Abstract
Objective
The risk factors associated with iodine contrast extravasation immediately after endovascular thrombectomy (EVT) and subsequent hemorrhagic transformation within 24 hours remain unclear.
Methods
Mixed images, iodine overlay maps, and virtual non-contrast images were reconstructed from 106 consecutive acute ischemic stroke patients who underwent dual energy computed tomography immediately and 24 hours after EVT. Multivariate analyses of clinical and radiological data were performed to explore independent predictors of iodine contrast extravasation and hemorrhagic transformation.
Results
Sixty-eight (64.2%) patients exhibited pure iodine contrast extravasation after EVT; 30.9% developed hemorrhagic transformation within 24 hours after EVT. The number of stent retriever passes was independently associated with both iodine contrast extravasation (odds ratio 1.608; 95% confidence interval (CI) 1.047–2.469) and subsequent hemorrhagic transformation (odds ratio 1.477; 95% CI 1.003–2.175). Patients with more than two stent retriever passes were more likely to exhibit iodine contrast extravasation (sensitivity = 68.2%, specificity = 81.5%), while those with more than three stent retriever passes more often exhibited hemorrhage after iodine contrast extravasation (sensitivity = 64.6%, specificity = 87.2%).
Conclusions
The number of stent retriever passes was an independent predictor for both iodine contrast extravasation and subsequent hemorrhagic transformation.
Keywords
Introduction
In 2015, five similar randomized trials demonstrated the efficacy of endovascular thrombectomy (EVT) over standard medical care for patients with large artery occlusions. 1 EVT is increasingly performed in clinical practice because it is recommended for use in acute stroke patients with occlusion of the internal carotid artery or middle cerebral artery segment 1 by the current guidelines. 2 As a consequence, more than half of stroke patients present hyperdense regions on noncontrast computed tomography (CT) after EVT, 3 which are caused by contrast staining (iodine extravasation) and/or early hemorrhagic transformation.
Dual energy CT (DECT) allows the differentiation of iodine contrast medium from hemorrhage. 4 Using DECT, an iodine overlay map (IOM) and a virtual non-contrast (VNC) image can be reconstructed to show the attenuation characteristics of iodine and blood at two selected energy levels (80 kV and 140 kV).5,6 In addition, three types of radiological outcomes are observed on 24-hour follow-up images of patients who present with pure iodine contrast extravasation immediately after EVT: iodine contrast washout, persistent iodine extravasation, and evolution to hemorrhage. Despite the blood–brain barrier injury hypothesis, 7 the risk factors associated with iodine contrast extravasation and subsequent hemorrhagic transformation remain unclear.
In the current study, we examined DECT images obtained immediately and 24 hours after EVT to differentiate between iodine contrast extravasation and hemorrhage. We aimed to investigate the risk factors of iodine contrast extravasation and subsequent hemorrhagic transformation after thrombectomy.
Methods
We retrospectively reviewed data from ischemic stroke patients who received EVT and underwent DECT immediately and 24 hours later between January 2016 and October 2018. Patients with previous ischemic stroke within 3 months were excluded from this study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 8
The DECT protocols performed on acute ischemic stroke patients who underwent EVT were approved by the Human Ethics Committee of Hangzhou First Hospital, Zhejiang University School of Medicine (2016006-01). All clinical investigations were conducted according to the principles expressed in the Declaration of Helsinki. Written informed consent was obtained from all patients.
Image analysis
DECT was performed with a dual source 128-slice CT scanner (Siemens Healthcare, Germany). The CT parameters used for acquisition and reconstruction were as follows: a dedicated dual-source protocol with simultaneous imaging at 80 kV/392 effective mAs and 140 kV(Sn)/196 effective mAs, collimation of 0.6 mm, and pitch of 0.7. The raw spiral projection data were rebuilt in three series, with two sets corresponding to 80 and 140 kV (0.6-mm slice thickness) and a third set corresponding to a mixed map of both energies (80 kV/140 kV), simulating a conventional 120-kV CT scan. Using the characteristic iodine absorption behavior at high- and low-kV peak CT, the Hounsfield unit value of any voxel can be decomposed into the contribution from the iodine content and the virtual nonenhanced contribution. VNC images and IOMs were calculated using a dedicated brain hemorrhage algorithm. Iodine contrast extravasation and hemorrhage were defined as previously described. 9 Briefly, hemorrhage manifested as hyperdense regions on VNC images and isodense regions on IOMs, while iodine contrast extravasation manifested as isodense regions on VNC images and hyperdense regions on IOMs, Both iodine contrast extravasation and hemorrhage were limited to the brain parenchyma. Two neurologists separately reviewed the reconstructed DECT images, and disagreements were resolved by an interventional neurologist. Hemorrhagic transformation was classified by hemorrhagic infarction, parenchymal hemorrhage, and symptomatic intracranial hemorrhage according to the European Cooperative Acute Stroke Study II criteria. 10 Successful reperfusion was defined as a modified thrombolysis in cerebral infarction score of 2b or 3. 11
Statistical analysis
A Pearson χ2 test or Fisher's exact test was used to compare dichotomous variables between groups, while an independent samples two-tailed t-test or the Mann–Whitney U test was used to compare continuous variables, as appropriate. One-way analysis of variance or the Kruskal–Wallis test was used for comparisons of multiple groups. Variables with a value of p < 0.1 in the univariate regression analyses were included in the logistic regression model. Statistical significance was set at a p value of <0.05. All statistical analyses were performed with the SPSS 23.0 statistical package (IBM Corp., Armonk, NY, USA).
Results
The inter-reader agreement κ values were 0.86 for iodine contrast extravasation on IOMs and 0.76 for hemorrhage on VNC images, and the intra-reader κ values were 0.94 for iodine contrast extravasation on IOMs and 0.92 for hemorrhage on VNC images immediately after EVT. The inter-reader agreement κ values were 0.84 for iodine contrast extravasation on IOMs and 0.73 for hemorrhage on VNC images, and the intra-reader κ values were 0.91 for iodine contrast extravasation on IOMs and 0.92 for hemorrhage on VNC images 24 hours after EVT.
Of the 106 included patients, 67 (63.2%) were men, with a median age of 74 years (mean 72 ± 10 years, range 27–86 years), and acute large vessel occlusions were distributed as follows: 27 (25.5%) in the internal carotid artery, 57 (53.8%) in the middle cerebral artery, 5 (4.7%) in the anterior cerebral artery, 4 (3.8%) in the posterior cerebral artery, 3 (2.8%) in the vertebral artery, and 10 (9.4%) in the basilar artery. Each patient underwent stent retriever thrombectomy (Solitaire, Medtronic, Minneapolis, MN, USA) without using a direct aspiration first pass technique. Most patients were treated under conscious sedation (69.8%) rather than general anesthesia (30.2%). Reperfusion was successfully achieved in 89 (84.0%) of the included patients. The detailed univariate analysis of characteristics of patients with different radiological signs on DECT immediately after thrombectomy is shown in Table 1.
Univariate analysis of characteristics of patients with different radiological signs on DECT immediately after thrombectomy.
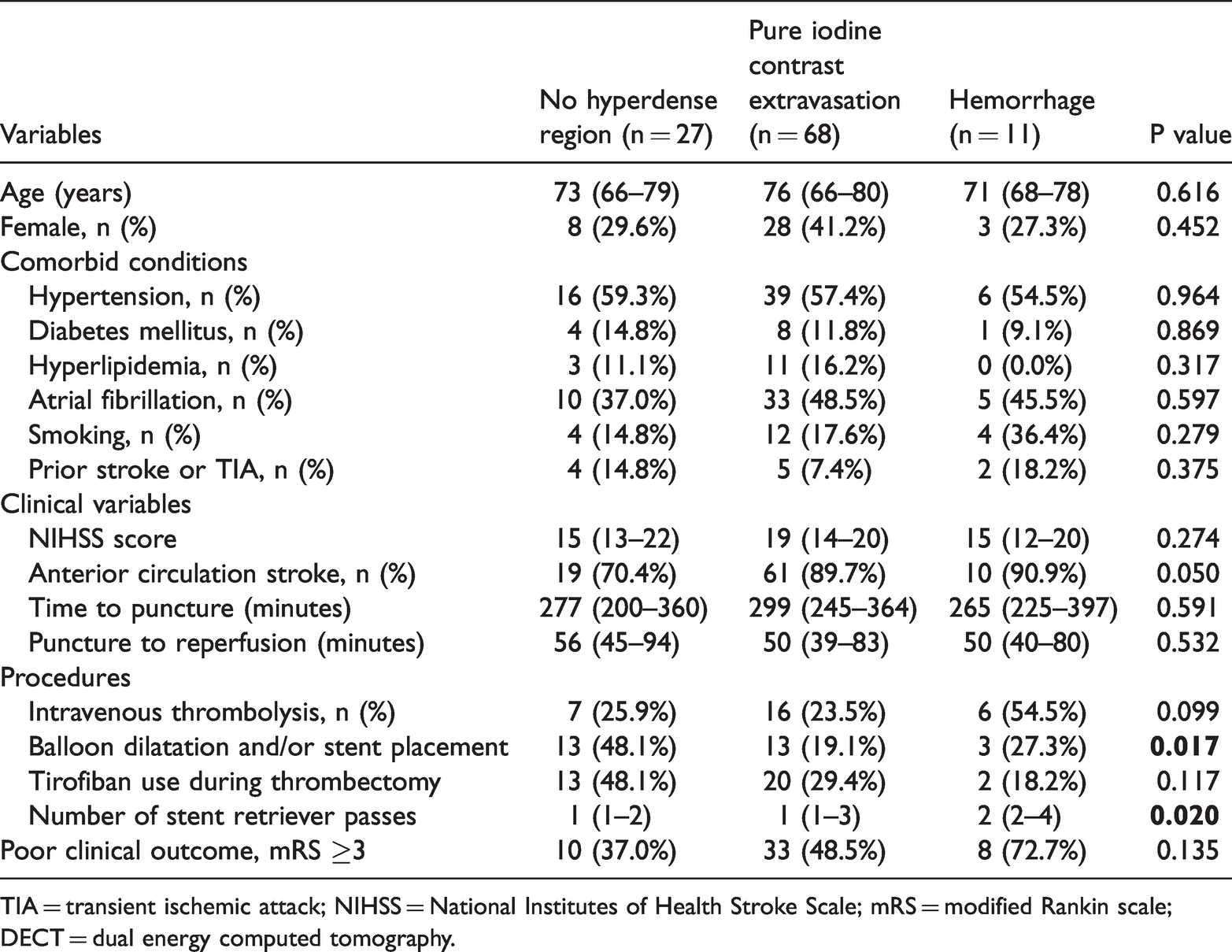
TIA = transient ischemic attack; NIHSS = National Institutes of Health Stroke Scale; mRS = modified Rankin scale; DECT = dual energy computed tomography.
On the basis of IOM and VNC images acquired by DECT immediately after EVT, 68 (64.2%) patients were classified as exhibiting pure iodine contrast extravasation, 11 (10.4%) patients were classified as having early hemorrhagic transformation (5 with hemorrhagic infarction, 6 with parenchymal hemorrhage), and the remaining 27 (25.5%) patients showed no hyperdense regions. The results of these patients are shown in Figure 1. Of patients with pure iodine contrast extravasation immediately after EVT, 30.9% (21 of 68) exhibited hemorrhagic transformation (17 with hemorrhagic infarction, 4 with parenchymal hemorrhage) on VNC images 24 hours after EVT (Figure 2). Eight patients developed symptomatic intracranial hemorrhage within 24 hours after EVT.

Distribution of the results of patients who underwent dual energy computed tomography (DECT) immediately and 24 hours after endovascular thrombectomy (EVT). HT, hemorrhagic transformation.

Examples of pure iodine contrast extravasation immediately after endovascular thrombectomy (EVT) and three types of radiological outcomes 24 hours after EVT. (a), (b), and (c) show three different patients and, from left to right, show mixed images immediately after EVT, virtual non-contrast (VNC) images immediately after EVT, mixed images 24 hours after EVT, and VNC images 24 hours after EVT. (a) A patient with pure iodine extravasation immediately after EVT presented iodine contrast washout 24 hours after EVT. (b) A patient with pure iodine extravasation immediately after EVT presented persistent iodine extravasation 24 hours after EVT. (c) A patient with pure iodine extravasation immediately after EVT presented hemorrhagic transformation 24 hours after EVT.
Anterior circulation stroke, combined intravenous thrombolysis, balloon dilatation and/or stent placement procedures, and the number of stent retriever passes were included in the binary logistic regression model for iodine contrast extravasation among patients without hemorrhagic transformation immediately after EVT. Balloon dilatation and/or stent placement procedures independently decreased the presence of iodine contrast extravasation (odds ratio 0.309; 95% confidence interval (CI) 0.113–0.842; p = 0.022), whereas the number of stent retriever passes was an independent risk factor for iodine contrast extravasation (odds ratio 1.608 for each pass; 95% CI 1.047–2.469; p = 0.030) (Table 2). The median number of stent retriever passes was 1 (interquartile range (IQR) 1–3) for patients with balloon dilatation and/or stent placement and 2 (IQR 1–3) for those without these procedures, resulting in no significant difference between groups.
Binary logistic regression analysis of radiological outcomes immediately after EVT.

EVT = endovascular thrombectomy; HT = hemorrhagic transformation; OR = odds ratio; CI = confidence interval.
Prior ischemic stroke or transient ischemic attack, time to puncture, tirofiban use during thrombectomy, and number of stent retriever passes were included in the binary logistic regression model for subsequent hemorrhagic transformation 24 hours after EVT among patients who presented with pure iodine contrast extravasation immediately after EVT. Prior ischemic stroke or transient ischemic attack (odds ratio 12.789; 95% CI 1.233–132.637; p = 0.033) and the number of stent retriever passes were independent risk factors for subsequent hemorrhagic transformation after iodine contrast extravasation (odds ratio 1.477; 95% CI 1.003–2.175; p = 0.048) (Table 3). In addition, patients with more than two stent retriever passes were more likely to exhibit iodine contrast extravasation (sensitivity = 68.2%, specificity = 81.5%), while those with more than three stent retriever passes were more likely to progress to hemorrhage from iodine contrast extravasation (sensitivity = 64.6%, specificity = 87.2%).
Binary logistic regression analysis of radiological outcomes 24 hours after EVT.
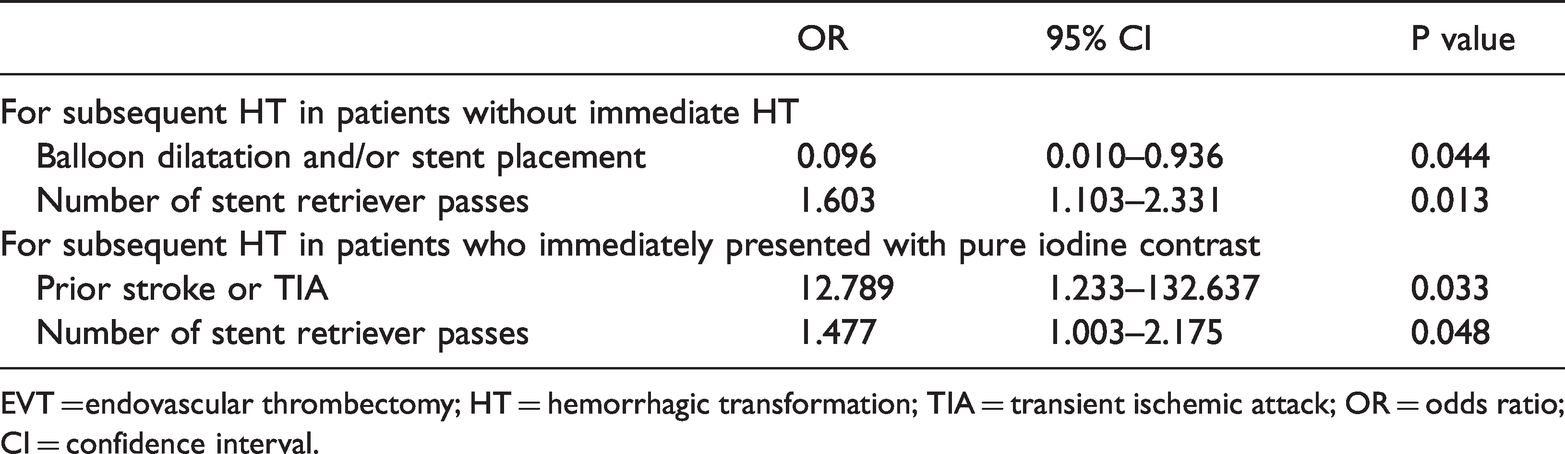
EVT = endovascular thrombectomy; HT = hemorrhagic transformation; TIA = transient ischemic attack; OR = odds ratio; CI = confidence interval.
Discussion
More than 60% of acute ischemic stroke patients presented with pure iodine contrast extravasation immediately after EVT. Approximately 30% of these patients developed hemorrhagic transformation within 24 hours after EVT. Patients with more than two stent retriever passes were more likely to have iodine contrast extravasation, while those with more than three stent retriever passes were more likely to progress to hemorrhage from iodine contrast extravasation.
A previous study reported that the frequency of hyperdense lesions was 84.2% in patients after EVT, 3 most of which were caused by pure iodine contrast extravasation. The proportion of pure contrast extravasation in our study is higher than those in three previously published studies of post-interventional DECT within 30, 60, or 120 minutes (68%, 47%, and 32%, respectively).5,12,13 Several previous studies investigated the risk factors for iodine contrast extravasation and subsequent hemorrhagic transformation after EVT. In the current study, we found that balloon dilatation and/or stent placement procedures independently decreased the presence of iodine contrast extravasation immediately after EVT. Two possible mechanisms are suggested: 1) reperfusion achieved with the aid of balloon dilatation and/or stent placement leads to less vascular endothelial injury; 2) patients who require balloon dilatation and/or stent placement procedures usually have atherosclerosis and previous artery stenosis, thus they have better collateral circulation. 14 The number of stent retriever passes was correlated with the presence of iodine contrast extravasation, which might be attributed to vascular endothelial injury, especially in patients with more than two passes. Moreover, we found that a history of ischemic stroke or transient ischemic attack and the number of stent retriever passes (more than three) were independently associated with evolution to subsequent hemorrhagic transformation from iodine contrast extravasation, which was explained by subacute and acute blood–brain barrier breakdown, respectively.
Several previous studies demonstrated that a high number of device passes was associated with worse outcomes in patients with EVT.15,16 Flottmann et al. 16 further assessed the recanalization rate and clinical outcome per retrieval maneuver and found that patients with one to three retrieval attempts had higher good clinical outcome rates. Further attempts (more than three) resulted in good recanalization rates, but the favorable clinical outcome rate did not improve. 16 The increased risk of hemorrhage might limit the benefit of recanalization when the number of stent retriever passes exceeds three.
There are several limitations of our study. First, this is a retrospective study conducted in a single stroke center, although we prospectively collected data using a stroke registry. Second, the sample size is moderate. Future studies with large sample sizes and individual patient data meta-analyses are needed to confirm the risk factors of contrast extravasation and subsequent hemorrhagic transformation after EVT. Third, hemosiderin-sensitive magnetic resonance sequences, which are clinically difficult, were not performed immediately after EVT to distinguish contrast extravasation from hemorrhage. 17 Fourth, the effects of the Alberta Stroke program early CT score, collaterals, and the clot burden score were not evaluated in the current study because the sample size did not allow subgroup analyses for anterior circulation and posterior circulation stroke. These variables, along with CT perfusion parameters, should be investigated in future studies.
Conclusion
The number of stent retriever passes was correlated with both iodine contrast extravasation and subsequent hemorrhagic transformation.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (81701150) and the Basic Public Welfare Research Project of Zhejiang Province (LGF18H090017).
