Abstract
Low serum levels of vitamin D have been reported as a risk factor for breast cancer. This narrative review provides an update on the impact of vitamin D on hormone receptors, notably estrogen receptor subunits, and gives insights on possible therapeutic interventions to overcome breast cancer. In addition, evidence that supports the beneficial use of vitamin D as adjuvant treatment of breast cancer is summarized. Vitamin D deficiency is significantly widespread in patients with triple-negative tumors. Several studies have observed a possible modulatory effect of vitamin D or its analogues on the expression of different hormone receptors in breast cancer and increased sensitivity to tamoxifen. Vitamin D possesses anti-inflammatory and immunomodulatory effects in patients with breast cancer, and the mechanism of action of vitamin D in patients with breast cancer is discussed. In conclusion, vitamin D appears to have a beneficial role in the prevention and management of breast cancer, however, large-scale, randomized controlled trials are needed to confirm the effects of vitamin D in breast cancer prevention or treatment.
Keywords
Introduction
Breast cancer is the most common type of cancer diagnosed in women, and is the main cause of cancer death among women worldwide. In Jordan, breast cancer is the most prevalent cancer type, constituting about 22.4% of cancer-related mortality in females, followed by colorectal, and lung. 1 Breast cancer is a complex disorder with multiple subtypes, each one characterized by its unique morphology, histopathological and biological features, clinical progress and implications.2,3 The three major breast cancer tumor subtypes are classified according to the expression of estrogen receptors (ERα and ERβ), progesterone receptor (PR), or human epidermal growth factor receptor 2 (HER2; encoded by erb-b2 receptor tyrosine kinase 2 [ERBB2]).3–7
Several epidemiological studies have indicated the enhancing carcinogenic potential of estrogens, as confirmed by the higher incidence of breast cancer in women with prolonged exposure to estrogens and the significantly decreased incidence in women with ovarian functional abnormalities. ER is highly expressed in about two-thirds of breast tumors, with estrogen effects being mediated via two specific receptors, ERα and ERβ. 8 The effects of estrogen are complex, directly affecting growth, motility, and invasiveness of cancer cells, neo-angiogenesis and the immune response. The expression of ERα receptors exhibits good prognostic value, as ERα-positive cancers are more differentiated and less invasive.3,9 Diagnosis and the selection of suitable breast cancer therapy is greatly dependant on the degree of expression of the three aforementioned hormone receptors, the cancer type, and the stage of cancer progression. A course of chemotherapy and hormone therapy may be included in the eventual treatment regime, whilst considering the potential side effects.4,5,10 In several clinical studies, vitamin D has been observed to exhibit a protective effect against breast cancer, has increased the anticancer response and has been associated with improved clinical outcomes and cancer survival. Additionally, bone modifying agents, notably bisphosphonates and nuclear factor-κB ligand inhibitors, decrease the incidence of skeletal-related hazards and improve breast cancer outcomes in vitamin D deficient patients with bone metastasis.11–13
The aim of the current narrative review was to provide an update of current breast cancer therapies and illustrate the possible use of vitamin D as an adjuvant, particularly in advanced breast cancer cases with metastasis, and in cases of triple-negative breast cancer (TNBC).
Vitamin D deficiency: a risk factor for breast cancer
Vitamin D deficiency is defined as a serum 25-hydroxy vitamin D level <20 ng/mL or 50 nmol/L, and whereas vitamin D deficiency is a characteristic feature in all patients with breast cancer, it is of uniquely higher prevalence in those with TNBC, the most aggressive form. 13 A relationship between plasma concentrations of vitamin D and breast cancer carcinogenesis has been established,14,15 and many epidemiological studies have investigated the inverse association between Vitamin D status and breast-cancer risk. The postmenopausal incidence of breast cancer is shown to be significantly decreased in patients with high levels of 25-hydroxy vitamin D, 16 with longer disease-free survival and reduced mortality.17,18 Vitamin D has been shown to be of great benefit in preventing breast cancer, however its role in treatment is not evidenced.19,20 Observational studies have concluded that deficiency in 25-hydroxy vitamin D is related to breast cancer, and an inverse relationship was found between intake of supplemental vitamin D and occurrence of breast cancer.21–24 Conversely, these associations were not supported in another study. 25
Activities of calcitriol in breast cancer
Vitamin D has diverse biological actions in relation to carcinogenesis. Calcitriol (1, 25-dihydroxyvitamin D3 or 1, 25-[OH] 2D3), is the active form of vitamin D3 and is considered to be a milestone agent for calcium homeostasis regulation. It possesses antiproliferative activities on the majority of body systems and has the ability to stimulate differentiation of hematopoietic cells. The aforementioned effects are mediated by a member of the nuclear receptor superfamily of transcription factors, the vitamin D receptor (VDR), that is considered to regulate cell proliferation, apoptosis and metastasis. 26 In addition, malignant cells show decreased production of intracellular calcitriol compared with normal cells, as they lack 1α-hydroxylase (the activating enzyme) and increase calcitriol destruction with consequently higher resistance to the beneficial antitumor activity of vitamin D. 27 In addition, immunohistochemical analysis of breast cancer cells has demonstrated down-regulation of VDR. 28 In their study, Hemida et al., 29 reported an inverse correlation between serum vitamin D levels and tissue VDR levels and gene expression of ERα subunits, and concluded that breast cancer risk increased significantly with serum 25 (OH) D levels ≤30 nmol/L, VDR tissue levels >5 ng/mL and ERα gene expression more than 17.7 copies. In addition, the beneficial effects of calcitriol in TNBC have been explained by immunomodulatory activities in the form of inducing the synthesis of interleukin-1β and tumor necrosis factor-α receptors. 30
Many laboratory studies have demonstrated the anticancer effects of vitamin D metabolites on three critical phases in the development of breast tumors: differentiation, apoptosis, and angiogenesis. 27 It is possible that the association between serum 25-(OH)-D and survival depends on the activities of vitamin D metabolites, notably in maintaining differentiation, promoting apoptosis, and inhibiting angiogenesis. 31 In studies of mice injected with cultured human breast cancer cells, oral calcitriol inhibited proliferation of cancer cells. 32 However, randomized controlled trials [RCTs] have shown less benefit in supplementing vitamin D in cases of breast cancer.33,34
Calcitriol suppresses cyclooxygenase-2 expression and thereby reduces inflammatory prostaglandin levels. Several in vitro and in vivo studies noted that calcitriol decreases aromatase expression in breast tumor cells through the inhibition of transcription and indirectly due to reduction of prostaglandin levels, which normally stimulate aromatase transcription. Additionally, calcitriol downregulates the expression of ERα. 35 A review by Negri et al., 2020, 36 reported that vitamin D has the ability to enhance the efficacy of conventional therapy beside its contribution in combating drug resistance, and has many activities at various molecular levels, i.e. it exhibits a regulatory effect on cancer stem cell growth, epithelial–mesenchymal transition, and short non-coding microRNA gene expression. The different activities of calcitriol that may explain its potential therapeutic benefits in breast cancer are illustrated in Figure 1.
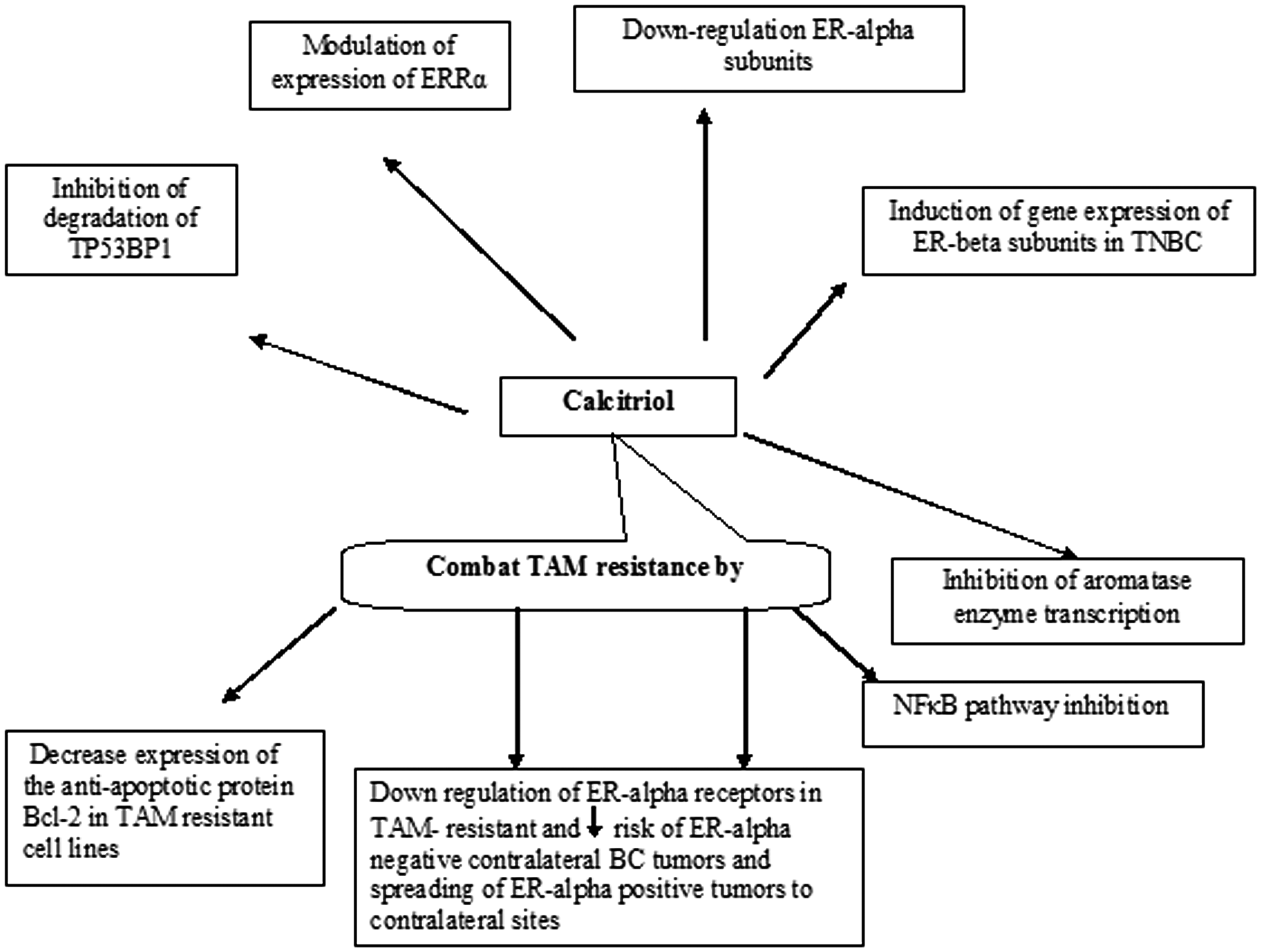
Illustration of the potential activities of calcitriol in breast cancer. Vitamin D downregulates ERα expression via inhibition of NF-κB, and increases sensitivity to tamoxifen through induction of functional ERα in ER-negative cancer cells. Its combination with tamoxifen may be effective in tamoxifen-resistant tumors. BC, breast cancer; ERRα, estrogen-related receptor α; ER, estrogen receptor; NFκB, nuclear factor- kappa B; TAM, tamoxifen; TNBC; triple negative breast cancer; TP53BP1, tumor protein P53 binding protein 1.
Downregulation of ERα by calcitriol
The work of Beatson, 1896, was the first to reveal the role estrogen in breast carcinogenesis and cancer progression, as evidenced by the antitumor effect of ovariectomy in a breast cancer patient. 37 ERα mediates the proliferative response and growth inducing activities of estrogens,37,38 and ERα-positive breast cancers have been shown to respond well to hormone therapies. 39 The antiestrogen tamoxifen is the most common, and considered to be the most effective, treatment in both pre- and postmenopausal patients with ER-positive cancers, with its long-term use shown to increase patient survival and reduce cancer recurrence.40,41 Unfortunately, a significant percentage of patients with ERα-positive tumors lose ER expression in recurrent tumors, and metastatic tumors also develop resistance to tamoxifen and lose ERα expression. 42 Lack of ER expression may be secondary to increased mitogen-activated protein kinase (MAPK) signalling activity, or increased expression of specific microRNAs.43,44 Inhibition of MAPK activity and knockdown of specific microRNAs has been found to restore functional ERα in ER-negative breast cancer cells. 45
The most active metabolite of vitamin D is calcitriol, which mediates significant antiproliferative activities in breast cancer cells, via the VDR, by arresting growth, cell differentiation, migration, invasion and apoptosis. 46 In addition, epidemiological research has revealed that low levels of the calcitriol precursor calcidiol is associated with a higher risk of breast cancer. 47 Decreased calcitriol carries the risk of enhanced cancer progression and underexpression of ER, thus, increasing the potential risk of ER-negative and triple-negative breast tumors.22,48 In addition, patients with VDR-positive breast cancer had significantly longer disease-free survival than those with VDR-negative tumors, 49 and VDRs are reported to be highly expressed in breast cancer with a low risk of death and good prognosis.50,51 In a case–control study, it was reported that VDR-negative individuals were more prone to develop ER- and PR-negative breast cancers. 22 Calcitriol has also been shown to exert a significant antiproliferative effect on cells taken from breast cancer biopsies or cell lines.52–54 Some epidemiologic studies have suggested that vitamin D intake reduces the risk of ER-positive breast cancer,55–58 while its deficiency is associated with poor outcomes in patients with luminal-type breast cancer. 59 Notably, ER-positive cells tend to express higher levels of VDR than ER-negative cells. 60 Therefore, calcitriol would be expected to mediate actions that were particularly effective in ER-positive breast cancer. Calcitriol has been shown to downregulate ERα expression in breast cancer cells. 43 Krishnan et al., 35 postulated that calcitriol decreases the synthesis of estrogens by breast cancer cells and the surrounding breast adipose/stromal tissue, and decreases the levels of ERα in breast cancer cells. Combinations of vitamin D analogs, such as calcitriol with estrogen receptor antagonists or tamoxifen have been shown to inhibit the growth of breast cancer cells.61–64
In addition to the well-known ERα, a more recently discovered nuclear receptor, estrogen-related receptor α (ERRα), has been shown to interfere with the VDR pathway, however its effect on the cytotoxic activity of vitamin D in breast cancer remains vague. ERRα may enhance the disruption of VDR genomic action and consequently worsen breast cancer prognosis.65,66
Effect of calcitriol on tamoxifen-sensitive and resistant breast cancer
Teft et al., 2013, studied the influence of sunlight exposure and vitamin D status, and found that endoxifen, the active metabolite of tamoxifen, decreased during winter months concomitantly with lower vitamin D levels. 67 Serum vitamin D levels have been observed to increase following therapy with tamoxifen, however the impact of increased vitamin D levels on efficacy of tamoxifen remains undetermined. 68 In addition, calcitriol has been reported to effectively decrease the growth of both tamoxifen-sensitive and resistant breast cancer cells through NF-κB pathway inhibition. 69
In their 2001 study, Larsen et al., 70 reported that estradiol, unlike EB1089 (a vitamin D analogue), induced expression of the antiapoptotic protein B-cell lymphoma 2 (Bcl-2) in two well-characterized antiestrogen resistant cell lines, MCF-7/TAMR-1 and MCF-7/182R-6, and abolished or reduced the growth inhibitory effect of EB1089 on MCF-7 cell lines, however, EB1089 was found to have a partial effect on MCF-7/TAMR-1 with no effect on MCF-7/182R-6 cells. EB1089 was also found to downregulate ERα expression in tamoxifen- resistant cell lines. 70 Another study showed that calcitriol, combined with the janus kinase (JAK)1 and JAK2 inhibitor ruxolitinib, exhibited a synergistic suppressive effect on ER and HER2-positive MCF7-HER18 breast cancer cells. Calcitriol with ruxolitinib was shown to decrease the levels of JAK2, phosphorylated JAK2, c-Myc proto-oncogene protein, cyclin D1, apoptosis regulator Bcl-2 and Bcl-2-like protein 1. Additionally, they increased the protein levels of caspase 3 and Bcl2-associated agonist of cell death. 71 Calcitriol combined with ruxolitinib may be a therapeutic strategy for tamoxifen-resistant breast cancer, and may be added to trastuzumab for HER-2-positive cancers. Similarly, another study confirmed the latter results and reported that JAK2 may be a new therapeutic target for tamoxifen-resistant breast cancer. 72 JAK2 was described to selectively phosphorylate signal transducer and activator of transcription (STAT)-3, with the JAK2-STAT3 signaling pathway considered to be principal in regulating cancer progression and metastasis. The study also revealed that basal phosphorylation of STAT3 was significantly greater in tamoxifen-resistant MCF-7 cells compared with control MCF-7 cells, and ruxolitinib was observed to significantly attenuate STAT3 phosphorylation, and consequently, the proliferation of tamoxifen-resistant MCF-7 cells. 72
Calcitriol and aromatase inhibitors
Concurrent administration of calcitriol with aromatase inhibitors (AIs) has been shown to enhance the growth inhibitory effects in MCF-7 cells in vitro. 35 In the study, calcitriol exhibited an inhibitory effect on the expression of aromatase enzyme by direct repressor action on transcription in human breast cancer cells, the adjacent mammary adipose tissue, and in vitro cultured preadipocytes. 35 Additionally, Lundqvist et al., 69 found that combining a low dose of EB1089, a vitamin D analogue, with low doses of AIs effectively inhibited aromatase-dependent growth of breast cancer cells. Similarly, Swami and colleagues hypothesized that combining calcitriol with AI may be beneficial in treating breast cancer. 73 Various studies have attributed the inhibitory effect of calcitriol on aromatase enzyme expression to decreasing the production of prostaglandins by inhibiting the enzyme cycloxygenase-2.74,75
Vitamin D signaling in triple-negative breast cancer
As mentioned, TNBC represents about one-fifth of all breast cancer cases, 76 and has limited therapeutic options, with more aggressive progress, higher recurrence rate, and a worse prognosis than other types of breast cancer. Current TNBC therapy comprises standard chemotherapy, with or without radiation therapy, and with no available prophylactic agents. 77 Average vitamin D levels are reported to be deficient in TNBC cases with poor prognosis compared with other cases.78,79
Promising studies have focused on potential novel VDR-targeted therapies for TNBC. A study by Thakkar et al., 80 demonstrated that the majority of TNBCs express VDR, and VDR agonists may be potential agents for concomitant use with standard chemotherapy, as they have shown antiproliferative effects in various TNBC cell lines via increased apoptosis and cycle arrest. Another study reported that the calcitriol analog MART-10, combined with calcitriol, significantly attenuated metastasis in some TNBC cell lines, and MART-10 was of higher potency than calcitriol. 81 The suppressive effect of calcitriol, or vitamin D analogues, has also been shown on SUM-159PT and WT145 TNBC cell lines.82,83
In a collaborative study, vitamin D analogues EM1 and UVB1 were observed to significantly decrease the viability of HER2-positive and TNBC-patient-derived xenografts (PDXs). Additionally, UVB1 exhibited antiproliferative activity in an in vitro model of acquired trastuzumab-emtansine resistance, and also had an effect on VDR expression in PDXs. 84 A review by Blasiak et al., 2020, 85 reported the possible protective molecular mechanisms of vitamin D in TNBC, particularly in cases with mutations in the DNA repair-associated breast cancer type 1 susceptibility (BRCA1) gene, including its potential inhibitory effect on degradation of tumor protein P53 binding protein 1 (TP53BP1) mediated by cathepsin L. In addition, 1,25(OH)2D may interact with proteins of the growth arrest and DNA damage-inducible 45 (GADD45) family.
Conversely, vitamin D has been observed to be ineffective in inhibiting the proliferation of MDA-MB-157, MDA-MB-231 and MDA-MB-468 TNBC cell lines. 86 Richards and his colleagues observed that the resistance of TNBC cell lines to vitamin D may be due to a lack of p53 gene, or non-functioning p53, so that vitamin D may be anti-apoptotic rather than apoptotic, 86 as evidenced by the study of Stambolsky et al., 2010. 87 Similarly, Hirshfield and Ganesan, 2014, 88 reported that TNBC cell lines may possess mutant p53, and thus resist vitamin D.
Novel therapeutic interventions are needed to overcome the major challenge of lacking drug targets for treating ER–/HER2– TNBC. Thakkar et al., 2016, 80 reported that patients with TNBC may express VDR and/or androgen receptor, and hypothesized that cell proliferation in TNBC cell lines may be inhibited by androgen receptor and VDR agonists via cell cycle arrest, apoptosis and inhibition of cancer stem cells. In another study, calcitriol, through an effect on the VDR, was found to induce the expression of functional ERα in ER-negative breast cancer cells, and calcitriol-induced ERα restored the response to antiestrogens by inhibiting cell proliferation. 89 Zheng et al., 90 suggested that calcitriol combined with paclitaxel (PTX) may be a promising therapy for TNBC, and explained their conclusion by the finding that calcitriol downregulated matrix metalloproteinase-9 and Bcl-2 levels, upregulated E-cadherin levels, and counteracted the elevation of C-C motif chemokine ligand 2 (CCL2) and Ly6C+ monocyte levels induced by paclitaxel. Treatment of TNBC cells in vitro with ERβ agonists was shown to make breast cancer cells less invasive, contrary to knockdown of the ERβ gene that increased the invasiveness of cancer cells. 91 Calcitriol combined with ERβ agonists may have potential as an effective therapeutic strategy. Key research findings regarding the modulation of ERs in breast cancer cells by vitamin D or its analogues are summarised in Table 1.19,23,25,26,29,30,64,68,69,71,72,79–82,85,86,89–91
Summary of important research findings regarding the modulation of estrogen receptors in breast cancer cells by vitamin D or its anaolgs.

AIs, aromatase inhibitors; AR, androgen receptor; BCL-2, B-cell lymphoma-2; CYP24A1, cytochrome P-450 24A1; ER, estrogen receptor; GADD45, growth arrest and DNA damage-inducible 45 family; HER2, human epidermal growth factor receptor 2; IL, interleukin; JAK, Janus kinase; NF-κB, nuclear factor-κB; RCTs, randomized controlled trials; TNBC, triple negative breast cancer; TNF, tumor necrosis factor; VDR, vitamin D receptor.
Conclusion
The present review has provided evidence that the conventional anti-estrogen tamoxifen may enhance the spread of Erα-positive tumors to contralateral sites, in addition to increasing the risk of ERα-negative contralateral tumors. As vitamin D downregulates ERα expression via inhibition of NF-κB, it may increase the sensitivity to tamoxifen through induction of functional ERα in ER-negative cancer cells. Vitamin D combined with tamoxifen may be effective in tamoxifen-resistant tumors, and its concurrent use with aromatase inhibitors may be another suitable therapeutic option. Vitamin D analogs that induce ERβ subunits in addition to androgen agonists may be future promising therapeutic interventions to overcome TNBC. Vitamin D appears to possess anti-inflammatory and immunoregulatory effects in patients with breast cancer. Large-scale randomized controlled trials are needed to confirm whether vitamin D may prevent or treat breast cancer.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221113800 - Supplemental material for Vitamin D: an essential adjuvant therapeutic agent in breast cancer
Supplemental material, sj-pdf-1-imr-10.1177_03000605221113800 for Vitamin D: an essential adjuvant therapeutic agent in breast cancer by Romany H Thabet, Adel A Gomaa, Laila M Matalqah and Erin M Shalaby in Journal of International Medical Research
Footnotes
Acknowledgements
Great thanks to breast oncologists in King Hussein Cancer Centre, Jordan for their valuable help in answering our questions about current medical management of patients with advanced breast cancer, especially those with metastasis, and whether or not these patients take calcitriol as an adjuvant.
Author contributions
Romany Thabet, the corresponding author, was the major contributor in designing and coordinating the duties of each co-author, and edited the final version of the manuscript. Adel Gomaa, Laila Matalqah and Erin Shalaby assisted in collecting data from the published literature and drafting the manuscript.
Declaration of conflicting interest
The Authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
