Abstract
Objective
To evaluate salivary interleukin (IL)-1β levels in patients with psoriasis, before and after treatment with tumour necrosis factor (TNF)-α inhibitors.
Methods
In this pilot study, salivary secretions were collected from patients with psoriasis and untreated healthy control subjects at baseline, and from patients after 12 weeks’ treatment with TNF-α inhibitors. IL-1β levels were determined in saliva samples via enzyme-linked immunosorbent assays, undertaken before and after TNF-α inhibitor treatment. Psoriasis-specific analysis of disease severity and activity were also undertaken.
Results
At baseline, patients (n = 25) had significantly higher salivary IL1β levels than controls (n = 20). In patients with psoriasis, TNF-α inhibitor treatment resulted in significantly reduced IL1β levels compared with baseline, but IL1β levels remained significantly higher than in control subjects even after treatment. There was a positive correlation between IL-1β levels, psoriasis activity and disease index score after TNF-α inhibitor treatment.
Conclusion
Saliva is a valid noninvasive tool for monitoring inflammation in psoriasis. TNF-α inhibitor treatments appear to interfere with the oral inflammatory process in patients with psoriasis.
Introduction
Psoriasis is a chronic immune-mediated inflammatory disease with an estimated prevalence of up to 3% in the general population. 1 The proinflammatory milieu plays a crucial role in the immunopathogenesis of psoriasis and links this disease to other inflammatory conditions, including metabolic syndrome, cardiovascular disease, periodontitis and inflammatory bowel disease.2–5
Saliva, a biological fluid containing important biological markers, is easily collected and stored. Salivary biomarkers have been identified in systemic pathologies including cardiovascular disease, renal failure and malignancy.6,7 We have profiled the inflammatory cytokines present in salivary secretions from patients with psoriasis. 8
The aim of this pilot study was to evaluate the change in salivary interleukin (IL)-1β concentration in patients with psoriasis after treatment with tumour necrosis factor (TNF)-α inhibitors.
Patients and methods
Study population
This pilot study recruited consecutive patients with stable chronic plaque psoriasis who were attending the Dermatology Clinic, Polytechnic University of Marche Region, Ancona, Italy, between January 2014 and March 2014. Inclusion criteria were no periodontal involvement and no previous biological therapy. Age, sex and periodontal status-matched control subjects were recruited from patients with nonpsoriatic dermatological conditions who were attending the same clinic. A trained oral pathologist (A.S.) evaluated the oral cavity of each participant. Patients with psoriasis then began standard regimens of TNF-α inhibitor treatment.
The study was conducted in accordance with the Declaration of Helsinski, 9 and all study participants provided oral informed consent prior to enrolment.
Sample collection and analysis
Salivary secretion samples were collected at baseline (in patients and controls), and after 12 weeks of TNF-α inhibitor treatment (in the patient group). Samples were collected between 09:00 h and 11:00 h to control for circadian modification of salivary biomarkers, using a standardized collection method (Salivette®; Sarstedt, Nümbrecht, Germany), and stored at –80°C until use. IL-1β levels were evaluated via an enzyme-linked immunosorbent assay kit (Quiagen, Venlo, Netherlands) and expressed as absorbance units. Psoriasis Area and Severity Index (PASI) scores 10 were determined for each patient at baseline and at 12 weeks. Patients were stratified according to baseline psoriasis severity: mild (PASI ≤ 10); moderate (PASI > 10– ≤ 20); or severe (PASI > 20).
Statistical analysis
Data were presented as mean ± SD. For continuous variables, normality of distribution was verified using Kolmogorov–Smirnov test and between-group comparisons were made using Kruskal–Wallis test. Linear regression analysis was used to model the relationship between tested variables. Patients were stratified according to baseline psoriasis severity: mild (PASI ≤ 10); moderate (PASI > 10– ≤ 20); severe (PASI > 20). All data were analyzed using GraphPad Prism® version 5.0 and QuickCalcs® (both GraphPad Software Inc., La Jolla, CA, USA). P-values < 0.05 were considered statistically significant.
Results
The study included 25 patients with psoriasis (15 male/10 female; mean age 50.2 ± 16.5 years; age range 34–66 years) and 20 control subjects (12 male/eight female; mean age 50.4 ± 15.5 years; age range 35–66 years). A total of 15 patients were treated with adalimumab (40 mg/every other week for 12 weeks); ten received etanercept 50 mg/bi-weekly for 12 weeks).
Psoriasis Area Severity Index (PASI) 10 scores in patients with psoriasis stratified according to baseline disease severity, before and after 12 weeks of tumour necrosis factor-α inhibitor treatment (n = 25).

Data presented as mean ± SD.
Baseline PASI ≤ 10.
Baseline 10 < PASI ≤ 20.
Baseline PASI > 20.
P < 0.05 vs baseline; Kruskal–Wallis test.
At baseline, patients had significantly higher salivary IL1β levels than controls (2.12 ± 1.16 vs 0.49 ± 0.17; P < 0.0001). TNF-α inhibitor treatment resulted in significantly reduced IL1β levels compared with baseline (1.15 ± 0.78 vs 2.12 ± 1.16; P < 0.0001), but IL1β levels remained significantly higher in patients with psoriasis than in healthy controls (1.15 ± 0.78 vs 0.49 ± 0.17; P = 0.0002) (Figure 1).
Salivary interleukin (IL)-1 β levels in patients with psoriasis and controls. Tumour necrosis factor-α inhibitor treatment significantly reduced IL1β levels, compared with baseline (T0); however, IL1β levels remained significantly higher in patients with psoriasis than in healthy controls at the end of the 12-week treatment period (T12); ***P < 0.001; Kruskal–Wallis test.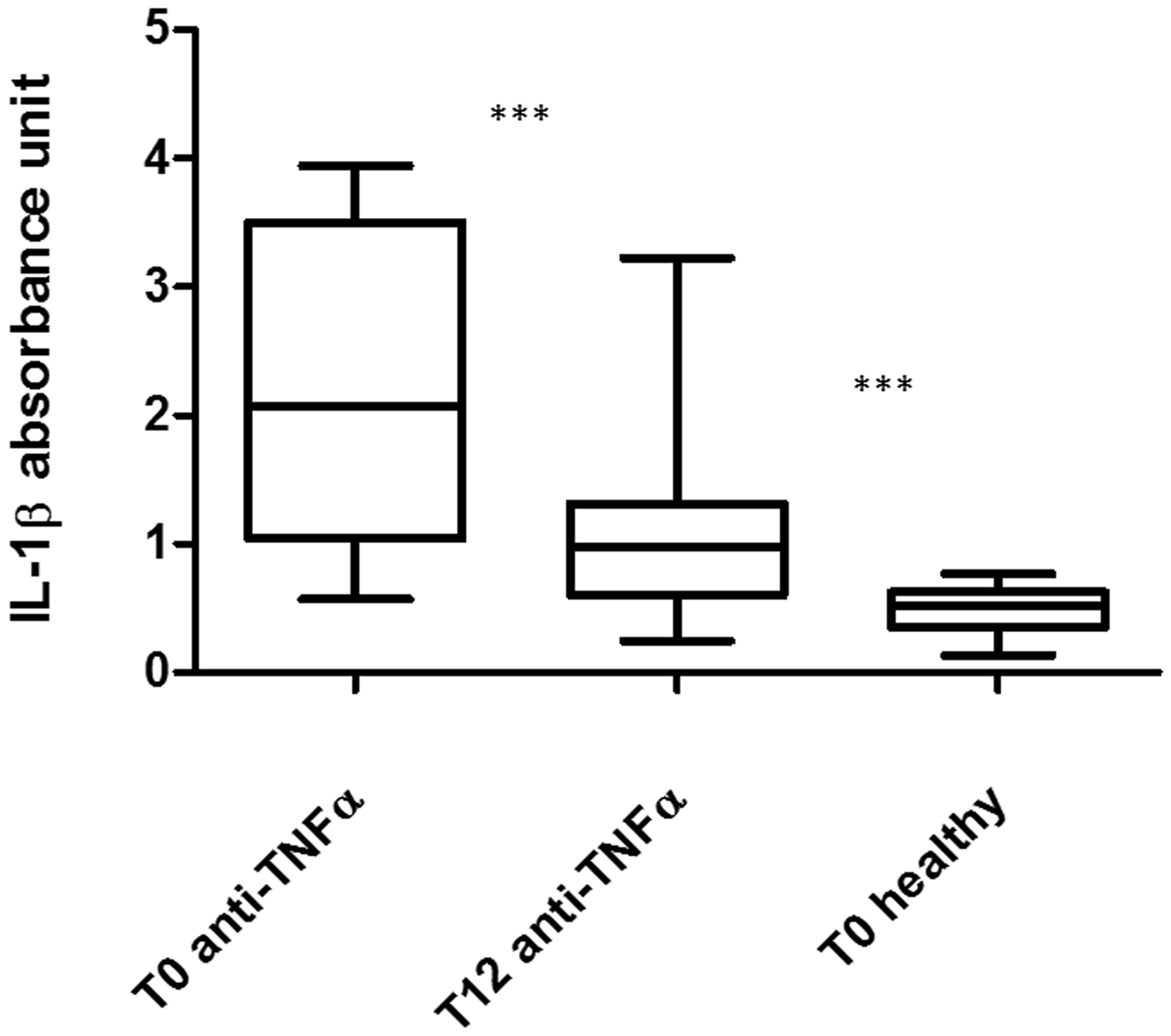
There was no correlation between PASI score and IL-1β level at baseline. There was a significant positive correlation between PASI score and IL-1β level after TNF-α inhibitor treatment. (R2 0.21, P = 0.0028).
Discussion
Psoriasis is a multifactorial disease characterized by a chronic inflammatory process involving innate and adaptive immunity, leading to persistent activation of the immune response in epithelial surfaces and dysregulation of the host inflammatory response.5,11–13
Interleukin-1β is a key inflammatory molecule implicated in the immunopathogenesis of many inflammatory diseases, acting on epithelial cells, macrophages and fibroblasts. In psoriasis, IL-1β regulates chemokine expression and contributes to T-cell extravasation, 14 and has been directly implicated in tissue destruction in other chronic inflammatory diseases such as periodontitis. 15
The efficacy of TNF-α inhibitors in psoriasis is well documented, although evidence of their action on periodontal disease is scarce.16–19 Patients with psoriasis demonstrated higher levels of salivary IL-1β than healthy subjects in the present study, suggesting an increase in IL-1β production in this patient population. Interestingly, TNF-α inhibitor treatment reduced salivary IL-1β levels in our patients. This may attenuate the oral inflammatory process that could be a psoriasis trigger and/or perpetuate the persistent inflammatory process of psoriasis itself. Our study found that, despite being reduced by TNF-α inhibitor treatment, salivary IL-1β levels in patients remained higher than in control subjects. Taken together, these data suggest that salivary IL-1β may serve as a biomarker of disease activity in patients with psoriasis.
The main limitations of this study were the small sample size, and the comparative analysis with nonbiological agents. Although larger cohort studies are needed to validate our results, TNF-α agents appear to interfere with the oral inflammatory process in patients with psoriasis. In conclusion, saliva is a valid noninvasive tool for monitoring inflammation in psoriasis.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
Editorial assistance was provided by Gayle Robins on behalf of HPS–Health Publishing and Services Srl and funded by Pfizer Italia.
