Abstract
Objective
This study aimed to establish and internally verify the risk nomogram of postoperative acute kidney injury (AKI) in patients with renal cell carcinoma.
Methods
We retrospectively collected data from 559 patients with renal cell carcinoma from June 2016 to May 2019 and established a prediction model. Twenty-six clinical variables were examined by least absolute shrinkage and selection operator regression analysis, and variables related to postoperative AKI were determined. The prediction model was established by multiple logistic regression analysis. Decision curve analysis was conducted to evaluate the nomogram.
Results
Independent predictors of postoperative AKI were smoking, hypertension, surgical time, blood glucose, blood uric acid, alanine aminotransferase, estimated glomerular filtration rate, and radical nephrectomy. The C index of the nomogram was 0.825 (0.790–0.860) and 0.814 was still obtained in the internal validation. The nomogram had better clinical benefit when the intervention was decided at the threshold probabilities of >4% and <79% for patients and doctors, respectively.
Conclusions
This novel postoperative AKI nomogram incorporating smoking, hypertension, the surgical time, blood glucose, blood uric acid, alanine aminotransferase, the estimated glomerular filtration rate, and radical nephrectomy is convenient for facilitating the individual postoperative risk prediction of AKI in patients with renal cell carcinoma.
Keywords
Introduction
The incidence of renal cell carcinoma has been increasing in recent years. Currently, partial nephrectomy (PN) is the gold standard for T1 renal masses. Recent reports have indicated that even in case of T2 renal masses, PN is a viable option with expert surgeons, and it confers a benefit in renal function compared with radical nephrectomy (RN).1,2 However, nephrectomy is still a risk factor for postoperative deterioration of renal function. New consensus standards and an increasing amount of reports on the incidence of acute renal injury after RN or PN and its adverse effects on long-term renal function and mortality have led to awareness of acute renal injury (AKI) after RN or PN. 3 However, some reports have indicated that PN can cause a certain amount of functional damage, even in patients with a preoperative normal baseline renal function. 4 AKI is a serious disease and complication, and is characterized by persistent oliguria and elevated serum creatinine levels, with high morbidity and mortality rates. 5 AKI is associated with a higher rate of postoperative complications, longer hospital stay, higher mortality rate, and higher costs. 6 Some studies have shown that the incidence of AKI has a higher risk of reduction in the estimated glomerular filtration rate (eGFR) between 3 and 15 months after an operation. 7 In this study, we creatively used a nomogram to examine the relationship between clinical factors and postoperative AKI. This nomogram can hopefully be used as a reliable tool to predict postoperative AKI and guide decision-making for nephrectomy.
Methods
Patients
In this study, we retrospectively analyzed patients who underwent renal cancer surgery in our hospital from June 2016 to May 2019. We evaluated the following demographic information of all patients: age, sex, body mass index, smoking, drinking, hypertension, and diabetes mellitus status. Additionally, the preoperative data of a routine blood test (neutrophilic granulocytes, platelets, and lymphocytes), blood coagulation (prothrombin time and activated partial thromboplastin time), and blood biochemical tests (levels of total albumin, albumin, blood uric acid, blood glucose, alanine aminotransferase [ALT], aspartate aminotransferase (AST), and alkaline phosphatase [ALP], and the eGFR) were simultaneously collected. Surgical information of the type of nephrectomy (RN or PN), surgical time, tumor position, and the approach (laparoscopy, open, or robotic) was also collected. The end point was whether AKI occurred after the operation. According to the RIFLE (Risk, Injury, Failure, Loss, and End-stage renal disease) standard, 8 the results of this study are expressed in terms of the occurrence of AKI, which was defined as a decrease of >25% in the eGFR from baseline preoperatively to discharge.9–11 The most recent eGFR before nephrectomy was selected as the baseline eGFR. The reporting of this study conforms to the STROBE statement. 12 This was a retrospective study, and no intervention was performed on the patients. Therefore, approval of an ethics committee was not required.
Statistical analysis
Continuous variables are shown as mean ± standard deviation, while descriptive statistics of classified variables are reported as frequency and proportion. Continuous variables and classified variables were compared by the Student’s t-test, χ2 test, or Fisher’s exact test.
The least absolute shrinkage and selection operator (Lasso) method was used for selection from the original data set, which resulted in a set of predictors that were more relevant and easier to explain. This method is suitable for regression of high-dimensional data. 13
Multiple logistic regression analysis was used to establish the prediction model,14,15 draw the calibration curve, and evaluate calibration of the postoperative AKI prediction model. 16 We measured Harrell’s C index to quantify the resolution performance of the predictive nomogram. The nomogram was bootstrapped to calculate the relative corrected C index, 17 quantify the net benefit under different threshold probabilities in the verification data set, and analyze the decision curve to determine the clinical practicability of the nomogram. 18 Statistical analysis was performed using R software (Version 4.0.2; https: //www.R-project.org).
Results
A total of 559 patients were included in this study. The mean age of the patients was 50.97±13.06 years, with 309 men and 250 women. According to the RIFLE diagnostic criteria, all patients were divided into the non-AKI and AKI groups. Postoperative AKI developed in 186 (33.27%) patients and it did not occur in 373 patients. All data of the patients, including clinical characteristics in the two groups, are shown in Table 1.
Clinical characteristics of the non-AKI and AKI groups.
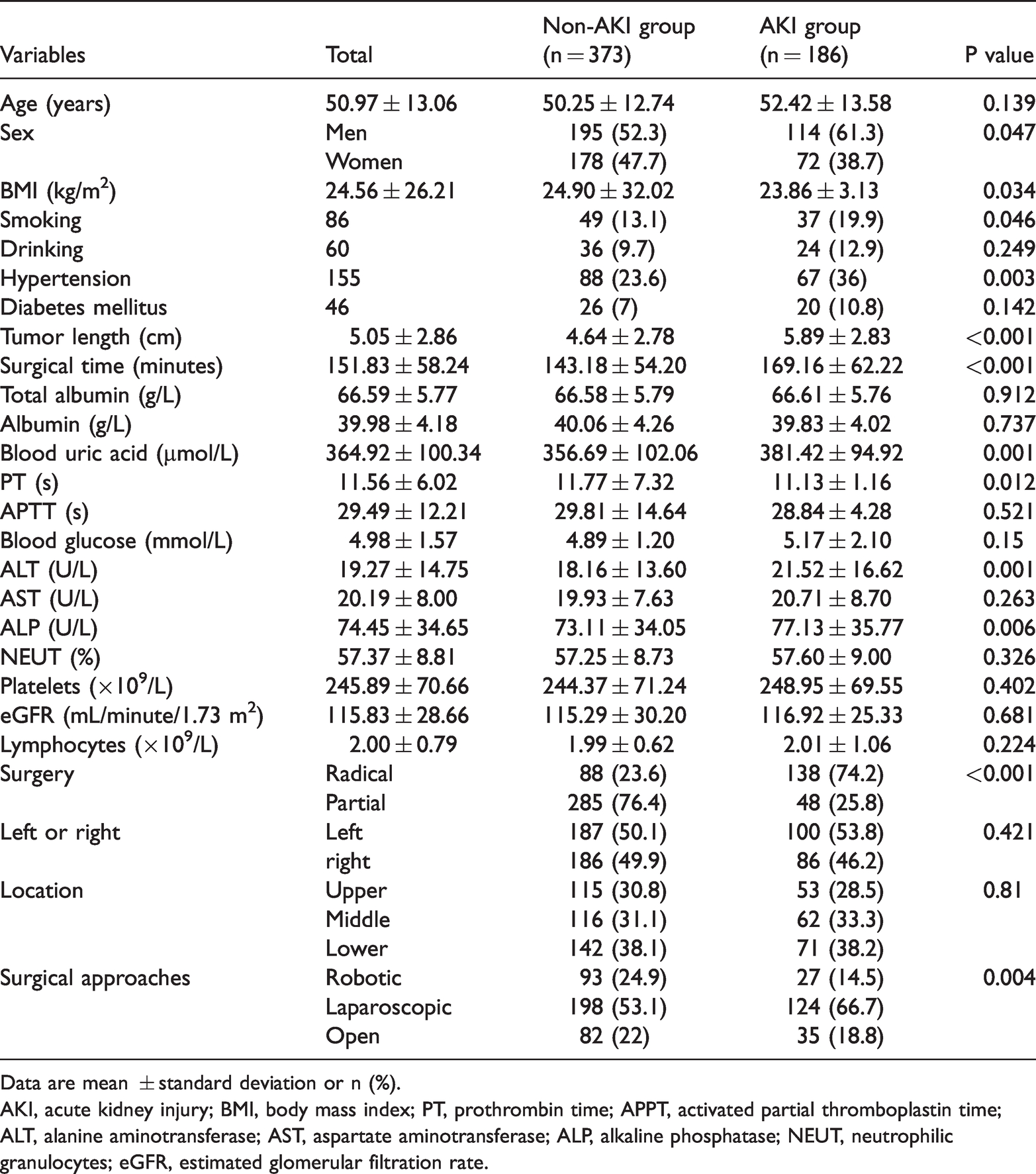
Data are mean ± standard deviation or n (%).
AKI, acute kidney injury; BMI, body mass index; PT, prothrombin time; APPT, activated partial thromboplastin time; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; NEUT, neutrophilic granulocytes; eGFR, estimated glomerular filtration rate.
Twenty-six variables were reduced to eight potential predictive factors on the basis of the 559 patients in the cohort (Figure 1). These variables were smoking, hypertension, surgical time, blood glucose, blood uric acid, ALT, eGFR, and radical nephrectomy. The results of logistic regression analysis are shown in Table 2. The surgical time, blood uric acid levels, the eGFR, and RN were significant for predicting postoperative AKI (all P < 0.05). A model that incorporated the above-mentioned independent predictive factors was developed and presented as a nomogram (Figure 2). The calibration curve of the nomogram for predicting the risk of postoperative AKI in the patients showed good agreement in this cohort (Figure 3). The C index for the predictive nomogram was 0.825 (95% confidence interval: 0.789–0.860) for the cohort and 0.814 through bootstrapping validation, which suggested good discrimination of the model. In the nonadherence risk nomogram, the apparent performance had a good predictive capability.

Selection of clinical characteristics using the least absolute shrinkage and selection operator (Lasso) binary logistic regression model. The tuning parameter (λ) in the Lasso model was selected by minimum criteria using five-fold cross-validation. A binomial deviance curve was plotted versus log(λ). Vertical lines were drawn at the optimal value with the minimum criteria and 1 standard error of the minimum criteria (1-SE criteria).
Predictive factors for postoperative acute kidney injury
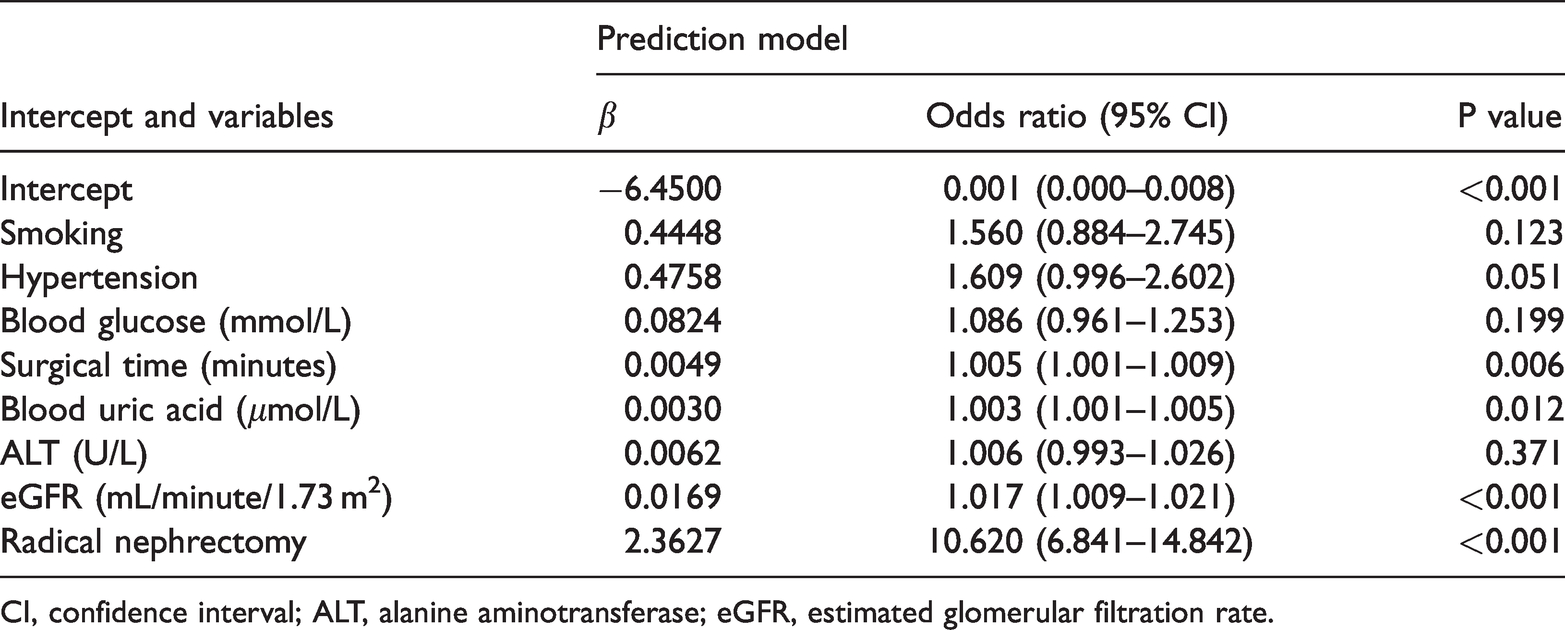
CI, confidence interval; ALT, alanine aminotransferase; eGFR, estimated glomerular filtration rate.

Developed postoperative AKI nomogram. The nomogram was developed for the cohort using smoking, hypertension, surgical time, blood glucose levels, blood uric acid levels, ALT levels, the eGFR, and radical nephrectomy.

Calibration curves of the nomogram for predicting the risk of postoperative AKI in the cohort. The calibration curves describe the calibration of the model based on the consistency between the predicted risk of postoperative AKI and the observed postoperative AKI results. The diagonal dashed line indicates perfect prediction of the ideal model. The black solid line indicates the performance of the nomogram; being closer to the diagonal dashed line indicates that the model has better prediction ability
Decision curve analysis for the postoperative AKI nomogram is shown in Figure 4. The decision curve showed that a threshold probability of >4% and <79% for patients and doctors, respectively, enabled better predictive ability of this nomogram for the risk of postoperative AKI. Using the nomogram in the current study to predict postoperative AKI was more beneficial than intervention with all regimens or non-intervention regimens of patients.

Decision curve analysis for the postoperative AKI nomogram. The blue line indicates the predictive nomogram for the risk of postoperative AKI. The thin solid line shows the assumption that AKI occurs in all patients after surgery. The thick black line shows the assumption that no patient has postoperative AKI. The decision curve shows that the threshold probabilities of patients and doctors are >4% and <79%, respectively.
Discussion
In recent years, the incidence of renal solid masses has shown an upward trend, and regardless of whether there is a benign or malignant mass, the main treatment is nephrectomy. However, postoperative renal dysfunction is still a continuing concern for health care providers. 19 This study focused on whether AKI will occur after nephrectomy and we established a prediction model to examine which factors may promote the occurrence and development of AKI.
Many studies have been conducted to investigate which factors can accurately predict postoperative AKI, but the results are not consistent.20,21 In this study, we established a prognostic model that included comprehensive preoperative predictors to determine the risk of AKI after nephrectomy to help urologists make timely clinical interventions before surgery. The incidence of postoperative AKI in this study was 33.27%, which is relatively high compared with other studies. 22 In the analysis of risk factors, smoking, hypertension, the surgical time, blood glucose, blood uric acid, ALT, the eGFR, and radical nephrectomy were related to the risk of postoperative AKI. Logistic regression analysis showed that the probability of RN and postoperative AKI was more than two times higher than that of PN and was an independent risk factor for AKI. This result is consistent with studies on the relationship between RN and an increased risk of chronic kidney disease, cardiovascular events, and even death, while better preservation of renal function after PN is thought to improve the prognosis after nephrectomy.23,24 As a result, urologists may be more likely to choose PN for patients with smaller renal lumps. RN can be used in patients with a low risk of postoperative AKI.
This study comprehensively analyzed the relationship between preoperative laboratory values and postoperative AKI. Regression analysis showed that preoperative blood glucose levels, ALT levels, the eGFR, and blood uric acid levels were factors that could predict the occurrence of postoperative AKI. We found that elevated preoperative blood glucose levels were associated with postoperative AKI. Before surgery, doctors should better control blood glucose levels, especially for patients with diabetes. An increase in ALT levels is also associated with AKI. ALT is mainly present in hepatocytes after an operation. When a liver lesion occurs, it leads to an increase in ALT levels. Therefore, if a patient has liver function damage before the operation, it should be treated in time, and the ALT value should be reduced to a normal value before a kidney operation. 25 In our study, hypertension appeared to be a risk factor for postoperative AKI, which is consistent with previous studies.3,26 Therefore, urologists should control blood pressure at a safe level, especially for patients with high blood pressure. Another important independent prognostic factor of postoperative AKI was the preoperative eGFR. Therefore, clinicians should evaluate renal function in patients before an operation.27,28
We also found that elevated blood uric acid levels were associated with postoperative AKI. Uric acid is thought to have proinflammatory effects.
Bertolo et al. 35 built a nomogram to predict postoperative loss of renal function in patients undergoing PN. He focused on chronic kidney disease upstaging as the outcome of interest owing to its clinical relevance and association with future morbidity and mortality. The logistic nomogram model had a C index of 0.76. Shum et al. 36 constructed and internally validated two nomograms to predict the eGFR at 1 year after PN, using only preoperative covariates as predictors. They showed that the percentage of patients with an eGFR <60 mL/minute/1.73 m2 increased from 25% before PN to 35% at 1 year after PN. Martini et al. 37 separately investigated the role of the occurrence of AKI in patients with normal preoperative renal function and that of the occurrence of AKI in chronic kidney disease. They found that the latter was associated with an increased risk of a significant reduction in the eGFR compared with the former.
Our research has several limitations. First, the study was conducted in a single institution, and multicenter research should be conducted in the future. Second, we could not analyze the relationship between other intraoperative factors and postoperative AKI, such as perioperative hydration and urine volume. Third, we only found that certain preoperative factors might lead to the specific mechanism of postoperative AKI, which is not clear. Therefore, further high-quality research is required in the future.
Conclusion
This study developed a new nomogram with a relatively good accuracy to help clinicians assess the risk of AKI after nephrectomy at the beginning of treatment. By performing estimation of individual risk, clinicians and patients may take more necessary intervention measures according to the patient’s condition.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (Nos. 81402116 and 82072817) and the Natural Science Foundation of Guangdong Province, China (No. 2019A1515010285).
Author contributions
Project development, data collection, data analysis, and manuscript writing: WYK and CJX; material preparation, data collection, and analyses: WYK, CJX, LC, CLW, and HB; draft preparation of the original manuscript: WYK and HB; financial support: CJX and HB; editing the manuscript: all authors; reading and approval of the final version of the manuscript: all authors.
Data availability statement
The data used to support the findings of this study are included within the article.
