Abstract
Objective
To evaluate the tolerability and efficacy of nasal pillow-noninvasive ventilation (NP-NIV) compared with high-flow nasal therapy (HFNT) in postsurgical patients.
Methods
This propensity score-matched retrospective study enrolled postoperative patients that received NP-NIV (NP-NIV group) or HFNT (HFNT group) in the intensive care unit. Data were collected from their medical records and the tolerability and respiratory status before and after extubation were compared between the two groups.
Results
The study enrolled 83 patients in the NP-NIV group and 27 patients in the HFNT group. After propensity score matching, there were 19 patients in each group. After matching, there were no significant differences in the baseline demographic and clinical characteristics before extubation. The tolerability was similar in both groups. When the NP-NIV group was compared with the HFNT group, the respiratory rate was significantly lower (median 16 [interquartile range, 14–17] versus median 19 [interquartile range, 18–26], respectively) and the partial pressure of arterial oxygen/fraction of inspired oxygen ratio was significantly higher (median 205 [174–256] versus median 155 [130–192], respectively) at 1 h after extubation.
Conclusion
NP-NIV was equally well tolerated and provided better respiratory support than HFNT in postsurgical patients.
Keywords
Introduction
It is reported that 10–15% of patients in intensive care unit (ICU) undergo reintubation due to extubation failure, 1 which is defined as the need for reintubation within 2–7 days of the scheduled extubation. This increases mortality by 25–50%, prolongs the duration of ventilation, increases the incidence of ventilator-associated pneumonia and prolongs ICU stay and hospitalization.1,2 Therefore, the prevention of extubation failure is important in respiratory management in the ICU. Guidelines for ventilator weaning recommend the use of preventive noninvasive ventilation (NIV) in patients at high risk of extubation failure. 3 The clinical practice guidelines for the use of NIV in acute respiratory failure also recommend prophylactic NIV use after extubation in patients at high risk. 4 On the other hand, high-flow nasal therapy (HFNT) has recently been increasingly reported to show clinical efficacy5,6 due to its good tolerability 7 and physiological support.8,9 HFNT is a device that can deliver up to 100 l/min (mainly 50–60 l/min) of heated and humidified high-flow oxygen set at a constant fraction of inspired oxygen (FiO2) through a nasal prong as respiratory support. 10
Noninvasive ventilation and HFNT have been used as effective alternatives to conventional oxygen therapy (COT) for extubation failure.3–5,11,12 It is controversial whether NIV or HFNT is more useful in preventing extubation failure. The respiratory support provided by HFNT is limited because the positive pressure it delivers may not be as stable as the pressure provided by NIV. However, HFNT can be used for a longer period because it is more comfortable and allows patients to speak, eat and drink while using it. In contrast, NIV is difficult to use continuously for a long period and can lead to complications such as facial skin injury. In a study on the efficacy of NIV for extubation failure, 10% of patients could not tolerate the use of NIV for 24 consecutive hours after extubation. 13 NIV with a nasal interface is mainly used for chronic neuromuscular disease and has excellent comfort and tolerability when attached. 14 However, nasal mask NIV has been reported to be more effective in preventing extubation failure than COT. 12 The nasal pillow NIV (NP-NIV), a type of nasal mask NIV, uses a specially shaped interface that covers only the nostrils and is well tolerated. NP-NIV is similar in shape to HFNT but allows for higher positive end-expiratory pressure (PEEP) and positive-pressure ventilation. Therefore, NP-NIV is speculated to have better respiratory support capacity than HFNT. However, the usefulness of NP-NIV for the acute phase is unknown, and no studies have compared it with HFNT.
This current study aimed to compare NP-NIV and HFNT in terms of tolerance and oxygenation after extubation in postsurgical patients.
Patients and methods
Patient population
This single-site retrospective cohort study enrolled consecutive patients admitted to a tertiary ICU in the Department of Anaesthesiology and Intensive Care, Faculty of Medicine, Oita University, Yufu, Japan between September 2018 and April 2020. Patients underwent NP-NIV or HFNT for the prevention of extubation failure after extubation with mechanical ventilation lasting at least 24 h after surgery. Patients were divided into two groups: those that underwent NP-NIV (NP-NIV group) and HFNT (HFNT group). Exclusion criteria were as follows: (i) non-operative patients; (ii) patients that did not require ventilatory support for more than 24 h postoperatively; (iii) patients <18 years; (iv) patients that received both NP-NIV and HFNT; (v) patients that underwent tracheostomy; (vi) post-head and neck surgery patients; (vii) patients with significant neuromuscular disorders; (viii) patients that had been using NIV or HFNT preoperatively.
The Ethics Committee of the Faculty of Medicine, Oita University, Yufu, Japan (no. 1682; Clinical Trials Registry, UMIN000045352; Retrospectively registered 2 September 2021, https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr_view.cgi?recptno=R000051788) approved the study and waived the need for informed consent from individual patients because of the retrospective nature of the study. All methods and regulation of this study were performed in accordance with the Declaration of Helsinki and the STROBE guidelines. 15
Criteria for extubation and reintubation
All patients underwent a spontaneous breathing trial with pressure support ventilation of 6 cmH2O and PEEP of 5 cmH2O for at least 30 min and were extubated if they met the following criteria: (i) no clinical signs of dyspnoea (retractions, thoraco-abdominal paradox, agitation, perspiration); (ii) saturation of percutaneous oxygen (SpO2) >90% at FiO2 0.5; (iii) respiratory rate <35 breaths/min; (iv) blood pressure not varying >20%. 16 Immediate reintubation was performed if the patient had any of the following major clinical events: (i) respiratory or cardiac arrest; (ii) respiratory pauses with loss of consciousness or gasping for air; (iii) massive aspiration; (iv) persistent inability to clear respiratory secretions; (v) heart rate <50 beats/min with loss of alertness; (vi) severe haemodynamic instability without response to fluid and vasoactive drugs. 17
NP-NIV and HFNT settings
Either NP-NIV or HFNT was used at the physician’s discretion if the following criteria were met: (i) partial pressure of oxygen/fraction of inspired oxygen (PaO2/FiO2) ratio <200 immediately before extubation or high risk of extubation failure; (ii) age >65 years; (iii) cause of intubation being heart failure; (iv) Acute Physiologic Assessment and Chronic Health Evaluation (APACHE) II score ≥12 at the time of extubation. 18 For NP-NIV, a Respironics Trilogy 202 system (Philips Respironics, Murrysville, PA, USA) was used as the ventilator and a DreamWear full face mask (Philips Respironics) was used as the nasal pillow-type interface with the humidifier (MR810 Respiratory Humidifier; Fisher & Paykel Healthcare, Auckland, New Zealand). NP-NIV was initially set at FiO2 0.5, PEEP 5 cmH2O and pressure support (PS) 5 cmH2O. Respiratory status was assessed hourly and the ventilator was adjusted to meet SpO2 > 90% and respiratory rate <35 breaths/min. The protocol of respiratory management with NP-NIV is shown in Figure 1a. When SpO2 > 90% or respiratory rate <35 breaths/min could not be met even with PEEP 10 cmH2O and PS 20 cmH2O, FiO2 was increased to 0.6. If respiratory status could not be maintained even under those conditions, reintubation was performed.

Respiratory management protocols for nasal pillow-noninvasive ventilation (NP-NIV) (a) and high-flow nasal therapy (HFNT) (b) used in a study to measure the tolerability and efficacy of NP-NIV compared with HFNT in postsurgical patients. Up and down arrows indicate increases and decreases. SpO2, saturation of percutaneous oxygen; FiO2, fraction of inspired oxygen; RR, respiratory rate; PEEP, positive end-expiratory pressure; PS, pressure support.
For HFNT, a Optiflow™ nasal high flow therapy system (Fisher & Paykel Healthcare) was used and HFNT was initially set at FIO2 0.5 and 40 l/min. As with the use of NP-NIV, respiratory status was assessed hourly and adjusted to meet SpO2 > 90% and respiratory rate <35 breaths/min. The protocol with HFNT is shown in Figure 1b. If respiratory status could not be maintained at 60 l/min and FiO2 0.6, reintubation was performed.
Data collection
The following data were collected retrospectively from the patients’ medical records: patient background, preoperative laboratory data (serum creatinine and serum total bilirubin levels, white blood cell count, haematocrit, platelet count, prothrombin time-international normalized ratio and activated partial thromboplastin time), APACHE II score, Sequential Organ Failure Assessment (SOFA) score at ICU admission, respiratory and circulatory parameters immediately before extubation, respiratory parameters at 1 and 24 h after extubation, NP-NIV or HFNT running time, and whether the patient was reintubated or not. To assess the tolerability of the interface, a four-point scale that was created based on information that could be extracted retrospectively was used: 1, wears the device without refusal; 2, frequently removes the device but accepts it only with instruction; 3, requires initiation or increase of sedative or analgesic medication for frequent removal of the device; 4, cannot accept the device and cannot continue to wear it. The higher the number on this scale, the lower the tolerability.
Outcome measures
The primary outcome was the tolerability of each interface after extubation. The secondary outcomes were respiratory parameters of each interface, device runtime, device weaning rate after 24 h and the incidence of skin injury associated with the interface. Skin injury was defined as a condition caused by the interface and requires medical examination or treatment by a dermatologist. In addition, reintubation rate within 72 h of extubation, duration of ventilation, ICU duration, length of hospital stay, in-hospital mortality and 60-day mortality were assessed according to the critical care ventilation trials outcome measures. 19
Statistical analyses
All statistical analyses were performed using Statflex Statistical Software (version 6.0; Artech, Osaka, Japan). Data are expressed as frequency (%) for categorical variables and as median (interquartile range) for continuous variables. Due to the retrospective design of the study, there was a baseline imbalance between patients on NP-NIV and HFNT. Therefore, propensity score matching was used to adjust the patient background, pre-extubation circulatory status and respiratory status. 20 The baseline patient variables used as candidates in the modelling process were considered clinically and statistically as confounders that may influence the choice of NP-NIV use. The propensity score for NP-NIV was calculated using multivariate logistic regression and included the following 29 independent variables: age, sex, height, weight, severity of illness at ICU admission (APACHE II and SOFA score), underlying disease (sepsis, immunosuppression, liver cirrhosis and chronic dialysis dependency), preoperative laboratory data, respiratory parameters immediately before extubation (respiratory rate, static lung compliance, PEEP, peak inspiratory pressure, PaO2/FiO2 ratio, partial pressure of arterial carbon dioxide [PaCO2] and pH) and circulatory parameters immediately before extubation (catecholamine index, heart rate and mean arterial pressure). The calliper width was set to 0.2 times the standard deviation of the propensity score and 1:1 nearest neighbour matching by non-restorative extraction was performed. Standardized differences were used to evaluate the balance of the covariates, wherein an absolute standardized difference (ASD) greater than 10% indicates a significant imbalance. 21 Continuous variables were compared using the Mann–Whitney U-test and categorical variables were compared using Fisher’s exact test. Friedman’s test was used to assess the difference between time-points for each group and the Wilcoxon signed-rank test with Bonferroni correction was used as a post hoc test. A P-value <0.05 was considered statistically significant. The results of a previous study showed that the difference in the PaO2/FiO2 ratio between face mask NIV and HFNT was 40–60. 22 Referencing these results, the required set of case controls was 16, assuming a difference of 50, a standard deviation of 50, an alpha error of 0.05 and a power of 80%.
Results
This single-site retrospective cohort study enrolled 110 consecutive patients that met the inclusion criteria over the study duration. After propensity score matching, the NP-NIV and HFNT groups were comprised of 19 patients each and the c-statistic was 0.819. The baseline characteristics of the study population are presented in Table 1. The severity of illness, as indicated by the APACHE II and SOFA scores, was significantly higher in the NP-NIV group than in the HFNT group (P =0.00139). After propensity score matching, the differences in baseline characteristics between the two groups were resolved, as the ASDs were less than 10% for all parameters. The respiratory and circulatory status before extubation is shown in Table 2 and the differences in pre-extubation status between the two groups were again resolved after propensity score matching.
Baseline demographic and clinical characteristics of the patients in the unmatched and propensity-matched groups in a retrospective analysis of the tolerability and efficacy of nasal pillow-noninvasive ventilation (NP-NIV) compared with high-flow nasal therapy (HFNT) in postsurgical patients.

Data presented as median (interquartile range) or n of patients (%).
PS, propensity score; ASD, absolute standardized difference; BMI, body mass index; APACHE, Acute Physiologic Assessment and Chronic Health Evaluation; SOFA, Sequential Organ Failure Assessment; CPB, cardiopulmonary bypass.
Respiratory and circulatory status before extubation in a retrospective analysis of the tolerability and efficacy of nasal pillow-noninvasive ventilation (NP-NIV) compared with high-flow nasal therapy (HFNT) in postsurgical patients.
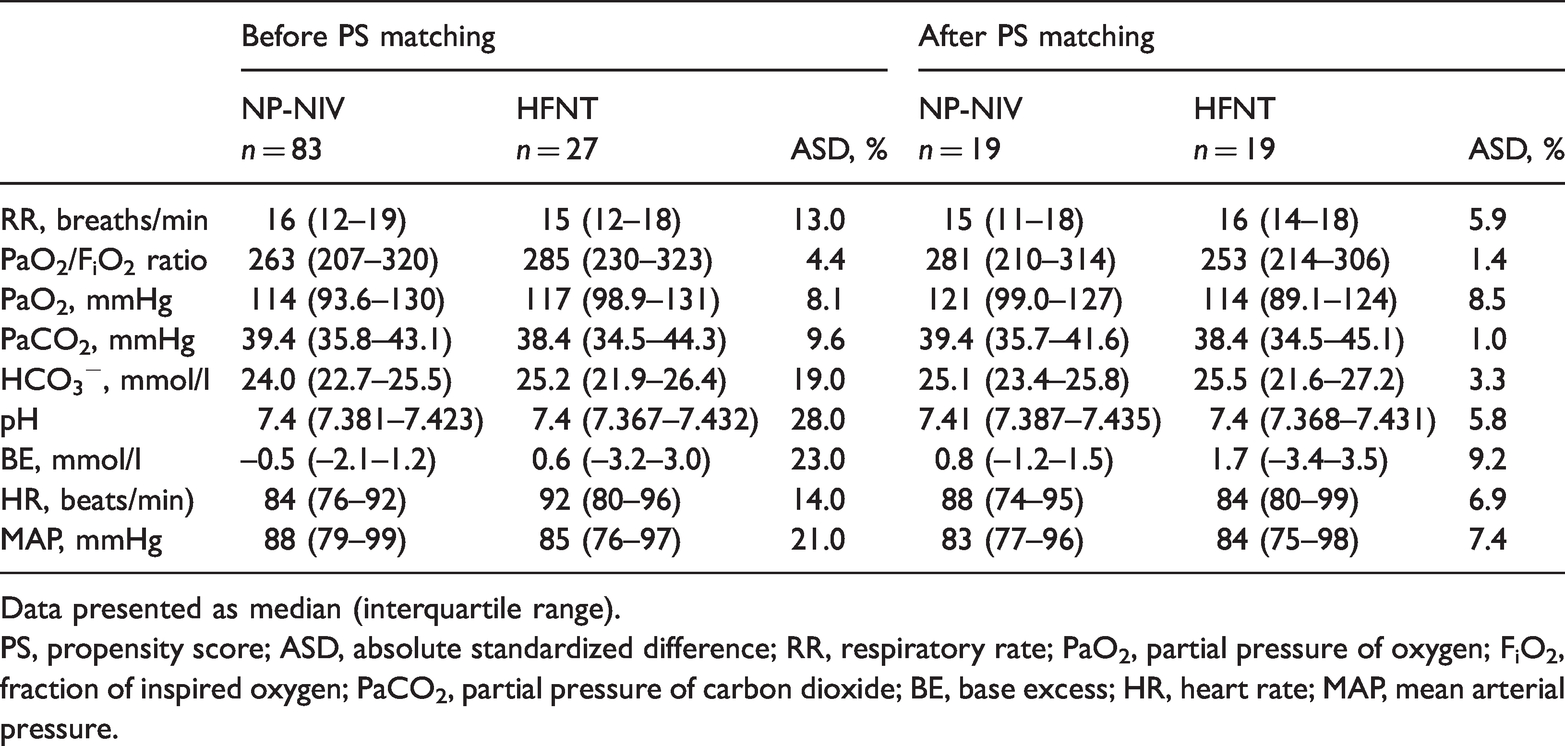
Data presented as median (interquartile range).
PS, propensity score; ASD, absolute standardized difference; RR, respiratory rate; PaO2, partial pressure of oxygen; FiO2, fraction of inspired oxygen; PaCO2, partial pressure of carbon dioxide; BE, base excess; HR, heart rate; MAP, mean arterial pressure.
The tolerability of NP-NIV and HFNT is presented in Table 3. There was no significant difference in the tolerability score between the groups and tolerability was good in all patients except for one reintubated patient in the NP-NIV group.
Tolerability of nasal pillow-noninvasive ventilation (NP-NIV) compared with high-flow nasal therapy (HFNT) in postsurgical patients.

Data presented as median (interquartile range) or n of patients (%).
No significant between-group difference (P ≥ 0.05); Mann–Whitney U-test.
The tolerability scores were defined as follows: 1, wears the device without refusal; 2, frequently removes the device but accepts it only with instruction; 3, requires initiation or increase of sedative or analgesic medication for frequent removal of the device; 4, cannot accept the device and cannot continue to wear it.
The changes in respiratory parameters immediately after extubation and 24 hours after extubation are shown in Figure. 2. The respiratory rate did not change significantly before and after extubation in the NP-NIV group but increased significantly after 1 and 24 h in the HFNT group (P < 0.05 for both comparisons). The respiratory rate of the NP-NIV group was significantly lower than that of the HFNT group at 1 h and 24 h after extubation (P < 0.001 for both comparisons). In both groups, the PaO2/FiO2 ratio was significantly lower at 1 h after extubation than before extubation (P < 0.05 for both comparisons). Only the NP-NIV group improved to the same level as before extubation at 24 h after extubation. The PaO2/FiO2 ratio of the NP-NIV group was significantly higher than that of the HFNT group at 1 and 24 h after extubation (P < 0.05 for both comparisons). The PaCO2 did not change significantly in either group. One patient was reintubated due to progressive hypoxia.
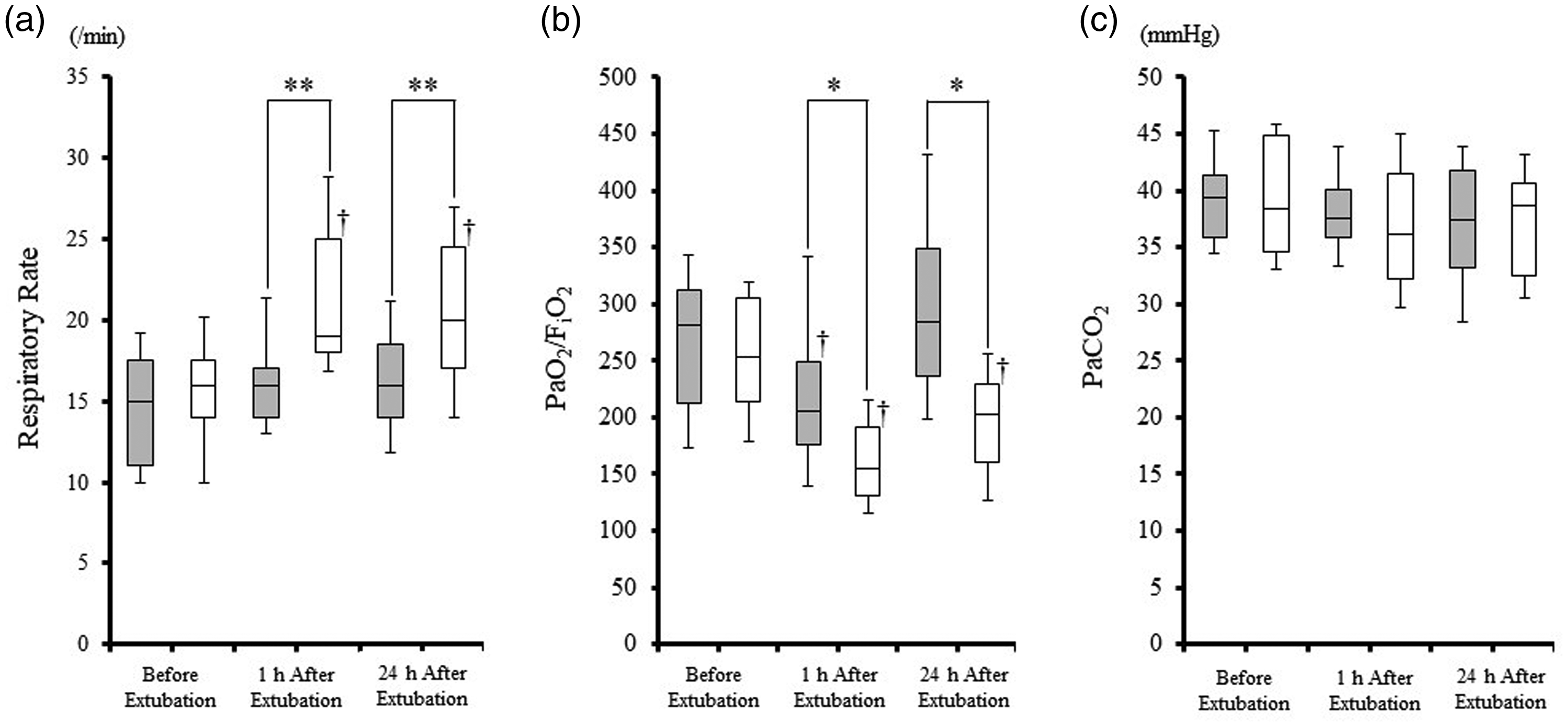
Changes in respiratory rate, partial pressure of oxygen/fraction of inspired oxygen (PaO2/FiO2) and partial pressure of carbon dioxide (PaCO2) before and after extubation in a retrospective analysis of the tolerability and efficacy of nasal pillow-noninvasive ventilation (NP-NIV) compared with high-flow nasal therapy (HFNT) in postsurgical patients. The box-and-whisker plots show the median (central black horizontal line), interquartile range (extremities of the box) and minimum/maximum outliers (error bars) of respiratory rate (a), PaO2/FiO2 (b) and PaCO2 (c) for the NP-NIV (grey) and HFNT (white) groups. *P < 0.05; **P < 0.001 comparisons of NP-NIV group and HFNT group at each time-point; †P < 0.05 comparison between before extubation and each time-point; Mann–Whitney U-test and Friedman’s test.
Clinical outcomes are shown in Table 4. The duration of NP-NIV was significantly shorter than that of the HFNT (P = 0.044). The duration of mechanical ventilation was also shorter in the NP-NIV group than in the HFNT group, but this difference was not statistically significant. No skin injury was observed in either group and there were no significant differences in reintubation or mortality rates between the two groups.
Clinical outcomes in a retrospective analysis of the tolerability and efficacy of nasal pillow-noninvasive ventilation (NP-NIV) compared withhigh-flow nasal therapy (HFNT) in postsurgical patients.

Data presented as median (interquartile range) or n of patients (%).
aContinuous variables were compared using the Mann–Whitney U-test and categorical variables were compared using Fisher’s exact test; NS, no significant between-group difference (P ≥ 0.05).
The tolerability scores were defined as follows: 1, wears the device without refusal; 2, frequently removes the device but accepts it only with instruction; 3, requires initiation or increase of sedative or analgesic medication for frequent removal of the device; 4, cannot accept the device and cannot continue to wear it.
Discussion
This current retrospective study identified two important findings. The first finding was that the tolerability of NP-NIV and HFNT was good in both groups and there was no significant difference between the two groups. The second finding was that NP-NIV was associated with better oxygenation and a lower respiratory rate 24 h after extubation as compared with HFNT.
First, there was no significant difference in the tolerance of NP-NIV and HFNT between the two groups and both were well accepted in the current study population. In addition, no complications such as skin injury, which is a problem with face mask NIV, were observed in the NP-NIV group. Typically, HFNT has better tolerability than NIV.7,23,24 This is because, for the NIV to function well, the interface and skin need to be sufficiently tight. Therefore, an excessively tight fit can lead to complications such as skin injury; and increased discomfort makes it difficult to use continuously. A previous study examined the preventive effect of NIV on postextubation respiratory failure in patients with chronic respiratory failure and found it to be more effective than COT. 13 However, approximately 10% of patients could not tolerate 24 h of continuous NIV use. 13 In many clinical studies that have examined the effects of NIV, NIV has been used intermittently;11,22,25 and the type of NIV used in those studies was mostly face mask NIV. The degree of tolerance depends on the type of interface because helmet-type NIV is more resistant than face mask NIV.26,27 Furthermore, a previous study compared the efficacy of face mask NIV and helmet NIV in patients with acute respiratory distress syndrome and found that the intubation rate was significantly lower in the helmet-type NIV group (18%) than in the face mask NIV group (61%). 28 Patients that used the helmet-type NIV maintained higher PEEP levels while the pressure support was lower. 28 Since the difference in tolerance between interface types correlates with the duration of device use, continuing ventilation at higher PEEP levels for a longer period using a better-tolerated interface may be more beneficial than increasing tidal volume with pressure support. NP-NIV is a device designed originally for chronic respiratory failure, making it suitable for long-term use, and it is the first choice for chronic respiratory failure. 29 Although the efficacy of NP-NIV in the acute postoperative period has not been evaluated to date, there may be many advantages of using NP-NIV in the acute period, considering that it can maintain higher PEEP with similar tolerability to HFNT.
Secondly, compared with HFNT, NP-NIV was more capable of preventing a decrease in oxygenation after extubation in the current study. The respiratory rate did not change after extubation in the NP-NIV group, but it increased significantly in the HFNT group. To date, no report has evaluated the oxygenation support capacity of NP-NIV in surgical ICU patients. These current results suggest that NP-NIV is superior to HFNT in supporting oxygenation in acute postoperative patients. Based on the significant change in respiratory rate between NP-NIV and HFNT, it can be speculated that it reflects the result of reduced work of breathing caused by the influence of respiratory support. The physiological functions of NIV and HFNT are different. 30 NIV increases pressure during inspiration that actively increases tidal volume and substantially decreases respiratory rate. 30 NIV is capable of controlling respiratory rate via the device settings. During HFNT, respiratory rate decreases spontaneously and primarily due to an improved gas exchange following the clearance of anatomical dead space. 31 As mentioned above, the patient population included in this current study was postoperative patients at a high risk of extubation failure. The efficacy of NIV in postoperative patients has already been demonstrated previously.17,32 In patients with acute respiratory failure after abdominal and thoracic surgery, NIV reduced the risk of intubation compared with COT. 4 Guidelines recommend the use of preventive NIV in patients at high risk of extubation failure.3,4 In contrast, HFNT has also been reported to improve the outcomes of postoperative hypoxemic patients and reduce the risk of pulmonary complications and reintubation.33,34 In a multicentre, randomized, non-inferiority study comparing HFNT with NIV in 830 patients after cardiothoracic surgery, 27.8% and 27.4% of patients with NIV and HFNT, respectively, were reintubated for mechanical ventilation, and HFNT was shown to be non-inferior to NIV. 22 In that study, a full-face mask NIV was used. 22 Although the outcome did not improve, the PaO2/FiO2 ratio after extubation was significantly higher in the NIV group. 22 Meanwhile, another study compared intermittent NIV plus HFNT with HFNT alone in patients at high risk of extubation failure and found that the rate of reintubation was significantly lower in the combined treatment group (12% versus 20%; P = 0.009). 35 It is speculated that the higher oxygenation capacity of NIV is due to the effect of higher PEEP alveolar recruitment. 36 Although NP-NIV fits more loosely than the face mask and has pressure loss due to mouth opening, it can provide stable PEEP due to leakage compensation by the ventilator. 37 In contrast, HFNT can generate positive pressure in the upper airway in proportion to the gas flow rate, but the positive pressure decreases significantly when the patient opens his or her mouth. A previous study measured nasopharyngeal pressure in postoperative patients at various flow levels using HFNT. 38 The pressure recorded during spontaneous breathing while using the HFNT correlated linearly with gas flow rate and was significantly higher when the patient breathed with the mouth closed. 38 This pressure exceeded 3 cmH2O at a gas flow rate of 50 l/min with the mouth closed and was <2 cmH2O with the mouth opened. 38 The advantage of an HFNT over an NIV concerning respiratory support is that the PEEP effect can be maintained for a long time owing to its better tolerability. 23 Furthermore, the PEEP levels produced by HFNT were low and limited. 39 NP-NIV is excellent for long-term use, similar to HFNT, and can maintain higher levels of PEEP for a longer period.14,29,37
The findings of this current study suggest that NP-NIV may be more advantageous for oxygenation than HFNT in patients at high risk in postextubation failure in the acute postoperative period. NP-NIV may have been more effective because most causes of acute postoperative respiratory failure are atelectasis and the giving of PEEP is more effective. This current study is a preliminary step to investigate the external validity of NP-NIV in the acute postoperative period.
This current study had several limitations. First, because this study was retrospective, the effect of unmeasured confounders could not be eliminated, even though attempts were made to negate bias through propensity score matching. Some parameters, such as PaO2 and base excess, were not statistically significant after matching, but the ASD was high, which may have modified the results. The APACHE II and SOFA scores of both groups before matching were significantly higher in the NP-NIV group. This implies that the physicians in charge tended to choose NP-NIV for more severely ill patients, which may suggest the existence of selection bias. Although there was no significant difference in the severity scores of the two groups after matching, the median APACHE II score was elevated in both groups: 19 and 18 in the NP-NIV group and HFNT group, respectively. Therefore, the patients to whom these current results can be applied are limited to those with much higher disease severity. Secondly, due to the small number of patients, the test power was insufficient to evaluate major outcomes such as reintubation and mortality. Thirdly, the method of evaluating tolerability used in this current study was an original development and its reliability has not yet been evaluated. Because of these limitations, randomized controlled trials are needed to confirm the effect of NP-NIV in improving clinical outcomes.
In conclusion, this current study showed that respiratory management with NP-NIV after extubation in postoperative patients provided better oxygenation support and a lower respiratory rate increase than that achieved with HFNT. The tolerability of NP-NIV and HFNT was good in both groups and there was no significant difference between the two groups.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221112777 - Supplemental material for Nasal pillow noninvasive ventilation versus high-flow nasal therapy after extubation in surgical intensive care patients: A propensity-matched cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221112777 for Nasal pillow noninvasive ventilation versus high-flow nasal therapy after extubation in surgical intensive care patients: A propensity-matched cohort study by Yoshifumi Ohchi, Yoshihide Kuribayashi, Takenori Makino, Norihisa Yasuda and Takaaki Kitano in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank Mr Ryo Saiko and Ms Ryoko Endo for their excellent technical assistance with data acquisition.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
