Abstract
Objective
Lymphocyte cytosolic protein 2 (LCP2) is often ectopically expressed in various human tumors. However, the clinical significance and role of LCP2 in lung adenocarcinoma (LUAD) remain unclear. This study explored the prognostic significance of LCP2 in LUAD patients.
Methods
LCP2 expression in LUAD tissues was analyzed using data from The Cancer Genome Atlas and Genotype-Tissue Expression databases. Western blotting was employed to detect LCP2 expression in LUAD. Gene Ontology and Kyoto Encyclopedia of Genes and Genomes analyses were performed to explore signaling pathways mediated by LCP2 co-regulatory genes. Immunohistochemistry was used to examine levels of LCP2 and programmed death ligand 1 (PD-L1) in 68 LUAD patients. Associations between LCP2 expression and clinicopathological features, prognoses, and PD-L1 levels among the LUAD in-patients were analyzed.
Results
Among the 68 LUAD in-patients, LCP2 expression was correlated with clinical stage and lymph node metastasis. LUAD patients with high LCP2 expression were associated with increased overall survival. LCP2 expression may be associated with an enrichment of several immune functions. Moreover, our immunohistochemistry results demonstrated that LCP2 expression was positively correlated with PD-L1 expression in LUAD tissues.
Conclusions
In the study, LCP2 was found to be a favorable prognostic biomarker in LUAD patients.
Keywords
Introduction
Lung cancer is the most common malignant tumor worldwide. According to the latest global cancer statistics in 2021, lung cancer continues to have the highest incidence and mortality rates among all cancer types, accounting for 23.8% of all cancer-related deaths. 1 Among the various lung cancer subtypes, non-small cell lung cancer accounts for approximately 80% of all lung cancers. Compared with squamous cell carcinoma, the incidence of lung adenocarcinoma (LUAD) has constantly increased, becoming a hot topic for clinical and basic and research. 2 Often, systematic chemotherapy combined with radiotherapy is the standard treatment for lung cancer. However, most patients who experience tumor recurrence or progression will have poor prognoses. 3 Meanwhile, immunotherapy has emerged as a promising cancer treatment, 4 although it is still not ideal due to the low response rate of single-agent therapy, long drug half-life, poor controllability, and many other problems. 5 Therefore, it is crucial to identify novel prognostic and immune-related biomarkers to guide clinical treatments for LUAD.
Lymphocyte cytosolic protein 2 (LCP2/Slp-76) is an actin-binding protein that participates in a variety of cell signaling pathways. LCP2 is located on chromosome 5q33 and has a total length of 2032 bp. LCP2 protein can activate T cells and promote the secretion of IL-2 and IFN-γ.6,7 Under normal conditions, the lymphocyte cytoplasmic protein family is primarily expressed by hematopoietic cells. Nevertheless, ectopic LCP2 expression has recently been discovered in a variety of malignant tumors.8,9 It has also been reported that LCP2 is associated with prognosis in gastric cancer and breast cancer patients. 10 However, no relevant studies have been conducted on the role of LCP2 in LUAD progression.
To address this lack of clinical knowledge, we first examined the role of LCP2 in LUAD through bioinformatics followed by validating the relationship between LCP2 and clinical characteristics in LUAD tissues using immunohistochemistry. We also evaluated the potential prognostic value of LCP2 in LUAD patients and confirmed its association with programmed death ligand 1 (PD-L1) expression in LUAD patients. Finally, LCP2 was found to be a potential research target for LUAD.
Materials and Methods
Patients
According to the 2015 World Health Organization classification, we retrospectively reviewed clinical records of LUAD patients who had undergone surgical resection between 1 January 2014 and 31 December 2015 at The Affiliated Tumor Hospital of Xinjiang Medical University, with complete follow-up for two subgroups of patients until December 2020. This study strictly followed the principles and guidelines for reporting preclinical research. All patients provided written informed consent before participating in the study, and the study protocol was approved by the Ethics Committee of the Affiliated Tumor Hospital of Xinjiang Medical University in December 2018 (No. 20180178).
Bioinformatics
The Genotype-Tissue Expression (GTEx) database and The Cancer Genome Atlas (TCGA; https://www.cancer.gov/tcga) were used to analyze the gene expression profiles of LCP2. 11 The mRNA was found to be positively correlated with LCP2 expression based on the cBioPortal database (R > 0.8, P < 0.05). Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genome (KEGG) functional enrichment analyses were performed using cluster profiler. The Linked Omics database (http: //www.linkedomics.org), which contains multi-omics data was used to perform Pearson’s correlation analysis between CD274 (PD-L1) and other genes in the LUAD RNAseq data.
Immunohistochemistry
LCP2 and PD-L1 expression were determined in LUAD patients by immunohistochemistry. Specimens were fixed with 10% formalin solution, embedded in paraffin wax, sliced into continuous 4-μm-thick sections, which were opened at 38°C with a skin bleaching temperature controller. The treated slides were then further sliced and dewaxed. The antigens were repaired using HP/HT, and immunohistochemical staining was performed followed the instructions of the Affinity Biosciences kit (Cincinnati, OH, USA).
Cell culture
The human LUAD cell lines H1299, H1975, and A549 were cultured in RPMI1640 medium containing 10% serum in a 37°C incubator with 5% CO2.
Western blot
Cells were lysed in 1% Triton X-100 lysis buffer. The total protein concentrations in the lysates were determined using a BCA protein assay kit. Proteins from each sample were separated via 10% SDS-PAGE and transferred to polyvinylidene fluoride membranes. Membranes were blocked with 5% non-fat milk powder in Tris-buffered saline containing 0.1% Tween-20 (TBST) at 37°C for 2 hours. Immunoblot analysis was performed with mouse anti-β-actin (#A01010, Abbkine, Wuhan, China) and rabbit anti-LCP2 (#4958, Cell Signaling Technology, Danvers, MA, USA) at 4°C for 12 hours. Membranes were then washed with TBST buffer three times, followed by incubation with HRP-conjugated polyclonal secondary antibodies for 1 hour at 37°C. Immunoreactive bands were developed using the enhanced plus chemiluminescence assay (Pierce Biotechnology, Waltham, MA, USA) following the manufacturer’s instructions. Finally, images were analyzed using a Chemidoc XRS + System (Bio-Rad Laboratories, Hercules, CA, USA). All experiments were repeated twice.
Scoring immunohistochemical staining
We scored LCP2 immunohistochemical staining based on staining intensity and the percentage of positively-stained cells. Staining intensity scores were as follows: 0, colorless; 1, yellow; 2, brown; and 3, dark brown. The percentages of positive cells were scored as follows: 0, 0% to 5%; 1, 5% to 25%; 2, 25% to 50%; 3, 50% to 75%; and 4 >75%. Final scores were the product of the staining intensity and the percentage of positive cells scores. Scores ≥5 were defined as high expression and those <5 were defined as low expression. PD-L1 expression was evaluated using the Tumor Proportion Score (TPS), which was calculated as the percentage of at least 100 viable tumor cells with complete or partial membrane staining. Cytoplasmic staining in tumor cells was not considered. 12 TPS >50% was defined as positive PD-L1 expression; TPS <50% was defined as low PD-L1 expression. Two independent pathologists conducted the assessments.
Statistical analysis
All statistical analyses were performed using SPSS 22.0 software (IBM Corp., Armonk, NY, USA). Differences in the distribution of categorical variables between the LCP2-high and LCP2-low subgroups were compared using the chi-square test. Survival analysis was performed using the log-rank test and Kaplan–Meier method. The association between LCP2 and PD-L1 expression in LUAD was analyzed using the Spearman correlation. In this study, p < 0.05 was considered statistically significant.
Results
LCP2 was downregulated in LUAD
To investigate the potential role of LCP2 in LUAD, we analyzed its expression using the Gene Expression Profiling Interactive Analysis database and found that LCP2 mRNA levels were significantly lower in LUAD (n = 483) compared with in the normal control group (n = 347; p < 0.05) (Figure 1a). LCP2 protein expression in tissues from LUAD patients and the control group were assessed using immunohistochemical staining data from The Human Protein Atlas. 13 These images demonstrated that LCP2 protein expression was downregulated in LUAD tissues compared with in normal lung tissues (Figure 1b). Additionally, LCP2 protein levels were analyzed in the human LUAD cells lines A549, H1299, and H1975 by western blot, which revealed low expression of LCP2 protein in human LUAD cell lines (Figure 1c).
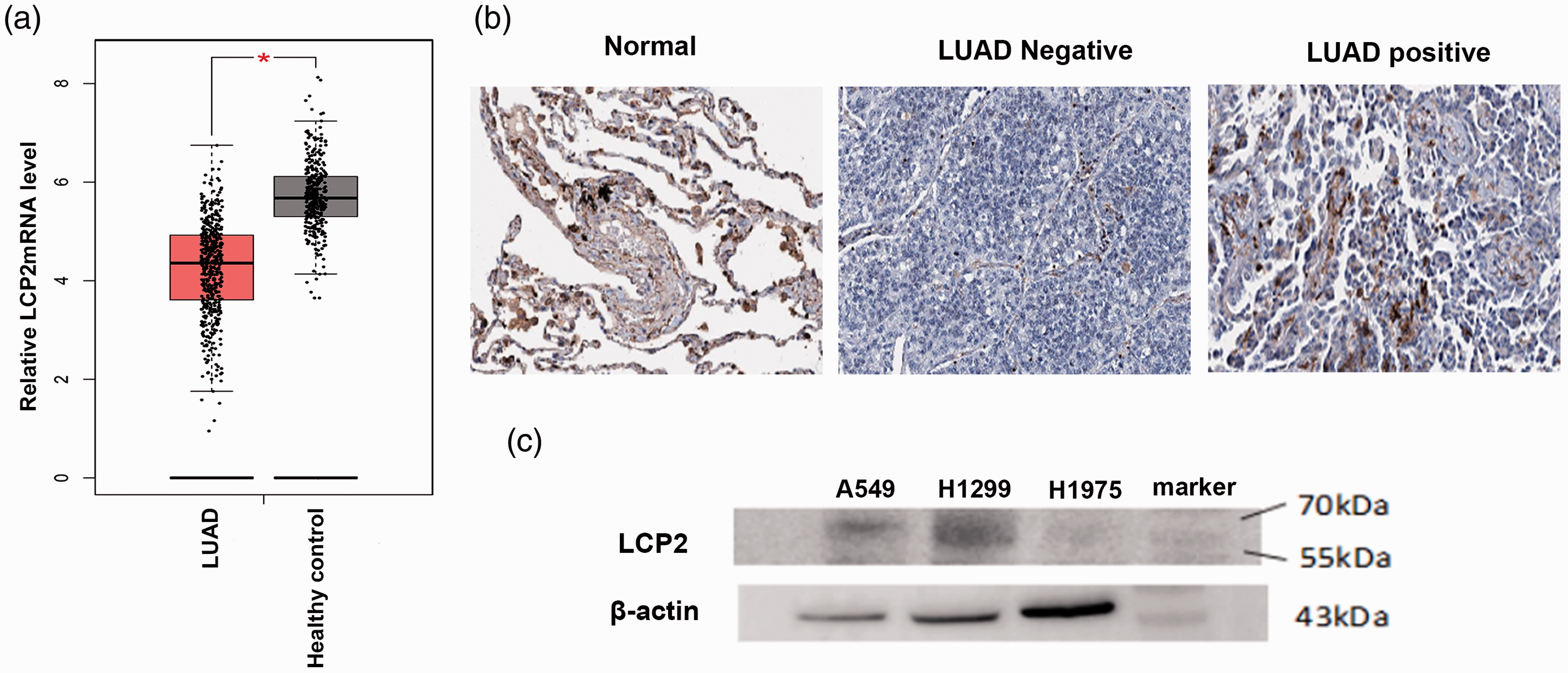
LCP2 was downregulated in lung adenocarcinoma (LUAD) tissues and cell lines. (a) Relative LCP2 mRNA expressions in tissues from LUAD patient versus in normal controls from The Cancer Genome Atlas and Gene-Tissue Expression databases. (b) Immunohistochemical staining of LCP2 in normal lung and LUAD tissues from the Human Protein Atlas database. (c) Western blot analysis of LCP2 expression in LUAD cell lines.
Relationships between LCP2 protein expression and clinical features in LUAD patients
This study included 68 LUAD patients with ages ranging from 32 to 81 years old. None of the included subjects had received prior lung resection or preoperative chemotherapy/radiotherapy. Immunohistochemical staining of LCP2 was performed on pathological paraffin sections from the 68 LUAD patients (Figure 2), among which the rate of positive LCP2 expression was 32%. Clinical characteristics of the 68 LUAD patients are listed in Table 1, and the relationships between LCP2 and clinical features are shown in Table 2. The results showed that LCP2 expression in LUAD was associated with clinical stage and lymph node metastasis (p < 0.05). In contrast, it was not correlated with age, sex, smoking history, tumor location, tumor type, T stage, or M stage.
Representative images of immunohistochemical staining for LCP2 and PD-L1 in tissues from patients with lung adenocarcinoma. (a) High LCP2 expression, (b) low LCP2 expression, (c) LCP2 negative control, (d) high PD-L1 expression, (e) low PD-L1 expression, and (f) PD-L1 negative control. Original magnification, 20×.
Clinical characteristics of lung adenocarcinoma patients.

Clinical characteristics of lung adenocarcinoma patients with high and low LCP2 expression.

Prognostic value of LCP2 in LUAD
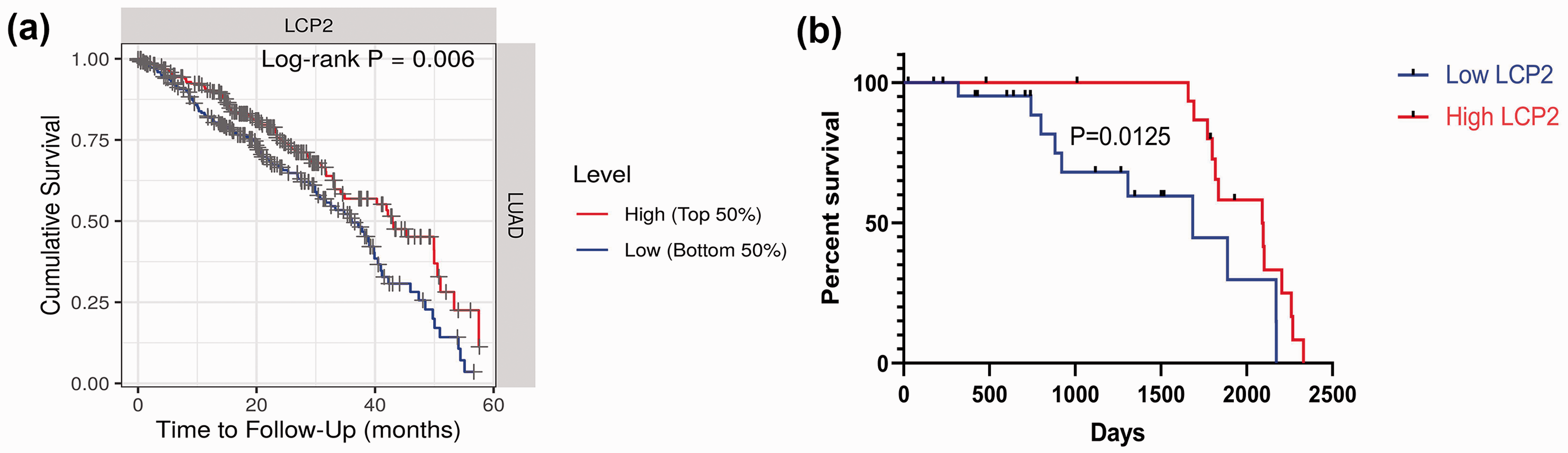
Kaplan–Meier survival analysis and the log-rank test were used to evaluate the potential association of LCP2 with overall survival (OS) in LUAD patients with high and low LCP2 mRNA expression using TCGA data (Figure 3a). Additionally, 41 of the LUAD patients enrolled in this study were divided into two groups: the high and low LCP2 protein expression groups. These patients were followed-up for at least 5 years. The results showed that LUAD patients with high LCP2 expression had longer a OS than those with low LCP2 expression (p < 0.05) (Figure 3b).

Prognostic value of LCP2 in lung adenocarcinoma (LUAD) patients. (a) Kaplan–Meier curves showing the overall survival of LUAD patients with high and low LCP2 mRNA expression. (b) Kaplan–Meier curves showing the overall survival of LUAD patients with high and low LCP2 protein expression. *P < 0.05.
Bioinformatics analysis of LCP2
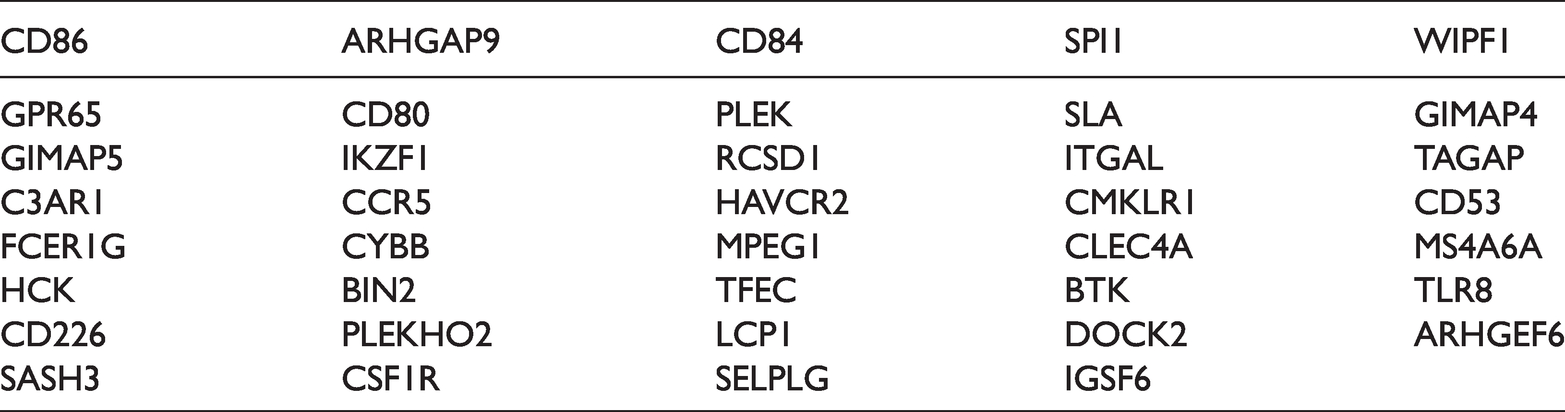
To further investigate the possible mechanism of action of LCP2 in LUAD, 73 mRNAs that were positively correlated with LCP2 expression were screened using the cBioPortal database (r > 0.8, p < 0.05). As seen in Table 3, GO analysis indicated that there was an association between LCP2 and enrichment of immune functions (Figure 4a). KEGG analysis indicated that LCP2 was associated with CAMS, JAK/SATA, and other pathways (Figure 4b).
mRNAs positively correlated with LCP2 expression in lung adenocarcinoma.

Analysis of the biological functions of LCP2. (a) Gene Ontology analysis of LCP2 co-regulated genes. The Y-axis shows Gene Ontology terms associates with biological processes. The length of the bars is proportional to the number of genes. (b) Kyoto Encyclopedia of Genes and Genomes enrichment analysis. The size of the nodes is proportional to the number of genes.
A positive correlation between LCP2 and PD-L1 expression in LUAD
We conducted a heat map analysis of PD-L1 mRNA expression and other mRNAs in 515 LUAD patients from TCGA. The results showed that in LUAD, LCP2 mRNA content was positively correlated with PD-L1 mRNA content. Among all the mRNAs positively correlated with PD-L1, LCP2 ranked fourth (Figure 5a, Figure 5b). The results also indicated that there was a significant positive correlation between LCP2 and PD-L1 mRNA levels (r = 0.66, p < 0.05) (Figure 5c). The immunohistochemistry results for LCP2 and PD-L1 in paraffin sections from the 68 LUAD patients showed that LCP2 and PD-L1 were primarily expressed in the cytoplasm and cell membrane of LUAD cells. Specifically, the rates of positive LCP2 and PD-L1 expression in LUAD tissues were 32.35% and 36.76%, respectively. In the LCP2-high group, there were 17 cases with high PD-L1 expression, and in the LCP2-low group, there were 38 cases with low PD-L1 expression, indicating a positive correlation between levels of the two proteins (r = 0.58, p < 0.05) (Figure 2).

(a) Heat map showing genes positively correlated with CD274 (PD-L1) in lung adenocarcinoma (LUAD). (b) Volcano plot showing the correlation between CD274 (PD-L1) with other genes in LUAD. (c) Scatter plot for correlation analysis of LCP2 and PD-L1 expression in LUAD.
Discussion
LCP2 is an important molecule in T cell signal transduction. 14 Previous studies have shown that LCP2 is expressed by a variety of hematopoietic cells.15–17 Additionally, current data have revealed that LCP2 is differentially expressed in various cancers. In our study, LCP2 expression in LUAD was first evaluated in TCGA and GTEx databases, and the results showed that LCP2 mRNA and protein levels were significantly lower in LUAD than in the control group. Furthermore, low LCP2 expression was found in three human LUAD cell lines, which was consistent with the TCGA and GTEx results. Previous studies have reported that LCP2 is highly expressed in leukemia and colon cancer and that high LCP2 protein expression is correlated with aggressive behaviors in chronic lymphocytic leukemia cells 9 and is involved in colon cancer metastasis. 18 Our results suggest that LCP2 expression in LUAD is different from that in other tumors.
In this study, LCP2 expression in tissue samples from 68 LUAD patients was determined by immunohistochemistry, and then relationships between LCP2 expression and clinical characteristics were analyzed. We discovered that LCP2 expression was correlated with clinical stage and lymph node metastasis (p < 0.05). The results further suggested that LCP2 expression was closely associated with early clinical stage. In the analysis of patient prognosis, we first used LCP2 mRNA levels in LUAD patients from TCGA to generate a using Kaplan–Meier survival curve. Subsequently, we retrospectively collected clinical samples and followed-up the enrolled patients for at least 5 years. These analyses found that LUAD patients with high LCP2 expression had an increased OS during the 5-year follow-up period. The Kaplan–Meier survival curve of TCGA cases also showed that LUAD patients with high LCP2 expression were associated with increased OS. Wang et al. showed that LCP2 overexpression also occurs in metastatic cutaneous melanoma and is associated with better OS. 19 Our study confirmed the assumption that the prognostic effect of LCP2 in LUAD was different from that in other tumors, which may be due to the different effects of LCP2 expression in different tumors. Previous studies have shown that LCP2 activates the IL-2 promoter by promoting T cell activation; IL-2 secretion causes the release of cytokines that lead to target cell apoptosis. 20 Quantitative reductions of LCP2 trigger immune dysregulation. 21 We found that LCP2 was associated with patient prognosis, acting as a protective protein. The results of GO and KEGG analyses showed that LCP2 was associated with enrichment of immune functions and was related to CAMS, JAK/SATA, and other pathways. The study by Yan et al. shed light on the development of LCP2-targeted therapeutics for sepsis and revealed the important function of LCP2 in RAGE-mediated proinflammatory signaling. 22 The immune system plays a critical role after tumor occurrence, and tumor development is a multi-gene and multi-stage process that includes important contributions of oncogenes and tumor suppressor genes. Thus, the role of LCP2 in LUAD cells and the tumor microenvironment still needs to be further explored in vitro and in vivo.
PD-L1 is an important member of the coordinated stimulus molecule B7 family that negatively regulates T cell activity.
23
PD-L1 expression leads to a failure of the body's anti-tumor response and is a mechanism of tumor immune escape; thus, PD-L1 promotes tumor occurrence and development.
24
Despite the existing data suggesting that high PD-L1 expression is associated with poor prognosis,
26
in this study, we discover that LCP2 was a protective factor in LUAD. The correlation between the two is worth considering for the following reasons. First, in tumor cells, PD-L1 expression is regulated by a variety of endogenous and exogenous signals, including chromosomal variation, epigenetic modification, abnormal pro- or anti-cancer signals, inflammatory factors, and other factors. These signals can regulate both the expression and function of PD-L1 through multiple pathways, including genomic, transcriptional, post-transcriptional modifications, translation, and post-translational modifications.
27
Recent studies have suggested that PD-L1 expression levels on tumor cell and some associated immune cell membranes may influence the clinical efficacy of anti-PD-1/PD-L1 therapy.
28
Second, the tumor microenvironment includes inflammatory cytokines secreted by tumor cells, which alter PD-L1 expression in tumor cells.
29
There were some limitations to this study. First, although TCGA database was used in our study, the number of tissue samples obtained was insufficient. This may cause some biases. Second, the biological role of LCP2 in LUAD and the potential association between LCP2 and PD-L1 needs to be further studied in vitro and in vivo.
Collectively, our study demonstrated that LCP2 can be a prognostic marker in LUAD patients, and the correlation between LCP2 and PD-L1 provides a new direction for future research.
Footnotes
Acknowledgements
We thank all the participants who took part in this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Data Availability
Data are available on request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This work is sponsored by the National Key R&D Program of China (2017YFC0909903).
