Abstract
Objective
Multidrug-resistant Klebsiella pneumoniae (MDR KP) bloodstream infections are a serious problem. The objective of this study was to investigate the effects of appropriate combination therapies on MDR KP bloodstream infections.
Methods
MDR KP strains isolated from clinical samples were assessed for antibiotic susceptibility using the broth microdilution method. Twenty consecutive MDR KP clinical isolates from patients with bloodstream infections were examined in this study. The experiments were conducted at the Bacterial Laboratory of Tongde Hospital from March to August 2021. Antibiotic combination tests were performed using the minimum inhibitory concentration (MIC) test, and the sum of the fractional inhibitory concentration was used to assess synergy.
Results
Following treatment with a combination of two antibiotic agents, the MIC50 and MIC90 values decreased compared with that before treatment. MIC50 decreased by at least 50%, with one value reduced to 6.25% of the pretreatment value. None of the antibiotic combinations were antagonistic. Combination of polymyxin B with rifampicin or tigecycline had a synergistic effect on 70% and 65% of the strains, respectively.
Conclusions
In vitro combination therapies with two active drug agents (polymyxin B plus rifampicin or tigecycline) had a better effect on MDR KP infections compared with that in other regimens.
Keywords
Introduction
The steady increase in extensively drug-resistant (XDR) and pandrug-resistant (PDR) strains of Klebsiella pneumoniae (KP) has become a significant clinical and global public health problem. 1 Many official reports by leading agencies, such as the World Health Organization, the Infectious Diseases Society of America, and the UK government (2015), have designated antibiotic resistance as a major problem affecting human health and health economy.2–4 Extensive KP resistance phenotypes can affect any patient where there is significant health care exposure. However, XDR and PDR KP strains mainly cause life-threatening infections, such as bloodstream infection and pneumonia, in critically ill patients. 5 There is a lack of effective antibiotics for XDR/PDR KP, which supports the urgent need to develop new therapies.
The dearth of novel antibiotics on the market is attributed to regulatory hurdles, high research costs, and low investment returns. 6 Combination therapies consisting of available drugs may enhance the antibacterial effects and may be promising therapies. In vitro data suggest antibiotic combinations can be effective treatments for XDR/PDR bacterial infections in individuals.7,8 However, additional research is needed to identify specific drug combinations to treat XDR/PDR bacterial infections.
In this study, combination therapies for treating bloodstream infections caused by MDR KP strains were evaluated. Emphasis was placed on identifying old antibiotic combinations with synergistic and bactericidal activity against MDR strains of KP in vitro. The combination regimens included several antibiotics, specifically imipenem (IPM), polymyxin B (POL-B), tigecycline (TGC), and rifampicin (RIF). IPM, POL-B, and TGC were used most commonly during the last decade to treat Gram-negative bacterial infections. Although RIF is not routinely used to treat Gram-negative Enterobacteriaceae infections such as Klebsiella, it plays a role in combination regimens for intracellular Gram-negative organisms such as Mycobacterium tuberculosis and Brucella. Certain issues, such as drug resistance, low serum drug concentrations, and side effects, now limit their clinical application. This work focuses on the anti-MDR KP effects that are caused by the combination of any two of these antibiotics.
Materials and methods
Materials
POL-B (MB1188) and RIF (MB1769) were provided by Dalian Meilun Biotechnology (Dalian, China). IPM and TGC were obtained from Merck Sharp & Dohme MSD (Rahway, NJ, USA) and Pfizer (New York, NY, USA).
Bacteria
MDR refers to strains that exhibit resistance to three or more antimicrobial drug classes. The strains were originally acquired from Tong De Hospital in Zhejiang Province and from Hangzhou Red Cross Hospital. Escherichia coli ATCC 25922, a bacterial strain that is susceptible to all antibiotics, was used as the quality control strain. Bacterial identification and antibiotic susceptibility testing were performed using the VITEK ®2 automated system (bioMérieux, Paris, France).
Twenty consecutive MDR KP clinical isolates from the bloodstream of infected patients were examined in this study. This research was reviewed by Ethics Review Committee of Hangzhou Red Cross Hospital which located in Hangzhou, Zhejiang Province, China. The Ethics Committee approved this study (reference number, 2022; Research Review No., 004). Verbal consent was obtained from each patient before the samples were analyzed.
Analysis of drug susceptibility and synergistic effects
The experiments were performed at the Bacterial Laboratory of Tongde Hospital from March to August 2021. The minimum inhibitory concentration (MIC) of the individual antibiotic agents was confirmed using broth microdilution tests, and the results of these tests were used to evaluate the synergistic effects. 9 The MIC required to inhibit the growth of 50% of organisms (MIC50) and the MIC required to inhibit the growth of 90% of organisms (MIC90) were also determined. The broth microdilution method was performed using the following test plate concentrations: 0.125, 0.25, 0.5, 1, 2, 4, 8, 16, 32, 64, and 128 mg/L. After inoculating the samples into the plates, all strains were incubated at 35°C for 16 to 20 hours before testing. The MIC was determined visually by examining the bottom of the plates. Antibiotic sensitivity was determined in accordance with the Clinical and Laboratory Standards Institute (CLSI) and European Committee on Antimicrobial Susceptibility Testing (EUCAST) standards. For IPM, the 2017 CLSI susceptibility breakpoints for Enterobacteriaceae are ≤ 1 μg/mL (susceptibility, S), 2 μg/mL (intermediate, I), and ≥4 μg/mL (resistance, R). For TGC, the 2019 EUCAST susceptibility breakpoints for Enterobacteriaceae are ≤ 0.5 μg/mL (S) and > 0.5 μg/mL (R). For POL-B, the EUCAST susceptibility breakpoints for Enterobacteriaceae are ≤ 2 μg/mL (S) and > 2 μg/mL (R). No established susceptibility breakpoints for RIF currently exist in the CLSI or EUCAST standards for KP. Therefore, we cautiously used breakpoints established for other similar organisms. The RIF breakpoints for Haemophilus influenzae are ≤ 1 µg/mL (S) and > 1 µg/mL (R). Because the breakpoints for RIF for Haemophilus influenzae are for prophylaxis for contacts of an invasive case and not for treatment, these breakpoints in our study were used only as an approximate frame of reference (Table 1).
Susceptibility breakpoints for each agent in this study.

The following six combinations of antibiotics were tested against 20 MDR KP isolates: IPM + RIF, IPM + TGC, IPM + POL-B, RIF + POL-B, TGC + POL-B, and TGC + RIF. The total FIC was calculated as follows: FIC = (MIC(Drug A) combination/MIC(Drug A) alone) + (MIC(Drug B) combination/MIC(Drug B) alone).
MIC, minimum inhibitory concentration; POL-B, polymyxin B; TGC, tigecycline; RIF, rifampicin; IPM, imipenem; MDR KP, multidrug-resistant Klebsiella pneumoniae; FIC, fractional inhibitory concentration; -, no data.
A fractional inhibitory concentration (FIC) index is a test to estimate the interaction between two or more drugs intended to be used in combination, and its range from 0.5 to 4 is commonly used to define additivity of the interaction. A FIC value ≤ 0.5 was considered to be a synergistic effect and 0.5 < FIC ≤ 4 was considered to be an indifferent effect. An FIC value >4 was classified as an antagonist. 10 The “checkerboard” experiment is a well-known and simple test to measure combined antimicrobial effects by calculating the FIC index. The FIC was calculated as follows: FIC = (MIC(Drug A) combination/MIC(Drug A) alone) + (MIC(Drug B) combination / MIC(Drug B) alone).
Statistical analysis
The association between FIC values and drug combinations was analyzed using cross-tabulation and chi-square testing. Statistical analysis was performed using Statistical Product Service Solutions (SPSS) v.22 (IBM Corp., Armonk, NY, USA), and p < 0.05 was considered to indicate statistically significant differences.
Results
Single antibiotic agent sensitivities and MICs
The MICs for IPM, RIF, POL-B, and TGC were assessed in 20 MDR KP clinical isolates. The MIC of IPM for the 20 strains ranged from 4 to 64 μg/mL, with a MIC50 and MIC90 of 32 and 64 μg/mL, respectively (Figure 1, Table 2). The MIC for RIF varied between 4 and 64 μg/mL, with MIC50 and MIC90 values of 16 and 32 μg/mL, respectively. The MIC for POL-B ranged between 0.25 and 4 μg/mL, with a MIC50 and MIC90 of 2 and 4 μg/mL, respectively. For TGC, the MIC ranged from 4 to 32 μg/mL, and the MIC50 and MIC90 were 16 and 32 μg/mL, respectively.

MICs of POL-B, TGC, RIF, and IPM against MDR KP isolates (n = 20).
MICs of polymyxin B, tigecycline, rifampicin, and imipenem against MDR KP isolates (n = 20).
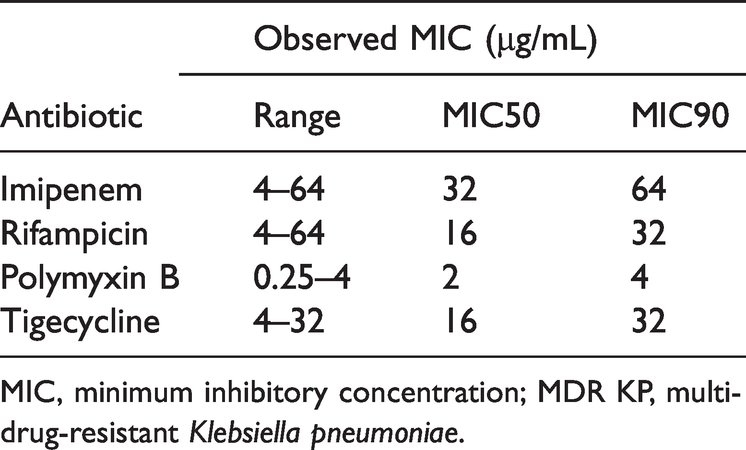
MIC, minimum inhibitory concentration; MDR KP, multidrug-resistant Klebsiella pneumoniae.
MICs from antibacterial agents used alone or in combination against MDR KP strains
The checkerboard titration analysis results for two-agent combinations indicated that lower doses of the drugs were required compared with the doses required for each drug alone (Table 3). Combinations involving IPM reduced the MIC50 by two to eight-fold compared with that of monotherapy. The MIC50 was decreased two to four-fold by RIF, four to 16-fold by POL-B, and four to eight-fold by TGC combinations compared with that of monotherapy (Table 3).
The MICs (μg/mL) of four antibiotics against MDR KP strains alone or in combination.

MIC, minimum inhibitory concentration; MDR KP, multidrug-resistant Klebsiella pneumoniae; POL-B, polymyxin B; TGC, tigecycline; RIF, rifampicin; IPM, imipenem.
FICs for antibiotic combinations
Table 4 shows the total FICs for the various antibiotic combinations. No antagonistic effect was observed among any of the combination regimens. Synergistic bactericidal activity (FIC ≤ 0.5) was demonstrated in most of the MDR KP strains treated with RIF and POL-B (14/20; 70%) or TGC and POL-B (13/20; 65%). These two combinations showed the greatest synergistic effect compared with that of each of the other combinations against MDR KP isolates (p < 0.05). The combination of RIF with TGC or IPM showed a strong indifferent effect (0.5 < FIC ≤ 4) against 80% (16/20) or 85% (17/20) of strains, respectively.
Interaction effects of the antibiotic combinations against 20 MDR isolates.

*, **, synergistic effect compared with that of each of the other combinations, p < 0.05.
FIC ≤ 0.5, synergistic effect; FIC 0.5 < FIC ≤ 4, indifferent effect; FIC >4, antagonist. 10
MDR, multidrug-resistant; FIC, fractional inhibitory concentration; POL-B, polymyxin B; TGC, tigecycline; RIF, rifampicin; IPM, imipenem.
Discussion
Understanding which antibiotic agents produce reliable synergistic interactions is essential for selecting the best combination regimens. Bloodstream infections by MDR Enterobacteriaceae are particularly concerning given the rapid degree of deterioration and high mortality rate that accompany these infections.
We assessed the ability of different drug combinations to lower the MIC against 20 MDR KP isolates. All 20 strains were resistant to TGC, RIF, and IPM monotherapies. Following treatment with the antibiotic combinations, the MIC50 and MIC90 decreased significantly compared with the pre-treatment values. In in vitro studies, Tängdén et al. concluded that antibiotic combinations demonstrated synergistic bactericidal effects against MDR Gram-negative bacteria resistant to the individual drugs. 11
Our results also indicated that the POL-B-based regimens, especially those with RIF and TGC, had strong synergistic effects. POL-B lowered the MIC50 of RIF and that of TGC by four- and eight-fold, respectively. Similarly, RIF and TGC lowered the MIC50 of POL-B by 16- and eight-fold, respectively. POL-B disrupts membrane integrity by displacing extracellular ions by binding to negatively charged lipopolysaccharide, which causes cell lysis. 12 POL-B acts primarily on the Gram-negative bacterial cell wall, leading to rapid permeability changes in the cytoplasmic membrane. 13 POL-B also has anti-endotoxin activity, which results in rapid bactericidal effects on many Gram-negative bacteria. 14 We speculate that our experimental results can be explained by these activities of POL-B. Further animal and clinical studies are needed to clarify the pharmacodynamics of this synergistic interaction in vivo as well as the limitations of these combinations.
The FIC values indicate high rates of synergism for POL-B in combination with RIF or TGC in MDR KP strains. The effect of this synergism was statistically significant compared with that of the other combinations. Previous studies found similar synergistic activities for POL-B-based combinations against KP strains. Tascini et al. 15 reported that the most reliable synergistic inhibition against all colistin-resistant KP was the in vitro combination of colistin and RIF.
Clinical studies indicate that combination therapies consisting of POL-B could reduce the mortality of patients with bloodstream MDR KP infections. 16 The combination of colistin with another active antibiotic improved survival in many patients infected with carbapenem-resistant Enterobacteriaceae. 17 POL-B is associated with nephrotoxicity and neurotoxicity, particularly when used alone, and thus, it is not an optimal first-line treatment due to poor clinical outcomes. Our results showed that POL-B-based combinations in our study, especially those with TGC and RIF, had a lower MIC50 and MIC90 for POL-B compared with those of monotherapy. The significant presence of synergy in these combinations indicates a better treatment choice for MDR KP.
RIF is a semisynthetic, broad-spectrum antibiotic that kills bacteria by inhibiting RNA polymerase, and it is commonly used to treat tuberculosis. 18 Gram-positive and -negative bacteria are similarly sensitive to RIF, with higher MICs reported in Gram-negative compared with those of Gram-positive bacteria because of their low outer membrane penetration. 19
The combination of RIF and colistin, a well-known membrane permeabilizer, demonstrated that perturbation of the Pseudomonas aeruginosa outer membrane potentiates RIF antimicrobial activity, 20 which is consistent with the results of our study. Following POL-B perturbation of the MDR KP membrane, RIF rapidly penetrated the outer membrane and impaired bacterial protein synthesis.
TGC is a glycylcycline antibiotic that inhibits protein translation by binding strongly to the 30S ribosomal subunit. 21 The limitations of TGC include its low serum concentration and low epithelial lining fluid penetration in mechanically ventilated patients, 17 which has hampered its clinical application. The combination of TGC and POL-B disrupts the bacterial membrane integrity, which is caused by POL-B, and this allows TGC entry and accumulation. The combination of TGC and POL-B resulted in the lowest blood culture positivity rates for KP carbapenemase-2-producing KP, which is consistent with the findings by Toledo et al. 22
Limitations of this study include the relatively small sample size, which precluded the evaluation of specific drug combinations. Molecular testing was not performed on all isolates to identify synergistic activity mechanisms. Finally, in our isolates, we did not evaluate susceptibility to ceftazidime/avibactam, which is a popular drug used to treat a wide range of MDR bacterial infections.
In conclusion, our data suggest that adding RIF or TGC favorably enhances POL-B activity against MDR KP strains. Our in vitro susceptibility and synergy test results suggest that these drug combinations are promising initial treatment regimens for patients with MDR KP bloodstream infections. Further studies are needed to evaluate other specific drug combinations.
