome 9), the green probe BRC (chromosome 22), and the yellow area shows where the two fluorophores are in close contact, indicating gene fusion and Chromosome examination (b) indicated the reciprocal translocation of the long arm of chromosome 22 and the long arm of chromosome 9; arrows indicate the regions of chromosome translocation.
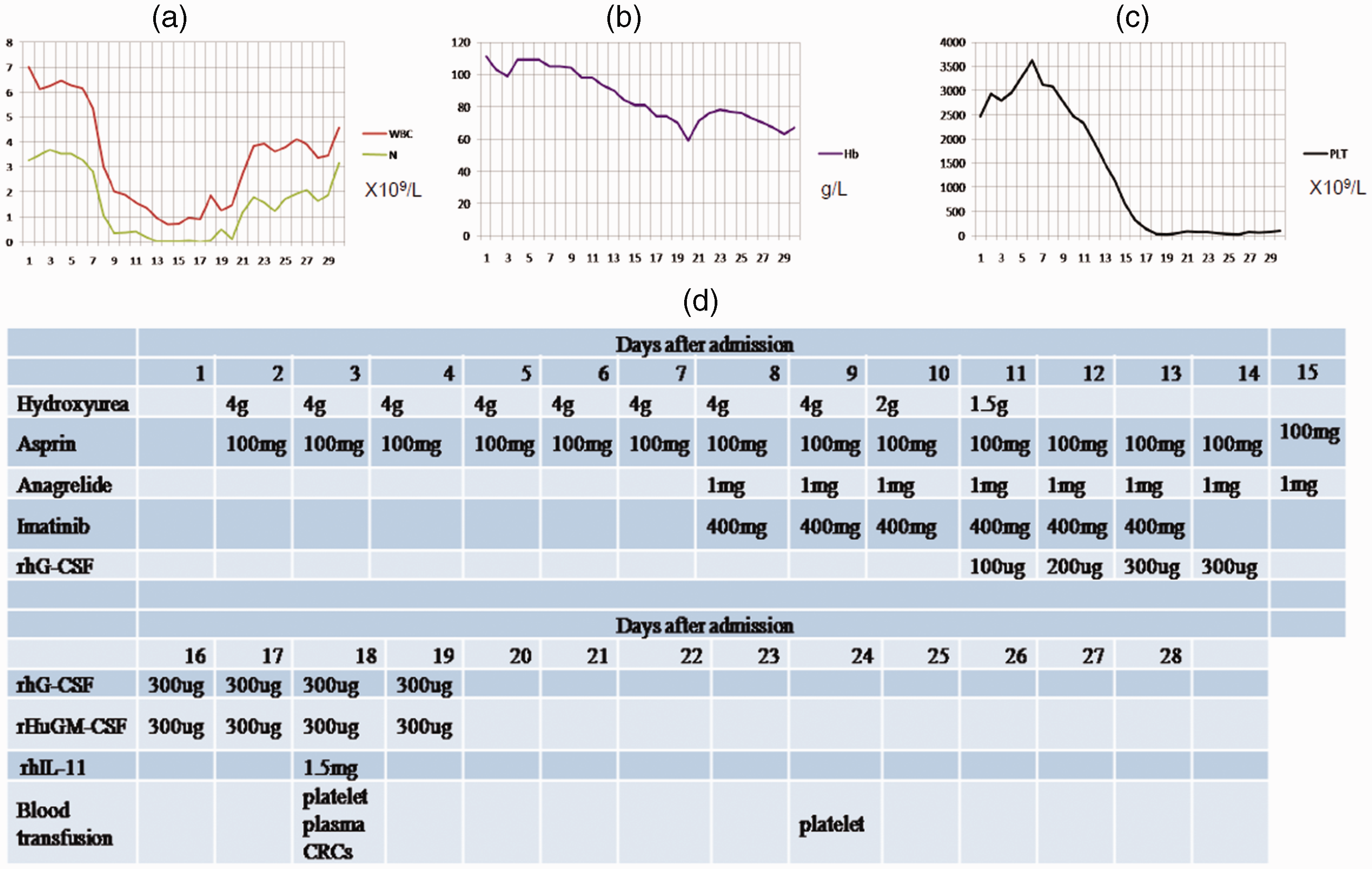
Treatments and the response to treatments in this patient. The trends of white blood cells (WBCs), hemoglobin, and platelets after admission, (a, b, c respectively) and The abscissa represents the days after admission, and the ordinates show the values of WBC (×109/L), hemoglobin (g/L), and platelets (×109/L), respectively. The main drugs and treatments during hospitalization (d).
The patient underwent a liver biopsy when her routine blood results returned to near normal, and hematoxylin and eosin staining suggested heterotypic adenoid and solid cell nests with necrosis in the chronic inflammatory liver tissue (Figure 5a). The Ki-67 index was approximately 30% to 40% (Figure 5b). Immunohistochemical studies showed positive staining for VILLIN, CK7, and EMA but negative staining for hepatocytes, Glypican-3, Arginasc1, AFP, CK19, CK20, CDX2, TTF-1, NapsinA, PAX8, ER, PR, and GATA3 (Figure 5c–r). The pathologists concluded that the patient had moderately poorly differentiated adenocarcinoma, and hepatic metastatic carcinoma could not be excluded.

Hematoxylin and eosin (HE) and immunohistochemical staining of puncture liver tissues. HE staining of liver biopsy specimen (a) showed heterotypic adenoid and solid cell nests with necrosis in the chronic inflammatory liver tissue (magnification: 100×). The Ki-67 labeling index was approximately 30% to 40% (b) and Immunohistochemical staining for CK7, EMA, VILLIN, hepatocytes, Glypican-3, Arginasc1, AFP, CK19, CK20, CDX2, TTF-1, NapsinA, PAX8, ER, PR, and GATA3 (c–r, respectively) (magnification: 100×).
The patient refused further pathological examination or screening for the primary tumor site and asked to be discharged from the hospital. She continued to take imatinib after discharge at a dosage of 0.4 g per day. However, she reduced the dosage by herself to 0.1 g per day due to a severe gastrointestinal reaction. During the fourth month after discharge, the patient’s platelet count was in the normal range (272 × 109/L), while color Doppler ultrasonography showed that the liver tumor had enlarged (9.8 × 7.3 cm). She died 6.5 months after discharge, and the exact cause of her death is unclear.
We have de-identified all patient details and ensured that the reporting of this case conforms to CARE guidelines. 16
Discussion
CML is a clonal disorder of hematopoietic stem cells that is characterized by the presence of the Philadelphia chromosome t(9:22) and manifests as an increased myeloid cell count, marked leukocytosis, myelocyte bulge, basophilia, eosinophilia, and normal to mild thrombocytosis in peripheral blood. 4 Rare cases of CML may present with isolated, marked thrombocytosis, which is defined as a platelet count greater than 1000 × 109/L. 17 Sora et al. 18 evaluated the marked thrombocytosis associated with CML in a large series of patients from 16 different Italian hematological centers treated between January 2002 and December 2015. Among the 1591 CML patients, 87 with extreme thrombocytosis were identified, with platelet counts ranging from 1054 × 109/L to 4720 × 109/L. CML patients with atypical clinical features and severe thrombocytosis often mimic the features of essential thrombocytosis (ET), as reported previously. 19 In our case, the patient's platelet count was as high as 3622 × 109/L, but her erythrocyte and granulocyte series in peripheral blood were generally normal. However, FISH, chromosome examination, and RT-PCR confirmed the diagnosis of CML. Moreover, bone marrow aspiration and biopsy revealed megakaryocytes that were smaller than normal cells and with typical hyperlobated round nuclei. Therefore, we and other researchers propose that every patient with ET should be tested for the Philadelphia chromosome to avoid missing CML diagnoses.
The main pathogenesis of CML is the reciprocal translocation between chromosomes 9 and 22, which leads to the tyrosine kinase coding gene ABL of chromosome 9 fusing into the BCR region of chromosome 22. An important result of this chromosome translocation is the product of the ABL-BCR fusion gene, which encodes a deregulated tyrosine kinase, resulting in manifestations of CML. Inhibiting the oncogenic expression and protein function of BCR-ABL are the primary treatments for CML on the basis of its pathogenesis. Bcr-Abl tyrosine-kinase inhibitors (TKIs) are the first-line therapy for CML; among which, imatinib is one of the most classic first-line drugs. In the recent study by Sora et al., the majority of patients received pretreatment with hydroxycarbamide and were then treated with TKIs (63 patients received imatinib, 16 received dasatinib, and 8 were given nilotinib), and 87% of patients achieved a complete cytogenetic response (CCyR) or a major molecular response (MMR) after 12 months of treatment. 18 In our case, the patient received pretreatment with hydroxycarbamide and platelet-lowering therapy with anagrelide, but it did not achieve a relevant reduction in platelet count. However, when imatinib therapy was started, the platelet count rapidly decreased to within the normal range. At the 4-month follow-up after discharge, her platelet count remained in the normal range.
Another issue in our report is the concurrent solid tumor with CML in this one patient. The abnormally elevated AFP levels, liver masses on CT imaging, and past history of HBV infection all support the clinical diagnosis of a liver tumor, particularly HCC. However, continuous monitoring of tumor markers revealed a downward trend in AFP levels (Additional file 2). Pathological diagnosis of the liver biopsy suggested moderately poorly differentiated adenocarcinoma, and it is also possible that the tumor was a metastatic liver tumor. However, neither abdominal enhanced CT scans, chest CT scans, nor gastroscopy investigations (Additional file 3) showed tumors in other parts of the abdomen. Owing to the particularity of liver tumors concurrent with hematological tumors, we consulted the literature and found some other reports of liver tumors coexisting with hematological tumors. Katoh et al. reported the case of a 55-year-old Japanese man with CML who had received an alkylating agent for 16 years and was later diagnosed with HCC with clinically evident splenic metastases. 8 This was the first report in the literature of HCC associated with CML. The authors suggested that long-term chemotherapy-induced DNA damage may have endowed HCC with an aggressive proliferative potential. 8 Ansari et al. presented the case of a 72-year-old man diagnosed with CML who subsequently presented with a liver mass that was pathologically diagnosed as histiocytic sarcoma (HS). 20 The liver mass retained the BCR-ABL1 translocation, suggesting clonality between the CML and HS. Therefore, the authors proposed that the concurrent expression of immunoglobulin heavy (IGH)-/light-chain rearrangements or cytogenetic markers common to primary CML suggested an evolutionary mechanism involving lineage switching that was potentially affected by genetic or epigenetic factors that may have occurred at the level of a progenitor or in the malignant cell itself. 20 Myeloid sarcoma is a rare extramedullary presentation of neoplastic myeloid cells that may occur before, concurrent with, or after the diagnosis of myeloproliferative disorders, such as AML or CML. Norsworthy et al. reviewed 51 cases of biopsy-proven myeloid sarcoma involving the liver, biliary tree or pancreas, 21 and most cases presented with systemic disease, mainly associated with AML and CML. The main symptoms included jaundice, right upper quadrant abdominal pain, fatigue, anorexia, nausea, and vomiting. Many patients were initially diagnosed with cholecystitis, pancreatitis, or carcinoma, especially those with unknown hematological diseases, and underwent surgery. As immunohistochemistry confirmed, the most commonly expressed cell surface markers of myeloid sarcoma in these patients were myeloperoxidase (63%), CD68 (40%), CD45 (30%), CD34 (30%), CD43 (30%), CD117 (28%), CD33 (15%), lysozyme (15%), and CD13(8%). 21 In this case, more immunohistological tests could have been performed to better diagnose the patient; unfortunately for our patient, there were no additional samples for further immunohistochemical detection of myeloid sarcoma markers. Although the platelet count was reduced to a normal range, imatinib therapy did not reduce the size of the liver tumor.
In summary, we presented a rare case of liver cancer concurrent with CML and extreme thrombocytosis in an elderly patient. However, the exact pathological relationship between the liver cancer and CML in this case is unknown.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221123684 - Supplemental material for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221123684 for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report by Ping Han, Zhi-Qiang Han, Xia Mao, Jin Wang, Qin-Lu Li, Min Xiao, Li Meng, De-An Tian and Zhen-Ya Hong in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221123684 - Supplemental material for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605221123684 for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report by Ping Han, Zhi-Qiang Han, Xia Mao, Jin Wang, Qin-Lu Li, Min Xiao, Li Meng, De-An Tian and Zhen-Ya Hong in Journal of International Medical Research
Supplemental Material
sj-pdf-3-imr-10.1177_03000605221123684 - Supplemental material for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report
Supplemental material, sj-pdf-3-imr-10.1177_03000605221123684 for Liver cancer concurrent with chronic myelocytic leukemia and extreme thrombocytosis: a rare case report by Ping Han, Zhi-Qiang Han, Xia Mao, Jin Wang, Qin-Lu Li, Min Xiao, Li Meng, De-An Tian and Zhen-Ya Hong in Journal of International Medical Research
Footnotes
Acknowledgements
The authors would like to thank all members of the study team, the patient, and their family.
Author contributions
Ping Han and Zhi-Qiang Han collected and analyzed the data and wrote the first draft of the manuscript; Xia Mao, Jin Wang, Min Xiao, and Qin-Lu Li performed the bone marrow cytology, chromosome, FISH, and flow cytometry tests. Li Meng and Dean Tian were in charge of the patient’s management; Zhen-Ya Hong supervised the patient’s management, critically revised the text, and made substantial scientific contributions. All authors approved the final version of the manuscript.
Consent for publication
The patient’s daughter gave written informed consent for the publication of this report.
Data availability statement
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology (no: TJ-IRB20190619), and written informed consent for the treatments was provided by the patient’s daughter.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Science Foundation of China (No. 81702396, 81772788, and 81873430).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
