Abstract
Objective
To explore the association between CpG island methylation in the promoter region of RELN and positive (type I) and negative (type II) types of schizophrenia, and investigate serum interleukin (IL)-1β, IL-6, and myelin basic protein (MBP) in schizophrenia.
Methods
Levels of CpG island methylation in the promoter region of RELN were detected in peripheral blood of patients with schizophrenia (experimental group) and healthy individuals (control group), and serum IL-1β, IL-6, and MBP were measured.
Results
The positive rate of CpG island methylation in the promoter region of RELN was higher in the experimental group than in the control group; however, there were no significant differences between type I and II patients. There were differences in Positive and Negative Syndrome Scale (PANSS) scores and serum IL-1β, IL-6, and MBP between type I and II patients. Furthermore, there were positive correlations between serum IL-1β, IL-6, and MBP and PANSS scores (negative symptoms) in type II patients.
Conclusion
CpG island methylation in the promoter region of RELN was associated with schizophrenia, but not with its clinical type. There may be different pathological mechanisms in type I and II schizophrenia, and type II schizophrenia may be associated with serum IL-1β, IL-6, and MBP.
Introduction
The neurodevelopmental disorder hypothesis is one of the most important pathological hypotheses of schizophrenia. 1 Reelin (encoded by RELN) is an extracellular glycoprotein composed of 3461 amino acid residues, with a molecular weight of approximately 420 kD. It is involved in signal regulation in neurotransmission, information formation, and synaptic plasticity, and regulates the migration and localization of neural stem cells during brain development. 2 In schizophrenia, the epigenetic process is mainly focused around the modification of DNA methylation and histones. It has been reported that RELN expression is often altered in both central and peripheral tissues of patients with schizophrenia, and the regulation of DNA methylation is an important factor that affects RELN expression. 3 Most genes are methylated on CpG islands in the promoter region. CpG island methylation in the promoter region of RELN is reportedly more severe when RELN is silenced. 4 Moreover, a previous study has demonstrated that an imbalance in the allelic expression of RELN occurs in schizophrenia. 5 However, previous studies have investigated schizophrenia as a disease unit even though the phenotypes of each clinical type may differ from one another, thus leading to inconsistent results.
Schizophrenia was traditionally classified into paranoid, catatonic, disorganized, residual, and undifferentiated subtypes (in the updated fourth Diagnostic and Statistical Manual of Mental Disorders [DSM-IV TR] only; the DSM-5 does not support schizophrenia classifications) but this classification system has little relationship with patient etiology. At present, some researchers divide schizophrenia into type I (positive symptoms) and type II (negative symptoms) based on patient etiology. It has been speculated that type I schizophrenia is caused by altered brain biochemical factors, while type II schizophrenia is caused by injury during brain development or altered brain structures. 6 In the present study, schizophrenia was divided into type I and type II subtypes in an attempt to increase the homogeneity of the study samples.
Previous studies have demonstrated that neuroimmune abnormalities and nerve injuries exist in patients with schizophrenia. 7 It has also been reported that nerve injury causes DNA methylation changes at 8% of CpG sites. 8 The serum protein factors interleukin (IL)-1β and IL-6 are important neuroimmune markers. IL-1β is a cytokine that is secreted by lymphocytes and non-lymphocytes and participates in various physiological processes, such as immunoregulation and neuroendocrine responses. 9 Furthermore, IL-1β has been reported to correlate with psychiatric symptoms in patients with schizophrenia, and may reflect the degree of this disease to a certain extent; it is also speculated that IL-1β may influence neurotransmitters, thus reflecting the psychiatric symptoms of patients with schizophrenia. 10 IL-6 is a proinflammatory factor that is mainly synthesized and secreted by macrophages, lymphocytes, and epithelial cells. When an inflammatory response is induced directly or indirectly in the nervous system, high levels of IL-6 affect the cognitive function of patients with schizophrenia. 11 Moreover, patients with schizophrenia have higher levels of serum IL-6 compared with healthy controls and individuals with euthymic bipolar disorder. 12
Myelin basic protein (MBP) is a protein of the central nervous system. When myelin fracture and disintegration occur in the central nervous system because of direct mechanical damage (namely primary brain injury, such as cerebral contusion or intracranial hematoma, or as the result of a brain tumor), the myelin component MBP is released into bodily fluids, thus causing secondary brain injury. 13 Studies have shown that serum MBP is a prognostic marker of brain injury. 14 Additionally, changes in blood MBP levels may reflect the degree of glial injury in the central nervous system; however, few clinical studies have investigated whether changes in MBP may be an index for the development of schizophrenia.
The present study aimed to explore the associations between CpG island DNA methylation in the promoter region of RELN and the positive and negative types of schizophrenia, and to investigate changes in the serum protein factors IL-1β, IL-6, and MBP in patients with schizophrenia.
Methods
This is a prospective clinical research report. The study was approved by the Research Ethical Committee of The Second People’s Hospital of Lishui, China (No. 20170107-1). Signed consent was obtained from each participant. All participant details have been de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 15
Participant selection
Patients with schizophrenia who were admitted to The Second People's Hospital of Lishui (Zhejiang, China) from January 2017 to December 2019 were selected consecutively as the experimental group. The inclusion criteria of the experimental group were as follows: 1) met the diagnostic criteria of schizophrenia in the DSM-IV; 2) aged 18 to 65 years; 3) first episode of schizophrenia; 4) did not use antipsychotic drugs or had not used them for at least 2 weeks prior to admission; 5) Positive and Negative Syndrome Scale (PANSS) score ≥60; and 6) had an informed consent signed by a legal guardian. The exclusion criteria were as follows: 1) met any other diagnostic criteria of the DSM-IV; 2) had a clear risk of suicide or endangering their own or others' safety; 3) had severe physical diseases; and 4) were pregnant or lactating women.
Healthy individuals who underwent a physical examination at The Second People's Hospital of Lishui from January 2017 to December 2019 were screened into the control group. The inclusion criteria of the control group were as follows: 1) age and sex matched to the experimental group; 2) had no family history of mental illness; 3) had no mental illness according to the Brief Psychiatric Rating Scale (i.e., had a total score <35); and 4) signed their informed consent. The exclusion criteria were as follows: 1) had severe physical diseases; or 2) were pregnant or lactating women.
Classification of type I and type II schizophrenia
The psychotic symptoms of patients with schizophrenia were assessed using PANSS. Psychiatric interviews were performed by psychiatrists who participated in scale training and met qualification requirements, and scores were obtained by combining these data with relevant information provided by family members. The PANSS scores of positive symptoms, PANSS scores of negative symptoms, and total PANSS scores were compared between patients with type I (with mainly positive symptoms) and type II (with mainly negative symptoms) schizophrenia.
Assessment of CpG island DNA methylation in the RELN promoter region
To assess CpG island DNA methylation, 5 mL of peripheral venous blood was taken from each participant and placed into a vacuum blood collection tube containing ethylenediaminetetraacetic acid as an anticoagulant, and was then stored at −20°C. The levels of CpG island methylation in the RELN promoter region in peripheral blood were detected using the iPLEX Gold Assay (Agena Bioscience, San Diego, USA) on the MassARRAY platform (Agena Bioscience). Samples with specific polymerase chain reaction (PCR) amplification products by both methylated and non-methylated primers were identified as positive (i.e., CpG island methylated) specimens. Only the samples of PCR products amplified by unmethylated primers were identified as negative (CpG island unmethylated). The positive rates of CpG island DNA methylation were compared between the experimental and control groups and between patients with type I and II schizophrenia.
Serum protein factor measurements
To measure the levels of serum protein factors IL-1β, IL-6, and MBP, 3 mL of venous blood was collected from each participant after 12 to 14 hours of fasting. The levels of IL-1β, IL-6, and MBP were detected using enzyme-linked immunosorbent assay. The IL-1β, IL-6, and MBP detection kits were purchased from Shanghai Enzyme-linked Biotechnology Co., Ltd., China.
Statistical analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY, USA). Count data are expressed as n (%), and comparisons between groups were performed using the chi-squared test. Measurement data are expressed as the mean ± standard deviation, and comparisons between two groups were conducted using the independent sample t-test, comparisons between three groups were performed using one-way analysis of variance, and pairwise (post-hoc) comparisons were carried out using the least significant difference method. Pearson correlation analysis was used to analyze the relationships between serum protein factor levels and PANSS scores. For all tests, differences were considered significant when P < 0.05.
Results
A flowchart of the study is shown in Figure 1. In the experimental group, there were 200 participants consisting of 97 (48.50%) men and 103 (51.50%) women. Participants were aged 18 to 64 (39.77 ± 9.50) years old and had disease durations of 8 to 43 (24.03 ± 7.35) months; 116 (58.00%) were type I patients and 84 (42.00%) were type II patients.
Flowchart of the study.
In the control group, there were 200 participants consisting of 94 (47.00%) men and 106 (53.00%) women. Participants were aged 19 to 63 (39.04 ± 9.19) years old. There were no significant differences in sex or age between the experimental and control groups (Table 1).
General information of the experimental and control groups.

SD, standard deviation.
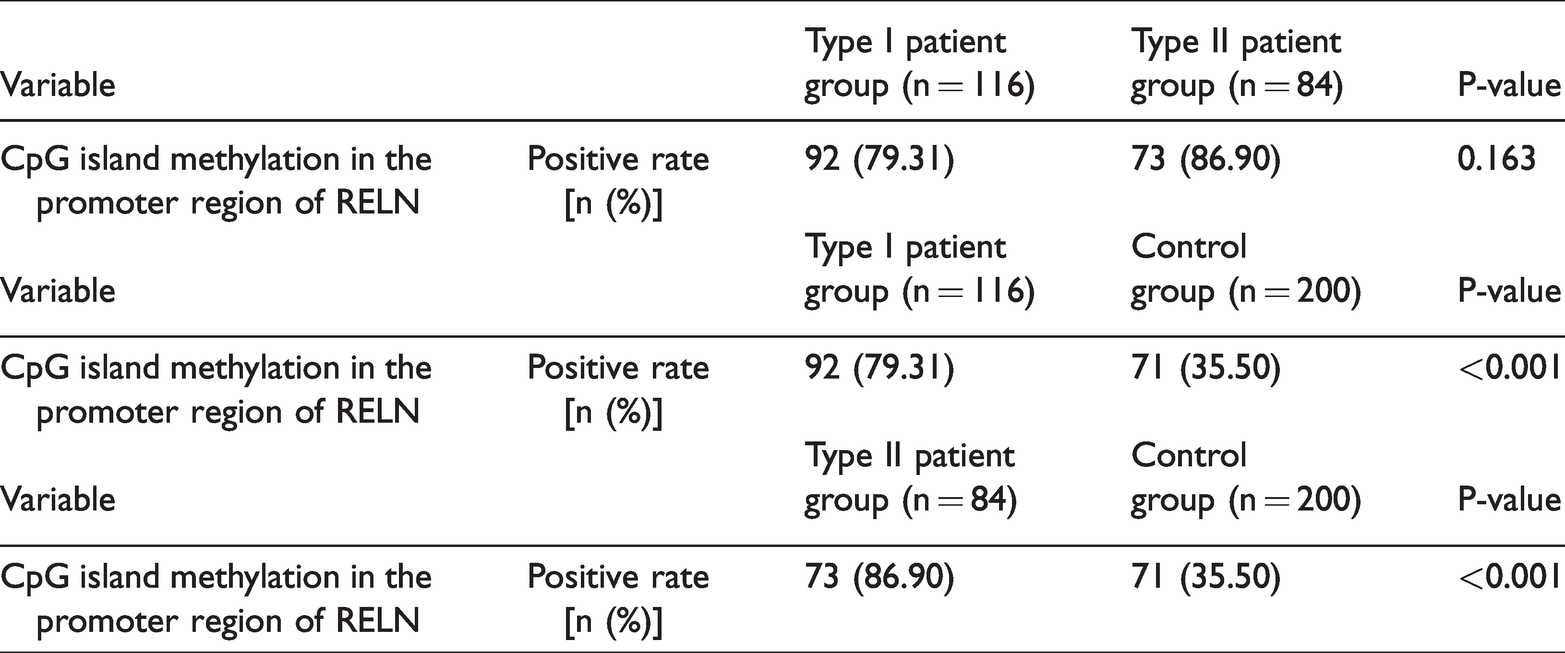
The positive rate of CpG island methylation in the promoter region of RELN in the experimental group (165 cases, 82.50%) was significantly higher than that of the control group (71 cases, 35.50%; P < 0.001, Table 1).
Among the 116 type I patients, the positive rate of CpG island methylation in the promoter region of RELN was 92 (79.31%), while among the 84 type II patients, the positive rate was 73 (86.90%). There was no significant difference between the two groups (Table 2), but the positive rates of both were higher than that of the control group (both P < 0.001, Table 2).
Positive rate of CpG island methylation in the promoter region of RELN.
The differences in PANSS scores of positive symptoms, PANSS scores of negative symptoms, and total PANSS scores were significantly different between the type I and II patient groups (all P < 0.001, Table 3).
Comparison of PANSS scores between the type I and II patient groups.

All data are presented as the mean ± standard deviation. PANSS, Positive and Negative Syndrome Scale.
There were also significant differences in the levels of serum protein factors IL-1β, IL-6, and MBP among the type I patient, type II patient, and control groups (all P < 0.001, Table 4). The levels of serum IL-1β, IL-6, and MBP were lower in the control group than in the type I (all P < 0.001, Table 4) and type II (all P < 0.001, Table 4) patient groups. Moreover, the levels of serum IL-1β, IL-6, and MBP were lower in the type I patient group than in the type II patient group (all P < 0.001, Table 4).
Levels of serum protein factors IL-1β, IL-6, and MBP in each of the three groups.

All data are presented as the mean ± standard deviation.
IL, interleukin; MBP, myelin basic protein; P1, type I patient group vs. type II patient group; P2, type I patient group vs. control group; P3, type II patient group vs. control group.
There were no significant correlations between the levels of serum IL-1β and IL-6 and the PANSS scores of positive symptoms, PANSS scores of negative symptoms, and total PANSS scores in the type I patient group (Table 5). However, there was a weak negative correlation between the levels of serum MBP and PANSS scores of positive symptoms in the type I patient group (r = −0.336, P = 0.041, Table 5).
Correlations between serum protein factors and PANSS scores in the type I and II patient groups.

IL, interleukin; MBP, myelin basic protein; PANSS, Positive and Negative Syndrome Scale.
There were positive correlations between IL-1β, IL-6, and MBP and the PANSS scores of negative symptoms in the type II patient group (r = 0.783, P < 0.001; r = 0.791, P < 0.001; r = 0.725, P < 0.001, Table 5), but no significant correlations between IL-1β, IL-6, and MBP and the PANSS scores of positive symptoms and total PANSS scores (Table 5).
Discussion
A previous experimental study reported that RELN participates in the migration and positioning of dopaminergic neurons in the developing brain. 18 The epigenetic changes in RELN that are involved in the development of dopaminergic neurons may fit the developmental theory of schizophrenia. RELN DNA methylation may contribute to the pathogenesis of schizophrenia. 3 In the present study, the positive rate of CpG island methylation in the promoter region of RELN was significantly higher in the peripheral blood of patients with schizophrenia than in that of the control group, which is largely consistent with previously reported results.19,20 These findings suggest that patients with schizophrenia have abnormal CpG island methylation in the RELN promoter region, which may be involved in the pathogenesis of schizophrenia.
Further analysis in the current study revealed no significant differences in the positive rate of CpG island methylation in the promoter region of RELN between type I and type II patients, but the rates of both were higher than that of the control group. This suggests that CpG island methylation in the promoter region of RELN is associated with schizophrenia but not with any specific clinical type of this disease. However, this finding needs to be confirmed in studies with larger samples.
Immune dysfunction mediated by cytokines occurs in patients with schizophrenia. The results of previous research indicate that the impaired activation of monocytes is closely related to increased IL-1β levels. 21 Higher levels of IL-6 may reflect the unique syndrome of patients with schizophrenia. 22 Additionally, serum MBP may be used to predict the prognosis of patients with schizophrenia. 23 It has been reported that reduced MBP mRNA in schizophrenia replicates findings in other brain regions and is consistent with oligodendrocyte involvement in the disorder. 24 Moreover, there is reduced MBP expression in the entorhinal cortex in schizophrenia, and this implies deficits in axonal myelination and disturbed connectivity. 25 One study demonstrated that the increased activity of MBP-hydrolyzing antibodies is caused by the activation of humoral immunity in schizophrenia. 26
In the present study, the levels of serum IL-1β, IL-6, and MBP were lower in the control group than in the type I and II patient groups. Previous studies have also reported that the levels of serum IL-1β and IL-6 are higher in patients with schizophrenia than in healthy individuals. 27 Furthermore, another study has demonstrated that the inflammatory process mediated by IL-6 is closely related to immunological abnormalities in schizophrenia. 28
In the current study, we revealed through further analysis that the levels of serum IL-1β, IL-6, and MBP were lower in the type I patient group than in the type II patient group. A previous study has also reported that serum IL-1β and IL-6 levels are higher in patients with schizophrenia characterized by negative symptoms than in those characterized by positive symptoms. 29 This suggests that type II patients (with schizophrenia characterized by negative symptoms) have more severe neuroimmune and nerve damage.
Further correlation analysis in the current study revealed that the levels of serum IL-1β, IL-6, and MBP were positively correlated with the PANSS scores of negative symptoms in type II patients (characterized by negative symptoms); however, there were no significant correlations between IL-1β or IL-6 levels and the PANSS scores of positive symptoms, PANSS scores of negative symptoms, or total PANSS scores in the type I patient group. These findings suggest that the pathological basis of negative symptoms may be more closely related to the inflammatory response and damage of nerve cells, and indicate that different clinical manifestations of schizophrenia may have different pathological bases. Furthermore, a previous study has also reported that patients with negative symptoms are likely to have more serious neuroimmune disturbances. 29
One limitation of our study was that we did not further investigate the associations between CpG island methylation in the promoter region of RELN and IL-6, IL-1β, or MBP. We will study such associations in the future.
Conclusion
CpG island methylation in the promoter region of RELN was associated with schizophrenia but not with any specific clinical type of this disease. There may be different pathological mechanisms underlying type I and type II schizophrenia, and type II schizophrenia may be inherently correlated with the serum protein factors IL-1β, IL-6, and MBP.
Footnotes
Author contributions
JZ: Acquired data, drafted the manuscript, revised the manuscript.
DZ, TY, WC, and HX: Research design, acquired data, drafted the manuscript.
YX: Drafted the manuscript; read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
