Abstract
Objective
This meta-analysis was conducted to investigate the relationship between the interleukin (IL)-17A rs2275913 polymorphism and rheumatoid arthritis (RA) susceptibility.
Methods
Eligible studies were retrieved from PubMed, Embase, and Web of Science. The fixed- or random-effects model was used to calculate the pooled odds ratios (ORs) and 95% confidence intervals (95%CIs) on the basis of heterogeneity.
Results
Overall, 11 studies containing 4019 RA patients and 4137 controls were included in this meta-analysis. The results suggested a significant association between the IL-17A rs2275913 polymorphism and RA susceptibility in the overall population (allelic model A vs. G: OR = 0.89, 95%CI: 0.83–0.95; heterozygote model GA vs. GG: OR = 0.87, 95%CI: 0.78–0.96; homozygote model AA vs. GG: OR = 0.82, 95%CI: 0.71–0.96; dominant model GA + AA vs. GG: OR = 0.86, 95%CI: 0.78–0.94). In the subgroup analyses, the IL-17A rs2275913 polymorphism was significantly associated with RA risk in Europeans (allelic model A vs. G: OR = 0.87, 95%CI: 0.78–0.97; heterozygote model GA vs. GG: OR = 0.79, 95%CI: 0.68–0.93; dominant model GA + AA vs. GG: OR = 0.79, 95%CI: 0.68–0.92), but not in Africans or Americans.
Conclusion
This study suggests that the IL-17A rs2275913 polymorphism is significantly associated with RA susceptibility in Europeans.
INPLASY registration number: INPLASY202170056.
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease that affects 1% of the population worldwide and is characterized by damage to articular cartilage, progressively destructive joint inflammation, and synovial hyperplasia. 1 The incidence of RA increases with age, affecting about 6% of the population over 65 years old. 2 Although the cause of the disease is not completely understood, genetic and environmental factors are regarded as two main reasons for developing RA. 3
T helper 17 (Th17) cells reportedly play a crucial role in autoimmunity, joint destruction, and angiogenesis. 4 Interleukin-17A (IL-17A) is one of the six members of IL-17 family that includes IL-17A–F, which are cytokines secreted by Th17 cells and other related immune cells. 5 IL-17A stimulates the production of multiple factors, such as tumor necrosis factor-alpha (TNF-α), IL-6, and IL-1β, which are important in the inflammatory response. 6 RA synovial fluid and tissue explants contain high levels of IL-17A and its receptor.7–9 Hence, IL-17A is a promising target for anti-cytokine therapy in autoimmune arthritis. 10
In recent years, the associations between six IL-17A polymorphisms and RA risk have been studied in various populations by many researchers,11–16 with the most frequent variant being IL-17A rs2275913, G-197A. Although some studies have shown that the IL-17A rs2275913 variant allele reduced the RA risk, there are also reports stating that this variant allele was not an important factor for RA susceptibility. 17 Overall, the results remain inconclusive among these studies. Therefore, we conducted the present meta-analysis to evaluate the association between the IL-17A rs2275913 polymorphism and RA risk.
Methods
This meta-analysis was performed in accordance with the guideline of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 18 In error, we did not prospectively register this trial, but we have now registered it retrospectively at International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY) (registration number: INPLASY202170056).
Identification of eligible studies
Systematic searches in the PubMed, Embase, and Web of Science online databases were performed by two independent investigators (PC and LLL) up to 1 June 2020. The following search terms were used: ‘‘Interleukin-17A or IL-17A,’’ ‘‘genetic polymorphisms or polymorphism or variant,’’ and ‘‘rheumatoid arthritis or RA.’’ There were no limitations for origin, languages, or items. References included in relevant studies, previous meta-analyses, and reviews were also manually searched.
Inclusion and exclusion criteria
The title and abstract of each study were carefully reviewed. Reference lists of relevant studies and reviews were also manually searched. Eligible studies were included by the following criteria: (1) case–control studies; (2) evaluated the association between IL-17A polymorphisms and RA susceptibility; (3) focused on humans; (4) included detailed genotype distribution data that could be acquired to calculate odds ratios (ORs) and 95% confidence intervals (95%CIs). Exclusion criteria included: (1) meeting articles, reviews, and duplications of previous publications; (2) studies with no detailed genotype data; (3) did not focus on the IL-17A rs2275913 polymorphism; (4) family-based studies. When several studies or articles from the same population appeared in the search, only the most recent or complete study was included.
Quality assessment
Quality assessment of the included studies was performed independently by two investigators (PC and ZL) according to Newcastle–Ottawa Quality Assessment Scale (NOS) 19 for cohort studies with moderate modification. Each included study was judged on three main perspectives: (a) the selection of the study groups: 0–4 points, (b) the comparability of the groups: 0–2 points, and (c) the outcome of the groups: 0–3 points. A study with a score of 6 or higher was considered as high quality.
Data extraction
The name of the first author, year of publication, country of origin, genotyping method, criteria of RA, number of cases and controls, and genotype frequencies for cases and controls were extracted from each eligible study. Two independent authors (PC and GZ) checked all the collected data and reached a consensus on each item. Any encountered discrepancies were resolved by consensus.
Trial sequential analysis (TSA)
TSA was performed to minimize the type I error and random error, as the present study had a smaller sample size. This was done using TSA software version 0.9.5.10 to calculate the required information size (RIS) by setting the relative risk reduction (RRR) to 10%, power to 95%, and type I error (α) to 1%. The monitoring boundaries were constructed to determine whether the present meta-analysis was sufficiently powered and conclusive to be able to reject false-positive reports from the meta-analysis. If the Z-curve crossed the TSA boundaries or futility area, there was sufficient information to support the conclusions and further trials are unlikely to change the findings. If the Z-curve did not cross the any of the boundaries or reach the RIS, the evidence was insufficient to reach a firm conclusion.
Statistical analysis
The chi-squared test was conducted on the control groups to evaluate the Hardy–Weinberg equilibrium (HWE) of each study, and the HWE was considered to be a significant departure when P < 0.05. ORs and 95%CIs were calculated to assess the strength of the association between the IL-17A rs2275913 polymorphism and RA susceptibility. The pooled ORs of the present meta-analysis were performed for the IL-17A rs2275913 allelic model (A vs. G), heterozygote model (AG vs. GG), homozygote model (AA vs. GG), dominant model (AA + AG vs. GG), and recessive model (AA vs. AG + GG). Cochran’s Q statistic and I2 statistic were used to evaluate the heterogeneity. The heterogeneity was considered to be significant when I2 > 50% and P < 0.1. A random-effects model or fixed-effect model was used to pool the effect sizes depending on the presence or absence of statistical heterogeneity, respectively. Potential publication bias was assessed by Egger’s regression test. All statistical tests were performed with Review Manager software version 5.2 (Nordic Cochrane Centre, Copenhagen, Denmark) and Stata 12.0 software (StataCorp, College Station, TX, USA).
Results
Characteristics of studies
According to the search strategies, 107 articles were retrieved from the PubMed, Embase, and Web of Science databases. The selection process is shown in Figure 1. Overall, 47 reports were discarded, with 14 being duplicate records and 33 having improper titles or abstracts. Among the remaining 18 studies, 1 was a scientific abstract, 20 1 was a meeting report, 21 2 were not case–control studies,22,23 and 3 did not include key information on the IL-17A rs2275913 polymorphism.14,24 From the remaining 11 studies, 4019 RA patients and 4137 controls were included in this meta-analysis.11–13,16,17,24–29 Only allele information for the rs2275913 polymorphism was available in the Louahchi et al. study. 27 In addition, two cohorts including populations from Norway and New Zealand were contained in Nordang’s study. 16 The characteristics of the 11 eligible studies are summarized in Table 1. The allele and genotype distributions of the rs2275913 polymorphism are listed in Table 2.
The flow chart of the literature search and study selection process.
Characteristics of the studies included in this meta-analysis.

RA, rheumatoid arthritis; HWE, Hardy–Weinberg equilibrium; PCR-RFLP, polymerase chain reaction restriction fragment length polymorphism assay; NA, not available; Y, yes; N, no.
Allele and genotype information of the interleukin (IL)-17A rs2275913 polymorphism among rheumatoid arthritis (RA) patients and controls.

RA, rheumatoid arthritis; NA, not available.
Association between the IL-17A rs2275913 polymorphism and RA susceptibility
To analyze the association between the IL-17A rs2275913 polymorphism and RA risk, we pooled the results of the 11 studies. No significant heterogeneity was present in the allelic model (I2 = 0%, P = 0.55), heterozygote model (I2 = 0%, P = 0.67), homozygote model (I2 = 12%, P = 0.33), dominant model (I2 = 0%, P = 0.69), or recessive model (I2 = 18%, P = 0.27). Therefore, a fixed-effects model was applied in these analyses. A significantly decreased risk of RA was observed in the allelic model (A vs. G: OR = 0.89, 95%CI: 0.83–0.95, P < 0.001; Figure 2A), heterozygote model (GA vs. GG: OR = 0.87, 95%CI: 0.78–0.96, P = 0.006; Figure 2B), homozygote model (AA vs. GG: OR = 0.82, 95%CI: 0.71–0.96, P = 0.01; Figure 2C), and dominant model (AA + AG vs. GG: OR = 0.86, 95%CI: 0.78–0.94, P = 0.002; Figure 2D). Although the recessive model (AA vs. AG + GG: OR = 0.89, 95%CI = 0.77–1.02, P = 0.09) showed no significant association between the IL-17A rs2275913 polymorphism and RA risk in the overall population, a trend of reduced risk was observed (Table 3).

Forest plots of the meta-analysis of the interleukin (IL)-17A rs2275913 polymorphism and rheumatoid arthritis (RA) in the overall population. (a) The allelic model (A vs. G). (b) The heterozygote model (GA/GG). (c) The homozygote model (AA vs. GG). (d) The dominant model (AA + GA vs. GG).
Summary of the association between the interleukin (IL)-17A rs2275913 polymorphism and rheumatoid arthritis (RA) susceptibility.

RA, rheumatoid arthritis; OR, odds ratio; CI, confidence interval; vs., versus; F, fixed effect model; PCR-RFLP, polymerase chain reaction restriction fragment length polymorphism assay; NA, not available.
Subgroup analyses based on ethnicity and genotyping method were then performed separately. The results suggest that a significant correlation exists between the IL-17A rs2275913 polymorphism and RA risk for the European population in the allelic model (A vs. G: OR = 0.87, 95%CI: 0.78–0.97, P = 0.01), heterozygote model (GA vs. GG: OR = 0.79, 95%CI: 0.68–0.93, P = 0.005), and dominant model (GA + AA vs. GG: OR = 0.79, 95%CI: 0.68–0.92, P = 0.003), as well as in a subgroup of individuals genotyped with the TaqMan method in the allelic model (A vs. G: OR = 0.89, 95%CI: 0.83–0.96, P = 0.003), heterozygote model (GA vs. GG: OR = 0.86, 95%CI: 0.77–0.96, P = 0.008), homozygote model (AA vs. GG: OR = 0.84, 95%CI: 0.71–0.99, P = 0.04), and dominant model (GA + AA vs. GG: OR = 0.86, 95%CI: 0.77–0.95, P = 0.004) (Table 3). No significant association was detected in the American, African, or polymerase chain reaction – restriction fragment length polymorphism (PCR-RFLP) subgroups (Table 3).
Sensitivity analysis and publication bias
Sensitivity analysis was conducted to examine the influence of the individual article on the pooled effects for the IL-17A rs2275913 polymorphism by omitting one study at one time. We found that in three models (allelic model: A vs. G, dominant model: GA + AA vs. GG, and heterozygote model: GA vs. GG), the pooled results remained significant except for the Norwegian population in Nordang’s study, 16 which indicated that the stability and reliability of this meta-analysis may be affected by this particular article. In addition, the Carvalho et al. study 24 results deviated from HWE, and the HWE of Louahchi et al. 27 was not available because of a lack of genotype frequency data. Therefore, sensitivity analysis was performed by removing these two studies from all model analyses. The results from the remaining studies showed that the pooled effects were not significantly altered by omitting these two studies. Moreover, the Dhaouadi et al. study 24 with low quality had no significant effect on the pooled results. Begg’s funnel plot was used to evaluate the publication bias. No significant publication bias was identified in our overall comparisons (Figure 3).
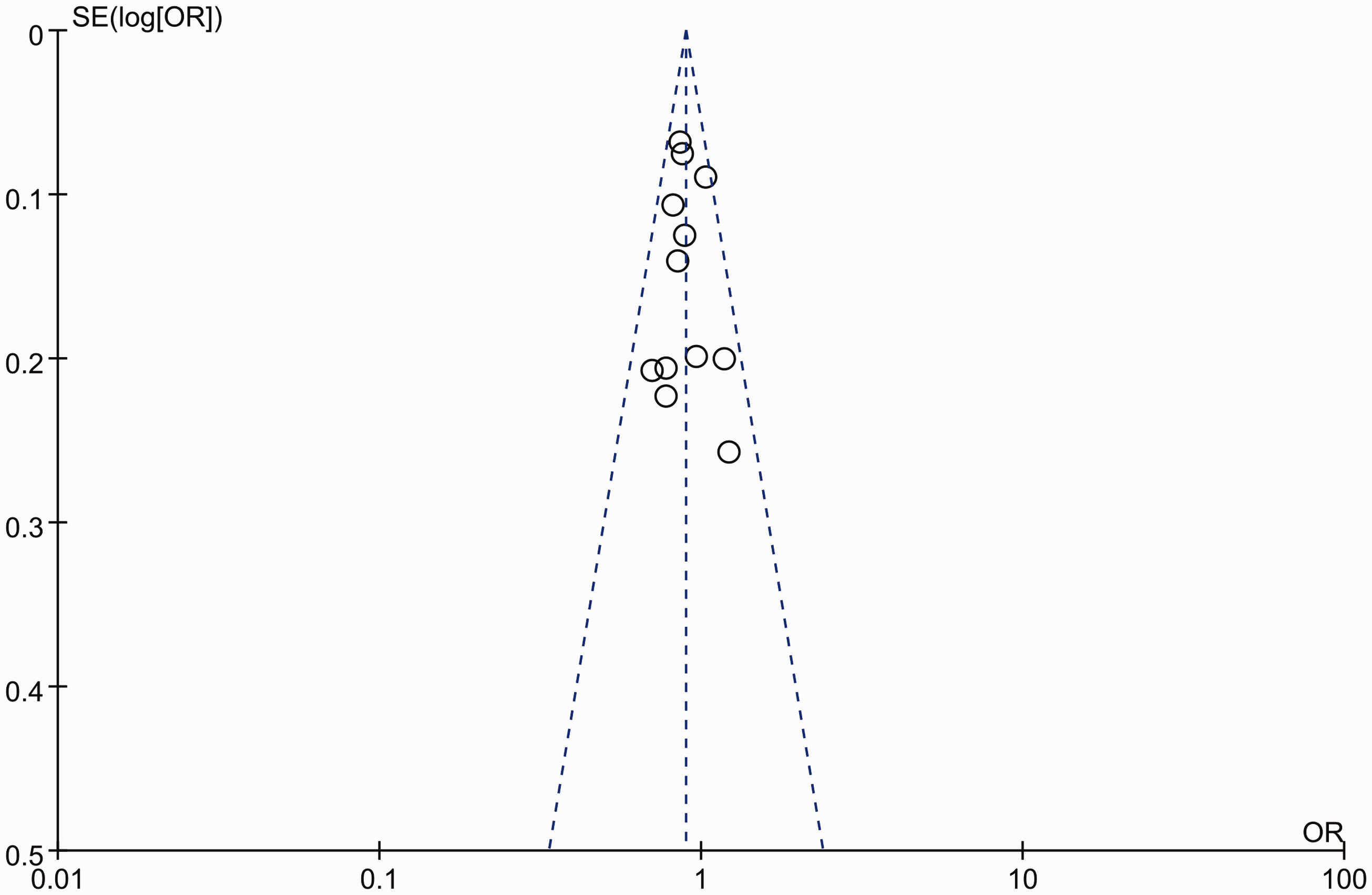
Begg’s funnel plot for publication bias of the interleukin (IL)-17A rs2275913 polymorphism for the allelic model (A vs. G).
TSA
TSA was conducted on the analyzed association between the IL-17A rs2275913 polymorphism and RA risk in the overall population with the allelic model (Figure 4A), heterozygote model (Figure 4B), homozygote model (Figure 4C), and dominant model (Figure 4D). Z-curves crossed the conventional test boundary and TSA boundary, and the total sample size reached the RIS in analyses with the allelic model. Although the Z-curves crossed the conventional test boundary in the analyses with the heterozygote, homozygote, and dominant models, they did not cross the TSA boundary nor the RIS boundary.

Trial sequential analysis (TSA) of the association between the interleukin (IL)-17A rs2275913 polymorphism and the rheumatoid arthritis (RA) risk in the overall population. (a) The allelic model (A vs. G). (b) The heterozygote model (GA/GG). (c) The homozygote model (AA vs. GG). (d) The dominant model (AA + GA vs. GG).
Discussion
Overall, 4019 RA cases and 4137 controls for the IL-17A rs2275913 polymorphism were included in our meta-analysis. By pooling all the eligible studies, a positive relationship between the IL-17A rs2275913 polymorphism and RA susceptibility was identified in the allelic, heterozygote, homozygote, and dominant models. This strengthens the evidence that IL-17A is a genetic susceptibility factor for developing this complex disease. In the European population, a significant relationship was found between the IL-17A rs2275913 polymorphism and RA risk in the allelic, heterozygote, homozygote, and dominant models. Consistently, a recent meta-analysis also found that IL-17A rs2275913 was significantly associated with RA risk in Whites by the allelic model. However, the results of the heterozygote, homozygote, and dominant models were not statistically significant. 30 We speculated that this difference was caused by a potential selection bias in Shao’s study because it included only eight publications with 1919 cases and 2126 controls. 30
Studies have shown that abnormal differentiation and dysfunction of CD4 + T cells is part of RA pathogenesis, and IL-17 is secreted by these CD4 + T cells. 31 IL-17A is located on chromosome 6. Previous reports revealed that there were high levels of IL-17A in RA synovial fluid and tissue explants, which led to joint degradation in ex vivo models. 8 IL-17A binds to a receptor complex that belongs to a specific subclass of cytokine receptors. 32 IL-17A could upregulate tumor necrosis factor receptor II (TNFRII) expression, which was the basis for synergy among these cytokines in synoviocytes. Therefore, IL-17A could induce pro-inflammatory cytokine production and promote the development of RA.32–34 The IL-17A rs2275913 polymorphism is located in the promoter region of the IL-17A gene, and cells with the 197A positive allele (genotypes GA/AA) secreted more IL-17A than cells with the 197A negative allele. 6 Hence, the IL-17A rs2275913 polymorphism results in more efficient secretion of IL-17A, which may be a potential mechanism by which this polymorphism can increase the risk of developing RA.
To more deeply understand the relationship between the IL-17A rs2275913 polymorphism and RA, we performed ethnicity-based subgroup analyses. Significant associations were found in the allelic, heterozygote, and dominant models in the European population. However, one report on the Chinese population suggested that there is no significant association in this group. 35 Because there was only one case–control study for Asians, no further conclusions can be made regarding the association between the IL-17A rs2275913 polymorphism and RA susceptibility. No significant association was found between the IL-17A rs2275913 polymorphism and RA susceptibility in the American and African groups. These observed ethnicity differences may be explained by the varied environmental exposures, lifestyles, and genetic backgrounds of these individuals. It is necessary to increase the number of studies and overall sample size to more conclusively examine the relationship between the IL-17A rs2275913 polymorphism and RA susceptibility in the Asian, American, and African populations.
There were several strengths of our work. First, compared with previous meta-analyses, 30 the present work included more studies with a larger cohort that included 4019 RA cases and 4137 controls. This helped strengthen the evidence that the IL-17A rs2275913 polymorphism is significantly associated with RA susceptibility. In addition, subgroup analysis, sensitivity analysis, and publication bias tests were carried out. Moreover, strict inclusion and exclusion criteria were implemented to ensure the quality of the included studies and reliability of the results. Finally, the selection bias was well-controlled via the literature search, and no publication bias was identified by Begg’s funnel plot. Similar to most other gene-related meta-analyses, this study has some limitations. First, the sample sizes of the Asian, American, and African populations were relatively small. Future studies are needed to increase the number of patients in these populations to clarify whether individuals with the IL-17A rs2275913 polymorphism have a significantly different risk of suffering from RA. Second, the sensitivity analysis suggested that the Norwegian population in Nordang et al. 16 had an impact on the pooled ORs of the allelic, dominant, and heterozygote models. This is possibly because of the relatively large sample size of this study compared with other studies. Third, there was one study that deviated from HWE 24 and one without HWE information. 27 Although the pooled effects were not significantly influenced by these two studies, potential uncertainty factors may be present in the pooled efficiency.
In conclusion, despite the limitations described above, our results suggest that the IL-17A rs2275913 polymorphism is significantly associated with a decreased risk of developing RA, especially in Europeans. However, caution should be taken because of the population heterogeneity and limited number of cases for different races included in this meta-analysis. Well-designed case-control studies with a larger sample size and multiple ethnicities are required to strengthen the results of the current study.
