Abstract
Objective
We aimed to assess prescribing practices, compliance with guidelines, and outcomes for patients who were admitted to the authors’ institution with community-acquired pneumonia (CAP).
Methods
We performed a single-center retrospective cross-sectional study of adults with CAP presenting during the 2019 influenza season. CAP severity was assessed using the CURB-65 risk score. The effect of CURB-65 risk score use on the rate of appropriate antimicrobial prescribing was assessed using the chi-square test and reported as odds ratio (OR). Fisher’s exact test was used to assess the relationship between prescribing appropriateness and patient outcomes.
Results
Patients with low-risk CAP were most likely to be inappropriately prescribed antimicrobials (OR: 4.77; 95% confidence interval: 2.44–10.47). In low-risk CAP, the most common prescribing error was overuse of ceftriaxone. In high-risk CAP, the most common errors were ceftriaxone underdosing and missed atypical coverage with azithromycin. Overall, 80% of patients were considered to have been inappropriately prescribed antimicrobials. No effect on mortality was observed.
Conclusions
In this study, we found low use of CAP risk scores and low adherence to antimicrobial prescribing guidelines for CAP at the authors’ institution.
Keywords
Introduction
Lower respiratory tract infections are the fourth leading cause of healthy life lost worldwide. 1 If acquired outside the hospital, such infections of the lung parenchyma are known as community-acquired pneumonia (CAP) and commonly have a bacterial or viral etiology. In Australia, the cost of treating patients hospitalized with CAP is estimated to be AUD 350 million annually. 2
Initial antimicrobial prescribing for bacterial CAP is typically empiric, intending to cover a range of common pathogens including Streptococcus pneumoniae, Mycoplasma pneumoniae, Legionella species, Staphylococcus aureus, and enteric gram-negative bacilli. 3 Australian clinicians are guided in their treatment of CAP by readily accessible electronic Therapeutic Guidelines (eTG), which detail the current standard of care. 4 The eTG provide guidance on antimicrobial selection, dosing, and duration of therapy, and recommendations are provided for several CAP severity assessment tools to assist in decision making. One such well-validated and easily applied tool is the CURB-65 risk score, which can be used in the emergency department (ED) to aid decision making regarding inpatient versus outpatient management. 5 The CURB-65 risk score combines patient data on confusion, serum urea nitrogen, respiratory rate, blood pressure, and age to determine whether patients with CAP have a low, intermediate, or high risk of 30-day mortality. The calculated severity score is also used to recommend the appropriate empirical antimicrobial therapy.
For both erroneous and intentional reasons, deviation from clinical guidelines is relatively common. 6 When treating pneumonia, however, frequent departure from the prescribing guidelines increases patient morbidity and mortality and contributes to rates of antimicrobial resistance. 7 To improve the understanding of adherence to eTG practice recommendations, we aimed to assess the rates of use of pneumonia severity scores, CAP investigations, antimicrobial prescribing, and outcomes in patients who presented with CAP at a tertiary Australian hospital during the influenza season of 2019.
Methods
Study area and period
We conducted a single-center retrospective review of all adult patients age ≥18 years with a primary discharge diagnosis of community-acquired pneumonia, who presented to our institution between 1 June and 31 August 2019. Patient data were deidentified at the time of collection. Our institution is a 250-bed outer-metropolitan hospital in Australia, with approximately 63,000 ED presentations in 2019. 8 The findings of the present study are reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement for reporting cross-sectional studies. 9
Inclusion and exclusion criteria
We excluded all patients with a background of chronic obstructive pulmonary disease, bronchiectasis, cystic fibrosis, severe asthma requiring daily corticosteroids, or other chronic suppurative lung diseases. We excluded patients diagnosed with hospital-acquired pneumonia and aspiration pneumonia because treatment of these conditions was outside the scope of our study. All patients transferred to or from our institution during their CAP treatment course were excluded because we could not assess the total duration of prescribing, appropriate de-escalation, whether re-escalation was required, or clinical outcomes.
Data points and collection methods
We collected data on patient demographics, vital signs at admission, radiology, laboratory investigations, antimicrobial prescribing, and patient outcomes. Patient demographic information included age, sex, respiratory comorbidities, and residence at an aged care facility. Radiological data included the presence of pneumonia on any of the following during the first 48 hours: chest x-ray, chest computed tomography (CT), and CT pulmonary angiogram. Laboratory investigations included: blood urea nitrogen levels, organisms identified on sputum microscopy, culture, and sensitivity, conventional and rapid (GeneXpert® Xpress Flu/RSV PCR; Cepheid, Sunnyvale, CA, USA) polymerase chain reaction (PCR) for respiratory viruses, urinary antigen testing for L. pneumophila and S. pneumoniae, and serology for M. pneumoniae, Legionella spp., Chlamydia spp., and Coxiella burnetii. The conventional respiratory viral PCR panel used at our institution detects influenza A and B viruses, parainfluenza 1 to 3 viruses, respiratory syncytial virus (RSV), human metapneumovirus, and adenovirus, within 24 to 48 hours. The GeneXpert® Xpress Flu/RSV PCR detects influenza A and B viruses and RSV within 2 hours. Laboratory investigation results were identified using the pathology clinical information system (AUSLAB; Citadel Health Pty Ltd, Belmont, Australia). Patient imaging was accessed using an online clinical imaging system (Synapse PACS; Fujifilm Medical Systems, Lexington, MA, USA). Antimicrobial data included agent, dose, route, duration, and patient antimicrobial allergy status. Patient outcomes included mortality within 3 months and re-presentation to the hospital within 1 week for respiratory illness. Additional data points were collected from the electronic medical records system (TheViewer; Queensland Health, Brisbane, Australia) and hard-copy patient medical records.
Definitions
We defined appropriate antimicrobial prescribing as any antimicrobial prescribed in accordance with eTG recommendations. CAP severity was determined using the CURB-65 score, in which one point is assigned for each of the following: presence of confusion, uremia >7 mmol/L, respiratory rate ≥30 breaths per minute, ≤90 mmHg systolic or ≤60 mmHg diastolic blood pressure, and age ≥65 years. A score of ≤1, 2, or ≥3 equates to a low (0.6%), moderate (6.8%), or high (>14%) risk of 30-day mortality, respectively. We considered initial antimicrobial prescribing that followed local sepsis guidelines as appropriate prescribing. We considered all antimicrobial coverage for suspected atypical pathogens as appropriate if prescribed in accordance with eTG recommendations (i.e., correct route, correct dose). Only antimicrobials prescribed at hospital presentation or later were considered, including discharge prescriptions.
Antimicrobial prescribing was considered inappropriate if the following conditions were met: additional non-indicated antimicrobials were prescribed, non-eTG-recommended antimicrobials were prescribed, an antimicrobial was incorrectly prescribed, or recommended antimicrobials were not prescribed. Additional non-indicated antimicrobials were defined as antimicrobials recommended for higher-risk CAP or for people with hypersensitivity reactions without documented allergy. Non-eTG-recommended antimicrobials were defined as any antimicrobial not listed under eTG CAP treatment, except where prescribing was for a concurrent non-CAP indication. Antimicrobials were considered incorrectly prescribed if the dosage was incorrect, duration of therapy was longer or shorter than recommended, antimicrobials were administered via the incorrect route, or an antimicrobial was prescribed to someone with a pre-existing documented allergy to that antimicrobial. The eTG recommends 5 to 7 days of total antimicrobial treatment. A missing recommended antimicrobial was defined as any antimicrobial not prescribed but recommended in the eTG, according to the assessed CAP risk. For confirmed viral CAP, if antimicrobials were stopped within 48 hours of microbiological diagnosis, this was considered appropriate treatment. Patients continued on antimicrobials for superimposed bacterial infection following microbiological diagnosis of viral CAP were evaluated according to the criteria above.
Statistical analysis
Continuous variables are reported as median and interquartile range. Categorical variables are reported as number and percentage. The effect of the use of CURB-65 risk score on the rate of appropriate antimicrobial prescribing and mortality was assessed using the chi-square test and reported as odds ratio (OR) with 95% confidence interval (CI). For the distribution of pathogens, Fisher’s exact test was used to test the hypothesis that viral or bacterial infection was independent of the CURB-65 score. The threshold for statistical significance was p < 0.05. Statistical analysis was performed using R version 4.0.2 (The R Foundation for Statistical Computing, Vienna, Austria).
Ethics
Ethical approval was granted by the Royal Brisbane Human Research Ethics Committee (no. LNR/2020/QRBW/64250). Participant consent not required as this was a retrospective study using existing de-identified patient data. Thus, individual informed consent was impractical to obtain and this study involved no more than minimal risk to the individual.
Results
Characteristics, investigations, and outcomes
We identified 381 patients with a presentation of pneumonia, among which 126 met the inclusion criteria (Figure 1). The median patient age at presentation was 69 (range, 53–83) years and 23% (n = 29) of patients were nursing home residents. CURB-65 scores indicated low-, moderate-, and high-risk CAP in 41% (n = 52), 28% (n = 35), and 31% (n = 39) of patients, respectively. No causative organism was identified in 74% (n = 93) of patients whereas bacterial and viral pathogens were detected in 5.6% (n = 7) and 21% (n = 26) of patients, respectively (Table 1). ED documentation of pneumonia severity scores occurred in 4% (n = 5) of patients.

Flow chart of patients diagnosed with community-acquired pneumonia during a single influenza season at a tertiary Australian Hospital in 2019, with reasons for exclusion from the study.
Patient characteristics, relevant CURB-65 score details, investigation types and frequency of use, detected causative organisms, and outcomes of included patients treated for community-acquired pneumonia during a single influenza season at an Australian hospital in 2019.

aMedian (interquartile range); n (%).
bOne patient had infection with two bacterial pathogens.
cMicroscopy, sensitivity, and culture.
dPolymerase chain reaction.
BUN, blood urea nitrogen; BP, blood pressure; bpm, breaths per minute; CT, computed tomography; CTPA, CT pulmonary angiogram; CXR, chest radiography; MCS, microscopy, culture, and sensitivity; PCR, polymerase chain reaction.
Antimicrobial prescribing
During the analyzed period, 20% (n = 25) of patients presenting with CAP received appropriately prescribed antimicrobials (Figures 2 and 3). Compared with receiving appropriate antimicrobials, the OR of receiving inappropriate antimicrobials in low-risk CAP was 4.77 (95% CI: 2.44–10.47, p < 0.001); this OR was taken as the reference when assessing other CAP risk categories. Patients with high-risk CAP had the lowest proportion of appropriate prescribing (Figure 2); however, the OR was not statistically significant (OR: 2.49; 95% CI: 0.56–15.35) compared with low-risk CAP. Patients with moderate-risk CAP were most likely to be appropriately prescribed antimicrobials (OR: 0.36; 95% CI: 0.12–1.07; p = 0.046) in comparison with low-risk CAP. There was no significant difference in 30-day mortality between appropriate and inappropriate antimicrobial prescribing (Table 2 and Figure 3).
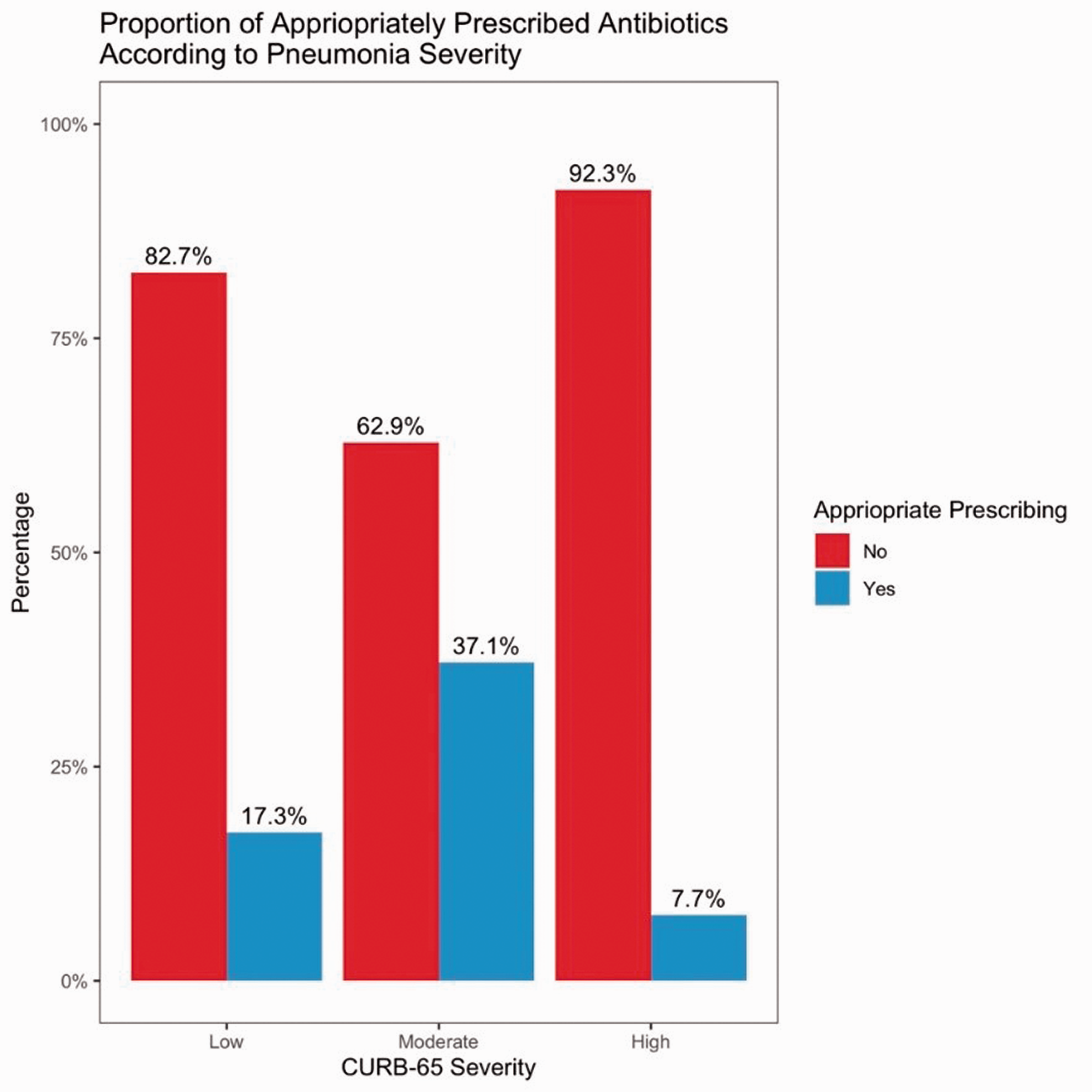
Proportion of included patients considered to have received appropriately versus inappropriately prescribed antibiotic therapy for community-acquired pneumonia during a single influenza season at a tertiary Australian hospital in 2019, according to CURB-65 risk scores.

Distribution of reasons for classification as inappropriately prescribed antibiotic therapy for community-acquired pneumonia during a single influenza season at a tertiary Australian hospital in 2019, according to CURB-65 risk scores.
In low-risk CAP, use of additional non-indicated antimicrobials was common in 60% (n = 41) of patients, followed by incorrectly prescribed antimicrobials in 30% (n = 21) (i.e., incorrect dose, route, duration, or given to a patient with an allergy). All patients with low-risk CAP who received additional non-indicated antimicrobials received benzylpenicillin or ceftriaxone. Ceftriaxone was prescribed in 37% (n = 19) of patients. In moderate-risk CAP, additional non-indicated antimicrobials were common in 35% (n = 11) of patients, with use of ceftriaxone without non-severe penicillin hypersensitivity most frequent (n = 7, 23%), followed by incorrectly prescribed antimicrobials (n = 9, 30%) (i.e., incorrect dose, route, duration, or given to patient with an allergy). Despite azithromycin only being indicated for high-risk CAP, 10% (n = 5) of patients with low-risk CAP and 11% (n = 4) of those with moderate-risk CAP received azithromycin. In high-risk CAP, incorrectly prescribed antimicrobials (i.e., incorrect dose, route, duration, or given to patient with an allergy) were common in 69% (n = 27) of patients, with ceftriaxone underdosing occurring most frequently (n = 17, 44%). Appropriate prescribing of azithromycin was missed in 67% (26) of patients with high-risk CAP.
Non-eTG-recommended antimicrobials were used in 13% (n = 7), 14% (n = 5), and 20% (n = 8) of patients with low-, moderate-, and high-risk CAP, respectively. Augmentin Duo Forte (amoxicillin 875 mg with clavulanate 125 mg) was the most prescribed non-eTG-indicated antimicrobial (n-10, 8%). Overall, 30% (n = 38) of patients exceeded the recommended total duration of antimicrobial treatment, with no significant difference in duration between risk scores (Table 2).
Duration of antimicrobial use and mortality according to CURB-65 risk score for included patients treated for community-acquired pneumonia during a single influenza season at a tertiary Australian hospital in 2019.

aMedian (interquartile range); n (%).
bKruskal–Wallis test, chi-square test of independence, Fisher's exact test.
Discussion
In this study, we reviewed antimicrobial prescribing and outcomes for CAP in adult patients during an influenza season at a tertiary Australian hospital. Prescribing was considered inappropriate in 80% of instances, with errors that varied according to CURB-65 score. We found that patients with low-risk CAP were most likely to be inappropriately prescribed antimicrobials (OR: 4.77; 95% CI: 2.44–10.47; p < 0.001). Patients with high-risk CAP were most likely to receive incorrectly prescribed antimicrobials (i.e., incorrect dose, route, duration, or given to patient with an allergy) and have missed coverage for atypical pathogens. Documented use of pneumonia severity scores was low at 4%. Overall, the appropriateness of antimicrobial prescribing did not have an impact on mortality.
Our finding that antimicrobial prescribing was aligned with the nationally recognized standard in only 20% of instances is similar to previous reports. Two multi-site studies of ED-prescribing practices for CAP in Australian hospitals by Maxwell et al. and McIntosh et al. demonstrated concordance with the eTG in 18% and 20% of cases, respectively, and an Irish study by O’Kelly et al. found that 22% of prescribing was aligned with national standards.10–12 Although the pneumonia severity index (PSI) was used, Maxwell et al. and McIntosh et al. similarly found that the most common deviation from guidelines was the prescribing of antibiotics not indicated according to severity class.11,12 We observed this most notably in low-risk CAP with 60% of patients receiving intravenous (IV) antibiotic therapy indicated for higher-risk presentations, as well as in moderate-risk CAP with 35% of patients receiving additional non-indicated antibiotics according to their risk score. Maxwell et al. and McIntosh et al. also raised concerns regarding the frequent use of third-generation cephalosporins as empirical treatment for CAP.11,12 The eTG only recommends third-generation cephalosporins as an option for high-risk diseases or for moderate-risk CAP with documented non-severe penicillin hypersensitivity. 4 We observed that 37% of patients with low-risk CAP and 20% with moderate-risk CAP received ceftriaxone without documented penicillin hypersensitivity. Whereas this is lower than the 44% described by Maxwell et al., the ongoing overuse of third-generation cephalosporins raises concerns regarding increased rates of emerging resistance owing to inappropriately broad coverage.4,11,12 The most common reason for inappropriate prescribing identified by O’Kelly et al. was the overuse of amoxicillin with clavulanate. 10 We similarly found that amoxicillin with clavulanate was the most common non-eTG-recommended antibiotic, despite the eTG advising directly against its use owing to the lower daily dose of amoxicillin, the active component against S. pneumoniae, as compared with amoxicillin 1 g three times daily. 4 In addition to its broader spectrum, this contributes to the development of antibiotic resistance. 4 Similar to Maxwell et al., we found that in high-risk CAP, missed doses of azithromycin contributed to inadequate coverage and the most common errors in dosing related to ceftriaxone was routine underdosing. This was similarly demonstrated by Buising et al., who found that 35% of patients with CAP requiring management in the intensive care unit did not receive adequate atypical coverage, in an Australian audit of empiric CAP prescribing. 13 The 2018 eTG updates saw an increase in high-risk CAP ceftriaxone dosing from 1 g daily to 2 g daily; our findings may reflect either a delay in this change in clinical practice or inadequate CAP risk assessment, which seems to be apparent in other data.4,5,10–12,14,15
In our cohort, the documented use of any pneumonia severity score was low at 4%. In the Australian studies by Maxwell et al. and McIntosh et al. low documented use of the PSI was also identified, at 5% and 6%, respectively; the authors argued that this reflected a lack of awareness about this tool.11,12 Several other studies have demonstrated similarly low documented use of pneumonia severity scores between 0.4% and 12%, despite these tools being clinically validated and embedded in the guidelines.5,10,12,14,15 The poor compliance with guidelines in the context of a lack of severity risk assessment potentially indicates that the low use of standardized tools leads to misclassification of pneumonia severity and consequently contributes to inappropriate prescribing. This is likely reflected in the high prevalence of additional non-indicated antimicrobial prescribing in patients with low- and moderate-risk CAP and missed antimicrobial prescribing in high-risk CAP.
Several recommendations can be made based on the findings of this study. We suggest that an increased use of pneumonia risk scores at initial presentation would likely increase concordance with antibiotic prescribing guidelines. 13 This is important because improved compliance with the guidelines in CAP has been shown to reduce 30-day mortality, sepsis, renal failure, duration of hospitalization, and development of antibiotic resistence.3,16,17 For low- and moderate-risk CAP, avoiding the use of non-indicated antibiotics is advised, particularly third-generation cephalosporins in patients without non-severe penicillin hypersensitivity. In cases where high-risk CAP has been appropriately identified, we recommend addition to the empirical regimen of active agents against atypical causes of CAP. If ceftriaxone is part of this regimen, then it should be dosed correctly. Total duration of antibiotic prescribing, including both IV and oral doses, must be calculated and monitored to prevent unnecessarily prolonged treatment courses. Finally, we suggest that amoxicillin with clavulanate not be used in the treatment of CAP.
This study has several limitations. Primarily, use of CAP risk scores is not a substitute for clinical judgment, and we cannot account for additional factors noted during patient assessment. We also considered the number of days of therapy rather than individual doses of antimicrobials; thus, in some instances where we considered the duration to be too long, patients may have received the correct number of doses. Additionally, our case numbers were small, reflecting local prescribing practices; therefore, the findings may not be generalizable to other settings. Finally, we could not verify antimicrobial adherence post-discharge despite including discharge antimicrobials in our analysis. This may be a confounder in terms of patient outcomes; however, we included these data because they reflected the prescribing intentions of the treating team.
Conclusion
In this study, we found low compliance in clinical practice with the accepted standard of care in antimicrobial prescribing for CAP at the authors’ institution. In particular, we noted a high rate of misuse of ceftriaxone and missed coverage for atypical pathogens.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211058366 - Supplemental material for Antimicrobial prescribing and outcomes of community-acquired pneumonia in Australian hospitalized patients: a cross-sectional study
Supplemental material, sj-pdf-1-imr-10.1177_03000605211058366 for Antimicrobial prescribing and outcomes of community-acquired pneumonia in Australian hospitalized patients: a cross-sectional study by Lawless Robert, Vickers Mark, Alawami Moayed, Appasamy Nivashen, Rajasingam Vinod, Paviour Sophie, El Washahy Mohamed and Chew Rusheng in Journal of International Medical Research
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: Funding of open access publication costs was provided by the Redcliffe Hospital PPTF Advisory Committee.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
