Abstract
Several clinical trials have proven that immunotherapy can improve survival and benefit non-small cell lung cancer (NSCLC) patients. In patients who progress after chemotherapy, immune checkpoint inhibitor (ICI) monotherapy can prolong overall survival compared with patients receiving single-agent chemotherapy. A 61-year-old man diagnosed with advanced NSCLC and without driver variants received first-line chemotherapy but experienced recurrence. During subsequent treatment, the disease progressed rapidly, and his general condition deteriorated; therefore, toripalimab monotherapy was initiated. Surprisingly, he responded well, and symptoms were relieved after several treatment cycles despite pseudoprogression, shown in chest images. For driver gene-negative NSCLC patients who progress after chemotherapy and who develop poor performance status (PS), ICIs are an option to alleviate symptoms and improve survival. Furthermore, immunotherapy in patients with pseudoprogression may also provide a survival benefit.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related death in the world. Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancers and is usually diagnosed at an advanced stage. 1 The prognosis of these patients remains poor. Standard treatment for NSCLC patients with metastatic disease is platinum doublet therapy with gemcitabine, vinorelbine, or taxanes, and with pemetrexed in patients with the nonsquamous histologic type. 2 For patients with driver variants, targeted therapies, such as epidermal growth factor receptor tyrosine kinase inhibitors (TKIs) and anaplastic lymphoma kinase (ALK) TKIs, are also recommended strategies. 2 Recently, immunotherapy has become a highly promising therapeutic approach for lung cancer. Immune checkpoint inhibitors (ICIs), such as programmed cell death 1 (PD-1) and PD ligand 1 (PD-L1) monoclonal antibodies, have tumor-suppressing effects, facilitating a positive immune response to kill tumor cells. 3 The use of these drugs significantly improves survival in patients with advanced NSCLC, especially in those without driver variants. 4 Two PD-1 monoclonal antibodies, pembrolizumab and nivolumab, were approved for second-line chemotherapy in NSCLC by the United States Food and Drug Administration (USFDA) in 2015; atezolizumab, a PD-L1 monoclonal antibody, was approved for the same indication in 2016 5 . Toripalimab, a recombinant, humanized PD1 monoclonal antibody, has been approved in China to treat metastatic melanoma patients who do not respond to systemic therapy. 6 There are also ongoing clinical trials evaluating toripalimab in various types of cancers, including NSCLC. Here, we report a patient with NSCLC who progressed after several chemotherapy cycles and who survived after receiving toripalimab.
Case report
A 61-year-old man with a 60 pack-year smoking history visited our hospital in June 2017 with a chief complaint of right hip pain for 5 months. Bone scintigraphy showed multiple imaging abnormalities, indicating possible metastasis. Positron emission tomography-computed tomography (PET-CT) revealed a hypermetabolic mass (5.3 × 4.0 cm) in the anterior segment of the upper lobe of the right lung invading the right hilar region and mediastinum. Signs of metastasis were as follows: several nodules were present in both lungs and the right pleura; multiple hypermetabolic lymph nodes were seen in the left neck, bilateral clavicular region, mediastinum, bilateral hilar region, and right cardiac diaphragmatic angle; hypermetabolic nodules were seen in bilateral adrenal glands; and bone destruction with metabolic activity was identified. The patient underwent bronchoscopy and was diagnosed with adenocarcinoma through pathology. Molecular analysis showed no driver gene mutations.
We diagnosed advanced lung adenocarcinoma on histopathology (Figure 1) according to the 8th edition of the American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM) staging system (stage: T4N3M1c, IVB) (Figure 2a). Between 15 July 2017 and 28 November 2017, we administered six cycles of chemotherapy combined with targeted therapy consisting of pemetrexed, carboplatin, and bevacizumab (pemetrexed: 500 mg/m2 on day 1, carboplatin (area under the curve (AUC) = 5) on day 1, and bevacizumab 7.5 mg/kg on day 1; repeated every 21 days), after obtaining patient consent for treatment. We administered zoledronic acid concurrently, for bone metastasis. After four cycles, enhanced chest CT revealed partial response (PR) (Figure 2b). According to the patient’s wishes, he received only one cycle of pemetrexed and bevacizumab maintenance therapy, on 23 January 2018. Periodic surveillance scans demonstrated stable disease until November 2018. Enhanced chest CT scan at that time showed enlargement of the primary lesion (from 30 × 23 mm to 48 × 41 mm) and an increase in the number of subpleural nodules (Figure 2c). In April 2019, the patient underwent enhanced brain magnetic resonance imaging, which revealed a new metastasis in the cortex of the frontal lobe. The progression-free survival with first-line chemotherapy (PFS1) was 16 months.

The patient’s pathology findings (hematoxylin and eosin, ×109).
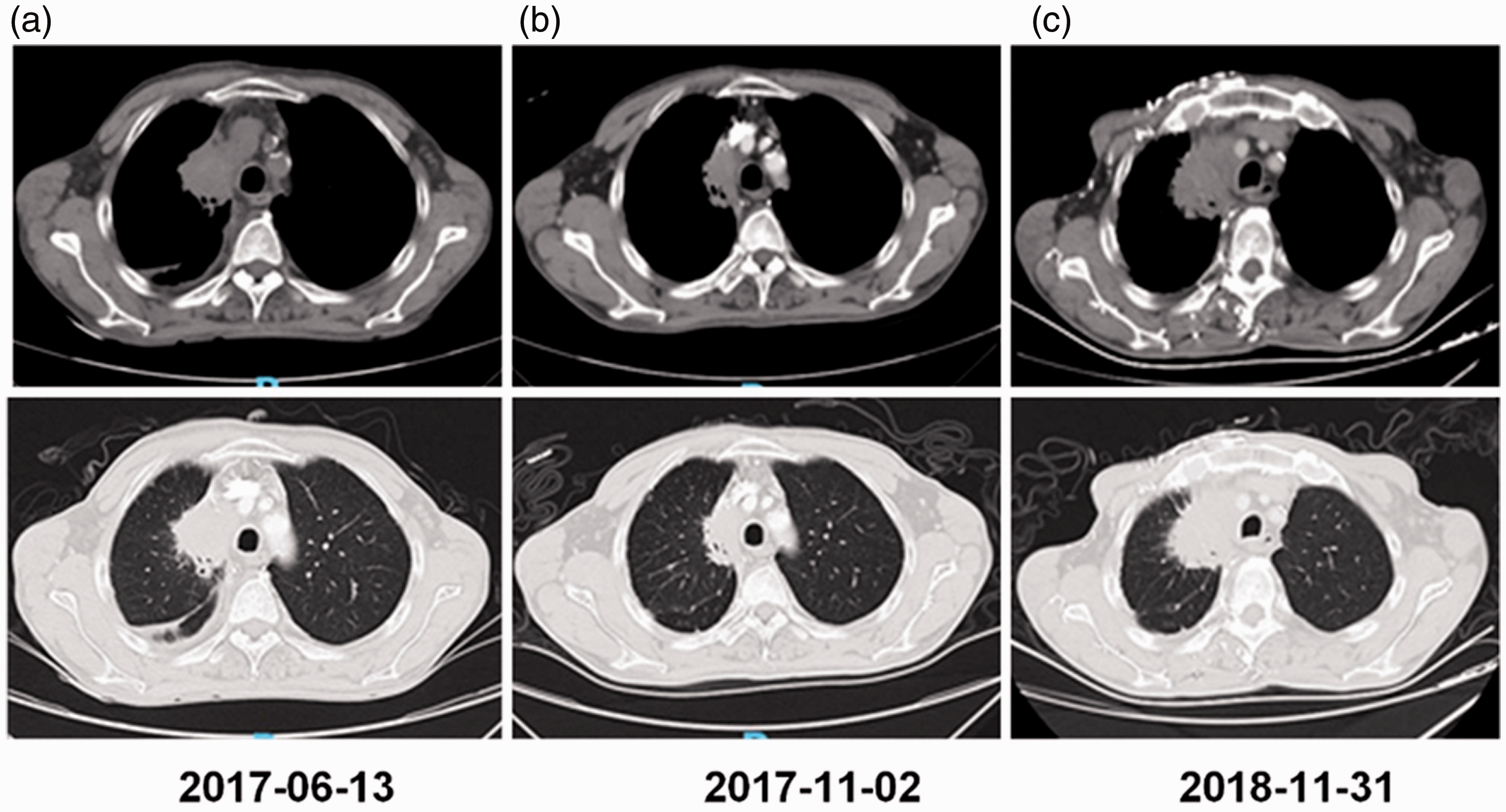
Computed tomography (CT) images (a) Baseline images. (b) Enhanced CT images after four cycles of first-line pemetrexed, carboplatin, and bevacizumab treatment. (c) Enhanced CT images indicating a relapse of the primary lesion.
The patient received two cycles of pemetrexed and carboplatin combined with bevacizumab rechallenge therapy, but enhanced CT performed after two cycles showed progressive disease (PD) (Figure 3a–c) with newly identified bone metastasis. He then received two cycles of chemotherapy with Abraxane (Celgene Corp., Summit, NJ, USA) which also failed (Figure 4a). We changed the drug to anlotinib targeted therapy for two cycles, which also resulted in PD, as demonstrated on chest CT (Figure 4b). The patient’s general condition deteriorated during the several lines of treatment, and he developed dysphagia and underwent gastric tube placement because of esophageal wall thickening. Other symptoms, namely cough and chest tightness, were also exacerbated, and he found it difficult to lie flat at night and could walk only approximately 100 m owing to shortness of breath. The Eastern Cooperative Oncology Group (ECOG) and Performance Status (PS) score was 2. The results indicated that indicated PS 2 patients receiving ICIs have worse overall survival (OS), which was in agreement with the findings of Dall’Olio et al., (2020) who demonstrated that PS2 patients treated with chemotherapy had worse outcomes compared with PS 0 to 1 patients. 7 However, the Checkmate 171 trial demonstrated that nivolumab is well tolerated in PS 2 patients; however, these patients may experience a higher risk of toxicity if they receive chemotherapy. 8 Considering the patient’s condition, we decided to administer the PD-1 inhibitor, toripalimab (200 mg on day 1, every 21 days), on 18 October 2019.

Enhanced computed tomography images (a) Newly identified bone metastasis. (b) Images after two cycles of pemetrexed and carboplatin combined with bevacizumab rechallenge therapy (Mediastinal window). (c) Images after two cycles of pemetrexed and carboplatin combined with bevacizumab rechallenge therapy (Lung window).
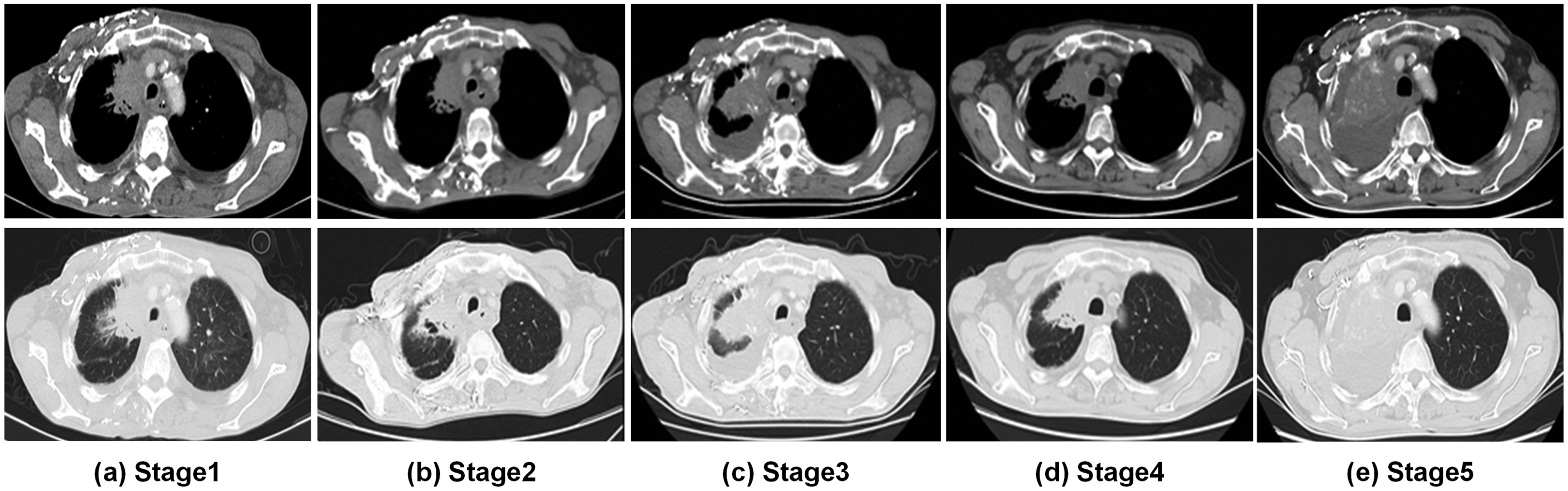
Computed tomography images (a) Images after two cycles of Abraxane treatment; (b) Images after two cycles of anlotinib treatment; (c) Images indicating pseudoprogression after two cycles of toripalimab; (d) Follow-up chest images showing stable disease; (e) Newly-emerging pleural effusion.
After two cycles of immunotherapy, chest CT revealed some signs of PD (Figure 4c), namely an increase in the size of the primary lesion, and pleural effusion and worse atelectasis; however, a slight reduction in the thickness of the esophageal wall was noted. The patient’s general condition did not worsen significantly, according to the imaging findings. Thus, we considered this phenomenon to be a pseudoprogression, and administered another two cycles of toripalimab. Imaging after four cycles of toripalimab demonstrated reductions in the size of the primary lesion, lymph nodes, pleural effusion, and esophageal wall thickening. In the following months, he achieved stable disease (SD) according to the Response Evaluation Criteria in Solid Tumors version 1.1, evaluated by CT. 9 CT indicated that the diameter of the mass in the upper lung lobe had decreased in size slightly from 48 mm to 43 mm, the thickness of the esophageal had decreased noticeably from 14 mm to 7 mm, and the short diameter of the supraclavicular lymph node had decreased from 27 mm to 12 mm (Figure 4d).
The patient’s general condition gradually improved, the gastric tube was removed, and his activity tolerance increased; he was able to walk approximately 1000 meters. Toripalimab was administered for 20 cycles, and he survived following this treatment. Unfortunately, during the follow-up in November 2020, chest CT indicated massive pleural effusion in the right thorax (Figure 4e). At this point, the patient had achieved PFS 2 for 13 months. Although we confirmed PD, the ECOG score remained at 1, and the PS score remained at 90, and he experienced a remarkable improvement in his quality of life for more than 1 year (Figure 5). Unfortunately, owing to the patient’s wishes, we did not perform a biopsy as the disease progressed. Therefore, we also could not confirm the pseudoprogression through biopsy.
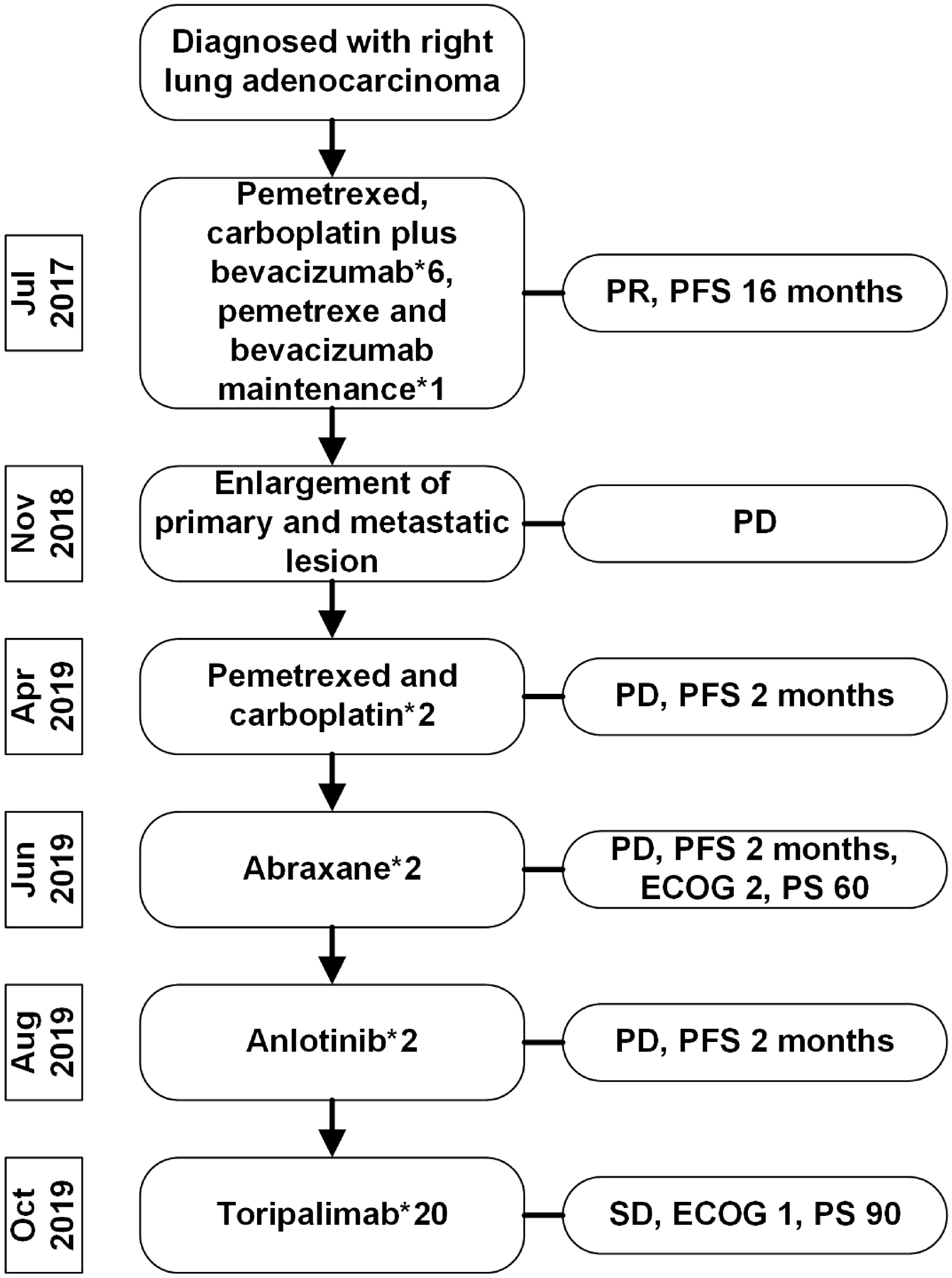
The patient’s timeline.
The reporting of this study conforms to the CARE guidelines. 10
Discussion
For advanced NSCLC patients without driver variants, chemotherapy is the optimal choice. First-line chemotherapy comprising platinum-based regimens produces response rates between 15% and 30% in advanced NSCLC patients, and second-line therapy, generally comprising taxane-based regimens, produces a relatively low response.5,11 Thus, multiple clinical trials are on-going to identify novel agents for metastatic NSCLC. A phase I clinical trial, the KEYNOTE-001 trial, selected PD-L1 tumor proportion score (TPS) of at least 50% as the cutoff and found that among patients meeting the cutoff, the response rate of pembrolizumab was 45.2%, and the median PFS was 6.3 months. The study also established the clinical efficacy and safety of pembrolizumab. 12 The KEYNOTE-010 study demonstrated that pembrolizumab prolonged OS in PD-L1-positive NSCLC patients who progressed after at least platinum-based chemotherapy. 13 Subsequently, the USFDA approved pembrolizumab for first-line treatment of advanced NSCLC patients with PD-L1 ≥50%, in accordance with the KEYNOTE-024 study. 14 Similarly, an international phase III study, the CheckMate 057 study, confirmed that nivolumab improved OS in advanced nonsquamous NSCLC patients who failed to response to platinum-based chemotherapy versus docetaxel. 15
Although toripalimab has not been approved as an NSCLC treatment, some phase I clinical trials reported the potential utility of toripalimab in lung cancer.4,16 However, these clinical trials often selected patients with ECOG scores ranging from 0 to 1, and there are few studies evaluating immunotherapy in patients with poor general condition. A systematic review demonstrated that poor PS did not affect the safety profile of immunotherapy; however, the OS of this group was half that of PS 0 to 1 patients. 17 Francesco et al. 18 found that in NSCLC PS 2 patients with PD-L1 TPS ≥50% who received first-line pembrolizumab, 27% of the patients experienced improved PS scores, although median PFS and OS were only 2.4 and 3 months, respectively. Regarding toripalimab, a multicenter retrospective study analyzed 166 NSCLC patients who received toripalimab. 19 The median PFS was 15.0 months (95% confidence interval (CI): 10.2–NA). The overall response rate was 21.3%, and the disease control rate was 81.7%; 28 patients received toripalimab as monotherapy and achieved a PFS of 15.0 months (95% CI: 12.6–15.0). 19 Another phase I study proved that toripalimab was well tolerated in alveolar soft-part sarcoma, NSCLC, and in lymphoma patients who failed to respond to systemic treatment. The median PFS of solid tumors reached 5.7 months. 16 After multiline therapy, our patient developed a poor PS and could not tolerate further chemotherapy; therefore, we chose immunotherapy alone as maintenance therapy. After consulting with the patient, he agreed to receive toripalimab, considering the cost. Surprisingly, his symptoms were noticeably relieved, and his general condition improved dramatically, with no obvious immune-related side effects; he achieved a PFS of 13 months.
The chest CT images of our patient indicated pseudoprogression after two cycles of treatment; however, subsequent images confirmed SD regarding the primary lesion, improvement in the esophageal wall thickening, and a reduction in the size of the lymph nodes. Pseudoprogression refers to patients who first experience an increase in tumor size and the development of new lesions, followed by a decrease in mass size. Pseudoprogression is commonly seen in patients when they begin immunotherapy, with an incidence rate of 0.6% to 5%, in NSCLC. 20 In contrast, hyperprogression refers to an increase in tumor growth rate by a factor of two after initial treatment, with an incidence rate between 4% and 29%, indicating acceleration of the progression pace and which leads to deleterious effects.21,22
Current studies reveal that patients with pseudoprogression experience a shorter response time than those who show a typical response; however, the OS in pseudoprogression patients is longer than with typical progression, 23 and patients with hyperprogression have a lower survival rate. 24 It is commonly agreed that PS and symptom assessment be used to differentiate the two clinical statuses. For patients with pseudoprogression, continuing immunotherapy has a potential benefit for survival. In our case, although imaging indicated newly emerging pleural effusion, the patient did not complain of worsened shortness of breath. Therefore, we administered another two cycles of toripalimab. Subsequent CT images revealed response to the treatment, with improved esophageal wall thickening, decreased lymph node size, and SD in the primary lesion. Most importantly, he showed dramatic quality of life improvement, with better general condition.
In conclusion, we report the case of an advanced NSCLC patient who received multiline treatment and subsequent toripalimab monotherapy because of a poor ECOG score. After immunotherapy, his symptoms were relieved, and he was progression-free for 13 months; he is alive at the time of this report. This case reminds us that for patients with poor general condition who are intolerant of chemotherapy, ICIs are feasible options. Although patients may develop pseudoprogression, continued use of ICIs may benefit patients who ultimately respond to therapy.
Footnotes
Declaration of conflicting interest
All authors have completed an ICMJE uniform disclosure form. The authors declare that there is no conflict of interest.
Ethics statement
All procedures performed in this study involving human participants were in accordance with the Helsinki Declaration (as revised in 2013). Our institution does not require ethics approval for case reports. We have obtained the patient’s written informed consent to publish the details of his case, and we have de-identified all patient details.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
