Abstract
Objective
This study aimed to explore the effect of sphingosine-1-phosphate on vascular homeostasis in the myocardium of diabetic mice.
Methods
Bioinformatics analyses were performed to analyze the targets and function of sphingosine-1-phosphate in diabetes. C57BL/6 mice were used to establish diabetic models and treated with sphingosine-1-phosphate. Pathological examination was used to evaluate the damage to the myocardium. Immunohistochemistry and western blot analyses were used to examine the expression of molecules involved in vascular function and sphingosine-1-phosphate receptors. Endothelial nitric oxide synthase level was detected via enzyme-linked immunosorbent assay.
Results
Multiple Gene Ontology entries of sphingosine-1-phosphate were associated with vascular homeostasis. Blood glucose level, food intake, and water intake increased and body weight decreased in diabetic mice, whereas these changes were relieved in the sphingosine-1-phosphate group. Sphingosine-1-phosphate alleviated the myocardial injury and restored the expression levels of vascular endothelial-cadherin, vascular endothelial growth factor, and CD31 in the myocardium of diabetic mice to some extent. The serum level of endothelial nitric oxide synthase increased in the sphingosine-1-phosphate group. Additionally, sphingosine-1-phosphate was demonstrated to act on its receptors S1PR1, S1PR2, and S1PR3, which were downregulated in the myocardium of diabetic mice and significantly upregulated after sphingosine-1-phosphate treatment.
Conclusion
Sphingosine-1-phosphate may restore myocardial vascular homeostasis by regulating the balance of sphingosine-1-phosphate receptors in diabetic mice, suggesting sphingosine-1-phosphate/sphingosine-1-phosphate receptors as potential therapeutic targets for diabetic myocardial injury.
Keywords
Introduction
Diabetes is a complex disease caused by interactions between genes and the environment and is becoming one of the most common and fastest growing diseases worldwide.1,2 Cardiovascular complication is the leading cause of mortality in individuals with diabetes and dramatically worsens the quality of life.3,4 Diabetes usually results in the imbalance of myocardial vascular homeostasis, which is one of the main pathogeneses of diabetic cardiomyopathy. 5 A growing number of patients with diabetes have led to increasing social and economic burden on families and healthcare systems. 6
Sphingosine-1-phosphate (S1P) is a lysophospholipid with various biological activities, such as anti-inflammation and antioxidative stress.7,8 Previous studies have reported that S1P is an important regulator of endothelial and vascular functions and is crucial for cardiovascular development and regulation of vascular permeability.9,10 Plasma S1P is believed to contribute to the occurrence and development of myocardial ischemia in different clinical and experimental models and may serve as a new predictive biomarker for cardiac and metabolic disorders. 11 Brinck et al. 12 found that glycation can reduce the content of S1P in high-density lipoprotein, leading to increased cardiomyocyte death. It has been reported that plasma S1P level is inversely associated with diabetic patients’ mortality, which usually results from diabetes mellitus-related cardiomyopathy. 13 These findings indicate that S1P plays a protective role in diabetic cardiomyopathy. Previous studies have shown that S1P can regulate angiogenesis, inflammation, permeability, and reactive oxygen species production in endothelial cells 14 as well as balance lipid homeostasis in diabetic patients with cardiovascular disease. 15 It has been confirmed that the majority of known S1P effects are based on three specific G-protein-coupled receptors, termed as S1P receptor (S1PR)1–S1PR3, which are the mainly expressed receptors of S1P in the cardiovascular system. Moreover, S1P plays vital roles in regulating cardiovascular functions by binding to and activating S1PR1–S1PR3.16–18 However, the effect of S1P in diabetic cardiomyopathy has not been fully elucidated.
Based on the above data, we hypothesized that S1P may alleviate myocardial vascular homeostasis imbalance caused by diabetes through its receptors. Therefore, based on the diabetic mouse model, we investigated the effect of S1P on the myocardium of diabetic mice via intraperitoneal injection of S1P and detected the expression of S1PRs, providing a reference for its clinical application.
Materials and methods
Main reagents
Streptozotocin (STZ) was purchased from Solarbio (Beijing, China). High-sugar, high-fat feed (DB0001) and endothelial nitric oxide synthase (eNOS) detection kit (20230310) were purchased from Bioswamp (Wuhan, China). S1P (S9666-1MG) was purchased from Sigma-Aldrich (Darmstadt, Germany). S1PR1 and S1PR2 antibodies were purchased from Invitrogen (Grand Island, US). Antibodies against S1PR3, vascular endothelial (VE)-cadherin, CD31, vascular endothelial growth factor (VEGF), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were purchased from Bioswamp (Wuhan, China).
Diabetes model construction
Thirty SPF-grade C57BL/6 male mice (age, 6–8 weeks; weight, 22–25 g) used in the present study were purchased from Hunan SJA Laboratory Animal Co., Ltd. (Changsha, China). We made efforts to minimize the number of animals utilized and decrease their suffering. Mice were housed in SPF-level animal houses, with a 12-h light/dark cycle, temperature maintained at 25°C, and relative humidity maintained at 50%–60%, throughout the study period. After adaptive feeding for 1 week, during which all mice were allowed free access to normal chow and water, the mice were randomly divided into the control group (n = 6) and model group (n = 24) via random number table. The mice in the model group were injected intraperitoneally with a low dose of STZ (70 mg/kg, 0.1 moL/L citrate buffer) once a day for 2 consecutive days and then continued to be fed high-fat diet for 4 weeks. 19 After 4 weeks, blood was collected from the tail vein, and the diabetes model was considered successful when the blood glucose concentration was >11.1 mmol/L. The mice in the control group were intraperitoneally injected with an equal volume of sterile citrate buffer (0.1 mol/L), once a day for 2 consecutive days and then continued with standard feeding. All animal experimental protocols complied with the Guide for the Care and Use of Laboratory Animals, 20 and the Animal Ethics Committee of Wuhan Myhalic Biotechnology Co., Ltd. provided full approval for this research (HLK-20221014-001). This study conforms to the Animal Research: Reporting of In Vivo Experiments (ARRIVE) 2.0 guidelines. 21
Bioinformatics analysis
We screened various keywords in Gene Expression Omnibus (GEO), Sequence Read Archive (SRA), The Cancer Genome Atlas (TCGA), and other public databases to determine the targets of S1P for diabetes. The data obtained from GEO (GSE215979) were preprocessed as well as normalized to obtain the corresponding expression matrix. Ctrl1–3 in the expression matrix referred to control mice, whereas DM1–3 referred to diabetic mice. The data from the model and control groups were analyzed for differences and screened for differential genes based on the criteria of |log2FoldChange|>1 and p-value ≤0.05. The differential genes were then subjected to Gene Ontology (GO) enrichment analysis.
Grouping and general observation of mice
Twelve mice with successful modeling were randomly selected and further divided into the diabetic model group (n = 6) and S1P treatment group (n = 6) via random number table. Regarding S1P preparation, 1 mg of S1P was dissolved in 1 mL of methanol, after which the mixture was stored at −20°C in the dark and diluted with phosphate-buffered saline (PBS) buffer as the working solution. The mice in the S1P group were intraperitoneally injected with 20 μg/kg/d S1P once a day for 3 consecutive weeks, 22 whereas those in the diabetic model group were intraperitoneally injected with an equal volume of saline. The flowchart of animal modeling and S1P administration treatment is shown in Figure 1. During intervention, the body weight, feed intake, water intake, and fasting blood glucose levels of mice in each group were detected every week.

Flowchart of animal modeling and S1P administration treatment. S1P: sphingosine-1-phosphate.
Sampling methods
At the end of the experiment, all mice were euthanized and sacrificed via exsanguination through the carotid artery under anesthesia through an intraperitoneal injection of 1% pentobarbital sodium (40 mg/kg), which induced unconsciousness in the shortest possible time to reduce pain and fear as much as possible. The mice were considered dead half an hour after their heartbeats stopped. Subsequently, the heart tissue and approximately 1 mL of blood from the carotid artery of each mouse were collected for the following analysis. The whole heart was collected, including the aorta, using ophthalmic scissors to cut along the backbone. Then, the root of the aorta was removed, and the heart was separated, weighed, and washed with ice-cold saline. The heart weight to body weight ratio (HW/BW) was calculated. A part of the heart was stored in liquid nitrogen at −80°C for western blot analysis. The other part of the heart was maintained in 10% formalin solution for histochemical analysis.
Hematoxylin and eosin (HE) staining
The hearts were fixed with 10% formalin solution for 24 h at room temperature. Then, the fixed myocardial tissues were dehydrated, embedded, and sectioned into slices (4.5 μm) according to routine procedures. Subsequently, the sections were dewaxed, stained with HE, washed, dehydrated with graded ethanol, sealed with neutral gum, and visualized using an optical microscope.
Immunohistochemistry
After fixation, dehydration, clearing, wax dipping, embedding, sectioning, dewaxing, and hydration, the myocardial samples were placed in a citric acid antigen retrieval solution. Antigen retrieval was conducted in a microwave oven, with medium heat for 8 min until boiling, ceasing fire for 8 min, and medium low heat for 7 min. Following cooling naturally, each sample was washed with PBS three times, 5 min each time, incubated in 3% hydrogen peroxide solution at room temperature for 25 min in the dark to block endogenous peroxidase, and blocked with 3% bovine serum albumin at room temperature for 30 min. Subsequently, primary antibodies (CD31, CD11b, S1PR1, S1PR2, and S1PR3) were added to each sheet and incubated overnight at 4°C, followed by washing with PBS three times. Then, a secondary antibody (HRP-labeled) was added dropwise to cover the tissue, followed by incubation at room temperature for 50 min. In total, 100 μL of freshly prepared diaminobenzidine solution was added dropwise to each sheet for color development. The color development time was controlled under a microscope. After being washed and counterstained with HE, the sections were dehydrated, making them transparent, and sealed with gradient ethanol, xylene, and neutral gum in sequence. The stained sections were scanned with a digital slide scanner (3DHISTECH). The positive results were indicated by brownish yellow area and were quantitatively analyzed via ImageJ software. Then, the integrated option density (IOD) value, which was proportional to the total amount of target protein, of the brownish yellow area was determined. Subsequently, the IOD value was divided by the area of target protein distribution to obtain the average optical density (AOD) value. The differences in protein expression were compared by AOD value.
Western blot analysis
In total, 50 mg of mouse myocardial tissue was taken and lysed with radioimmunoprecipitation assay lysis buffer. A bicinchoninic acid protein assay kit was used to quantify the protein concentration. Then, the protein samples were denatured at 95°C for 10 min. A polyacrylamide gel electrophoresis rapid preparation kit was used to prepare the gel, load the samples, set the parameters of the electrophoresis instrument to 80 V, and perform constant voltage electrophoresis for 15–20 min. Then, the parameters of the electrophoresis instrument were adjusted to 120 V, and constant voltage electrophoresis was performed for 60 min. The proteins separated on the gel were transferred to poly (vinylidene fluoride) membranes (constant voltage of 110 V, 90 min). The membranes were soaked in 5% skimmed milk, placed on a horizontal shaker, and sealed at room temperature for 2 h. Subsequently, the membranes were incubated with primary antibodies (S1PR1 1:1000; S1PR2 1:1000; S1PR3 1:1000; VE-cadherin 1:1000; CD31 1:1000; VEGF 1:1000; and GAPDH 1:1000) overnight at 4°C on a shaker and were then incubated with a secondary antibody conjugated to horseradish peroxidase on a shaker at room temperature for 1 h. Following incubation with each antibody, the membranes were washed five times with Tris-buffered saline–Tween at room temperature. The ultrasensitive enhanced chemiluminescence luminescent liquid was used for color development. After the image was collected, the gray value of each band was analyzed using Image J and normalized to that of GAPDH, which was used as an internal reference. All groups were then normalized to their respective controls.
Enzyme-linked immunosorbent assay (ELISA)
An ELISA kit was used to detect the level of eNOS in the mouse serum. First, the standard substance was diluted to construct a standard curve. The standard wells, blank wells, and sample wells were set on the microplate, and standards and samples of different concentrations were added to the standard wells. Then, 10 μL of biotinylated antibody and 50 μL of enzyme-labeled reagent were added, and the plate was sealed and incubated for 30 min. After washing, a color developer was added in the dark for 10 min. Finally, a stop solution was added, and the absorbance (OD value) of each well was sequentially measured at a wavelength of 450 nm.
Statistical analysis
Statistical software SPSS 15.0 was used for data analysis and processing. All continuous data were expressed as mean ± standard error. One-way analysis of variance (ANOVA) or least significant difference was used to compare data that followed homogeneous variance and normal distribution, and the rank sum test was used otherwise. Two-way ANOVA was used to compare the effects of different treatments and time on body weight, food and water intake, and blood glucose levels. p-values of <0.05 were considered to indicate statistical significance.
Results
S1P plays a key role in the vascular homeostasis of diabetes
First, we investigated the targets of S1P and performed differential gene GO analysis of the action of S1P in diabetic mice via bioassay analysis. The data obtained from GEO were preprocessed as well as normalized to obtain the corresponding expression matrix. Pearson correlation coefficients between the samples were calculated and were all above 0.9, which represented a relatively good correlation (Figure 2(a)). Based on the principal component analysis of the normalized data, the model and control groups could be clearly divided into two categories, with better reproducibility of samples from the same group and more pronounced component differences (Figure 2(b)). The data from each group were then analyzed for differences; the top 50 differences are shown in Figure 2(c). The screening of differential genes was performed based on the criteria of |log2FoldChange|>1 and p-value ≤0.05, and 2071 differential genes were obtained (Figure 2(d)). Next, we analyzed the abovementioned 2071 genes for GO enrichment and found that multiple GO entries were related to vascular homeostasis, including regulation of angiogenesis, regulation of vasculature development, positive regulation of angiogenesis, and positive regulation of vasculature development (Figure 2(e)). These findings indicated that S1P plays a key role in the vascular homeostasis of diabetes.
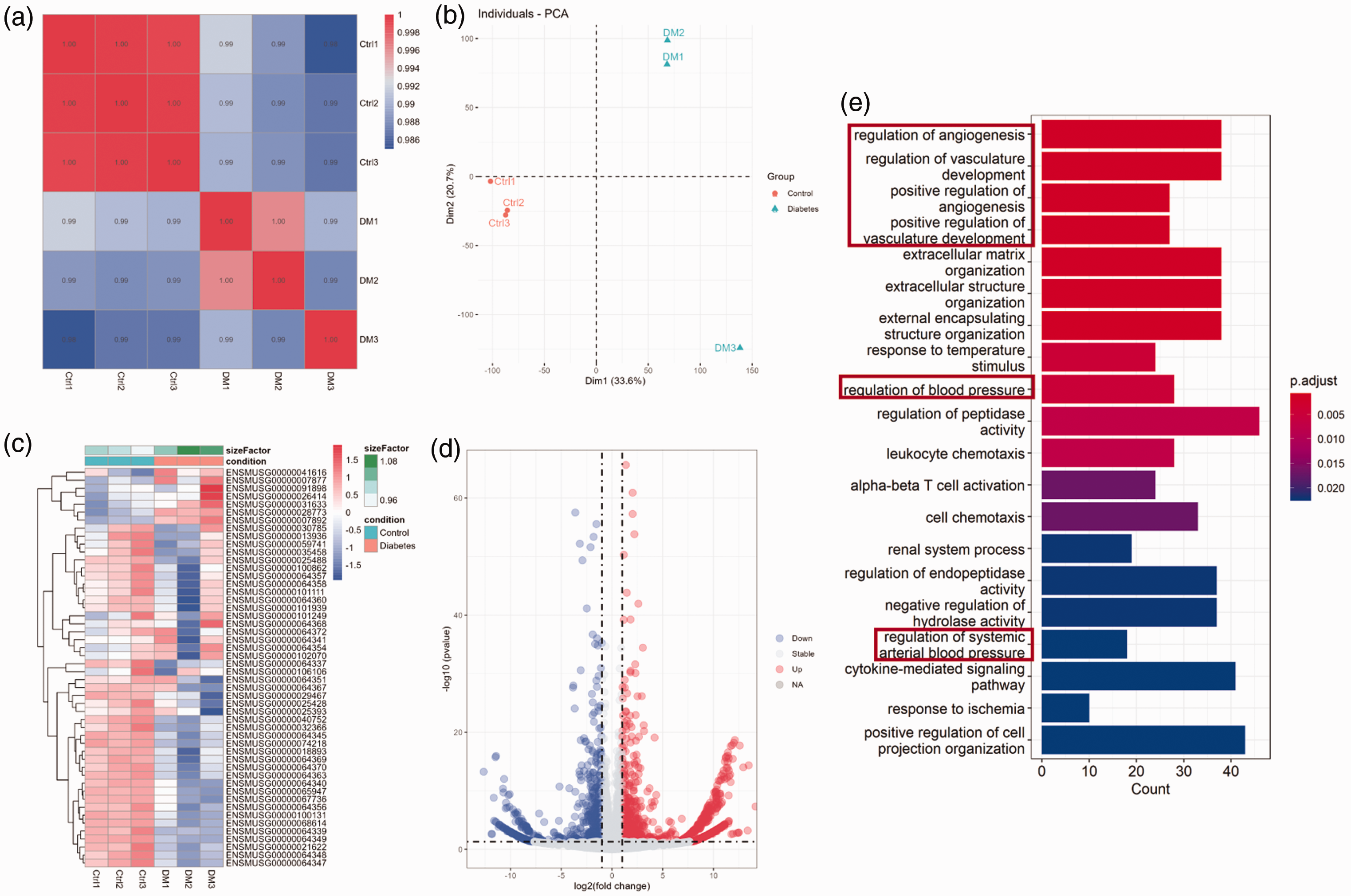
Screening of S1P-acting differential genes in diabetes. (a) Pearson correlation coefficients between samples. (b) Principal component analysis of samples. (c) Analysis of top 50 differential genes. (d) Analysis of differential gene volcano plots and (e) differential gene GO enrichment analysis. S1P: sphingosine-1-phosphate; GO: Gene Ontology.
S1P intervention improved the general condition of diabetic mice
As shown in Figure 3(a), we confirmed that the diabetic mouse models were established successfully. Blood glucose level in the serum of mice in the model group was markedly increased compared with that in the control group, and mice with blood glucose levels above 11.1 mmol/L were diagnosed as diabetic mice. Compared with the control group, the blood glucose level, feed intake, and water intake increased significantly in the model group, accompanied with significantly decreased body weight on days 7, 14, and 21. Meanwhile, compared with the model group, the blood glucose level, feed intake, and water intake decreased accompanied with significantly increased body weight in the S1P group (Figure 3(b) to (e)). These findings indicated that S1P intervention improved the general condition of diabetic mice.

Effects of S1P on the general condition of diabetic mice. (a) Development of the diabetic model. (b) Blood glucose level of mice in each group at different time points. (c) Body weight of mice in each group at different time points. (d) Food intake of mice in each group at different time points and (e) water intake of mice in each group at different time points. Data are presented as the means ± standard error (n ≥ 6 for each group). *p < 0.05. S1P: sphingosine-1-phosphate.
S1P alleviated the pathological damage and inflammatory exudation in the myocardium of diabetic mice
To determine the effects of S1P on the myocardium in diabetic mice, we tested the HW/BW and cardiac pathological morphology in each group. As shown in Figure 4(a), the HW/BW ratio in the model group increased significantly compared with that in the control group. However, in the S1P treatment group, the HW/BW ratio markedly reduced compared with that in the model group. Furthermore, the cardiomyocytes in the control group were neatly arranged, regular in shape, consistent in size, and evenly stained in the cytoplasm and nucleus. In the model group, the arrangement of cardiomyocytes was disordered and the structure was abnormal, a large number of cardiomyocytes dissolved and disappeared, and inflammatory cell infiltration could be seen easily in the myocardial space. However, compared with the model group, the injury degree of the myocardium in the S1P group obviously improved (Figure 4(b)). Furthermore, we detected the effects of S1P on inflammatory exudation in the heart. Compared with the control group, the positive area of CD11b, which mediates the adhesion and migration of inflammatory cells, in the model group significantly increased. In contrast, in the S1P treatment group, the positive area of CD11b in myocardial tissue significantly reduced (Figure 4(c) and (d)). These results suggested that S1P alleviated the pathological damage and inflammatory exudation in the myocardium of diabetic mice.

Effects of S1P on pathological morphology and CD11b expression in the hearts of diabetic mice. (a) Heart-to-body weight ratios. (b) Histological analysis of the myocardium via HE staining. (c) CD11b expression in myocardial tissue detected via the immunohistochemical method and (d) Quantification of CD11b expression. Data are presented as the means ± standard error (n ≥ 6 for each group). **p < 0.01 and ***p < 0.001 vs. control group. #p < 0.05 and ##p < 0.01 vs. model group. S1P: sphingosine-1-phosphate; HE: hematoxylin and eosin.
S1P restored the expression of VE-cadherin, CD31, and VEGF in the myocardium of diabetic mice
To determine the effects of S1P on myocardial vascular homeostasis in diabetic mice, we detected the expression of VE-cadherin, CD31, and VEGF in each group. Compared with the control group, VE-cadherin and VEGF were significantly upregulated, and CD31 was significantly downregulated in the myocardium of diabetic model group. In the S1P group, the expression levels of VE-cadherin and VEGF were downregulated, and the expression of CD31 was significantly upregulated compared with those in the model group (Figure 5(a) to (f)). Moreover, the serum eNOS level of mice in the model group reduced markedly compared with that in the control group. After S1P treatment, the eNOS level was significantly restored to a certain extent compared with the model group (Figure 5(g)). Additionally, we tested the positive area of CD31 in the myocardium and found that CD31 expression significantly decreased in diabetic mice and significantly increased after S1P treatment (Figure 5(h) and (i)), consistent with the results of western blot analysis. These findings indicated that S1P treatment restored the expression of molecules involved in vascular function to some extent in the myocardium of diabetic mice.

Effects of S1P on molecules involved in vascular function in the hearts of diabetic mice. (a–c) The expression levels of CD31, VEGF, and VE-cadherin in myocardial tissue detected via western blot analysis. (d–f) Quantification of CD31, VEGF, and VE-cadherin expression in myocardial tissue in each group. (g) The level of eNOS in the serum detected via ELISA. (h) CD31 expression in myocardial tissue detected via the immunohistochemical method and (i) quantification of CD31 expression. Data are presented as the means ± standard error (n ≥ 6 for each group). **p < 0.01 and ***p < 0.001 vs. control group. #p < 0.05 and ##p < 0.01 vs. model group. S1P: sphingosine-1-phosphate; VEGF: vascular endothelial growth factor; VE: vascular endothelial; eNOS: endothelial nitric oxide synthase; ELISA: enzyme-linked immunosorbent assay.
The imbalance of S1PR1, S1PR2, and S1PR3 expression was relieved after S1P intervention in the myocardium of diabetic mice
We confirmed that S1P can act on multiple targets in mice, including S1PR1, S1PR2, and S1PR3, via raw letter analysis (Table 1). Then, the expression levels of the three receptors in the myocardium were examined in each group. Compared with the control group, the positive areas of S1PR1, S1PR2, and S1PR3 in the model group significantly reduced. However, after S1P intervention, the positive areas of S1PR1, S1PR2, and S1PR3 significantly increased compared with those in the model group (Figure 6(a) to (d)). Meanwhile, we found consistent results in western blot analysis (Figure 6(e) to (j)). These results showed that S1P treatment can relieve the imbalance of S1PR1, S1PR2, and S1PR3 expression in the myocardium of diabetic mice, suggesting that S1PRs participate in the protective roles of S1P during myocardial vascular homeostasis in diabetic mice.
S1P was confirmed to act on S1PR1, S1PR2, and S1PR3 via raw letter analysis.

S1P: sphingosine-1-phosphate; S1PR: sphingosine-1-phosphate receptor.

Effects of S1P on the expression levels of S1PR1, S1PR2, and S1PR3 in the hearts of diabetic mice. (a–d) The expression detection and quantification of S1PR1, S1PR2, and S1PR3 in myocardial tissue via the immunohistochemical method. (e–g) The expression of S1PR1, S1PR2, and S1PR3 in myocardial tissue detected via western blot analysis and (h–j) the quantification of S1PR1, S1PR2, and S1PR3 expression via western blot analysis. Data are presented as the means ± standard error (n ≥ 6 for each group). ***p < 0.001 vs. control group. #p < 0.05, ##p < 0.01, and ###p < 0.001 vs. model group. S1P: sphingosine-1-phosphate; S1PR: sphingosine-1-phosphate receptor.
Discussion
This study showed that S1P treatment ameliorated myocardial vascular homeostasis imbalance in diabetic mice. We first confirmed that S1P was related to vascular homeostasis via bioinformatics analysis. Then, diabetic models were established based on C57BL/6 mice, which were subjected to S1P treatment. We found that S1P relieved several general symptoms of diabetes, such as hyperglycemia, polydipsia, polyphagia, and weight loss. Subsequently, we observed that S1P intervention alleviated the myocardial damage and restored the balance in the expression of VE-cadherin, CD31, and VEGF as well as S1PRs to some extent in the myocardium of diabetic mice. These results suggest that S1P/S1PRs are likely to be a potential modality for the treatment of diabetic myocardial injury.
Previous studies have indicated that chronically high levels of blood glucose can lead to impaired vascular function. 23 In addition, myocardial microvascular dysfunction usually precedes structural changes in diabetic patients. 24 In this experiment, S1P intervention significantly reduced blood glucose levels and improved the general condition of diabetic mice as well as the pathologic change in the myocardium. Meanwhile, the expression of CD11b, a member of the integrin family, which mediates the adhesion and migration of inflammatory cells, 25 increased in the myocardium of diabetic mice, and S1P treatment inhibited this increase in expression significantly. This suggested that S1P might alleviate inflammatory exudation and improve vascular permeability in the diabetic myocardium. Furthermore, we tested the level of vascular function-related proteins in the heart of diabetic mice.
CD31, also known as platelet endothelial cell adhesion molecule (PECAM), is a surface membrane protein of endothelial cells and plays important roles in angiogenesis and endothelial junctional integrity.26,27 This study found that S1P upregulated CD31 expression, which was previously downregulated in the diabetic myocardium, indicating that S1P can facilitate myocardial vascular homeostasis in diabetes.
Research has revealed that S1P can not only bind to the three G protein-coupled receptors (S1PR1, S1PR2, and S1PR3) expressed in the cardiovascular system but also act as a second messenger to play a signaling role, participating in various pathophysiological processes, such as endothelial barrier function, vascular tension, vascular inflammation, and angiogenesis.14,16,28 In addition, He et al. 22 confirmed that S1P may induce islet β-cell proliferation and inhibit apoptosis by activating S1PR1 and S1PR2, thereby enhancing the protection of islet β-cells. Targeting S1P/S1PR signaling pathway may be a new therapeutic strategy for the treatment of insulin resistance and diabetes. 29 Wafa et al. 30 showed that S1PR3 has a direct protective effect on the myocardium and coronary artery function. Therefore, we proposed that S1P plays a role in the protection of myocardial vascular homeostasis in diabetes through S1PRs.
Previous studies have shown that citronellal treatment can alleviate macrovascular and microvascular impairment in diabetic rats by increasing S1PR1 expression, eNOS expression, and NO production and reducing oxidative stress, finally improving vascular function. 31 Moreover, serum S1P can be used as a predictor of the occurrence and severity of coronary artery stenosis. 32 It has been confirmed that VE-cadherin is primarily a type of junctional protein maintaining the endothelial layer integrity and is critically involved in vascular permeability. 33 Furthermore, VEGF has been reported to play a vital role in the processes of angiogenesis. 34 Our recent study found that S1P plays a protective role against high glucose-induced injury of endothelial cells (human umbilical vein endothelial cells) by upregulating VEGF, VE-cadherin, and eNOS expression through S1PR1. 35 However, in this study, we observed that the expression levels of VEGF and VE-cadherin increased in the heart of diabetic mice, and S1P treatment suppressed this increase. Moreover, a study found that after treatment with VEGF inhibitors in diabetic mouse models, the decline in the number of glomeruli in mice was alleviated, and the symptoms of proteinuria were significantly relieved. 36 It has been shown that VE-cadherin expression can affect human retinal microvascular endothelial cell dysfunction. 37 These inconsistent phenomena may result from the different models. Both our studies illustrated that S1P might restore the balance of molecules associated with vascular function. Siragusa et al. 38 found that inhibition of the receptor-type vascular endothelial protein tyrosine phosphatase indirectly enhanced eNOS activity by activating the CD31/VEGFR2 complex and dephosphorylating eNOS Tyr81, thereby improving endothelial function and reducing blood pressure. This is similar to the results of this study, revealing that S1P intervention increased the level of eNOS in the serum of diabetic mice. Furthermore, based on immunohistochemistry and western blot analyses, we verified that S1P intervention significantly upregulated the expression levels of S1PR1, S1PR2, and S1PR3 in the myocardium of diabetic mice, which indicated that S1P can improve the imbalance of myocardial vascular homeostasis by regulating S1PRs. This is similar to a previous study finding that S1PRs may be involved in regulating brain and cardiac function, vascular permeability, and vascular and bronchial tone. 39 In future studies, we will further demonstrate its role in diabetic myocardial injury by designing S1PR inhibition experiments.
This study preliminarily explored the relationship between S1P and vascular homeostasis in the myocardium and revealed that S1P might ameliorate myocardial injury by restoring myocardial vascular homeostasis in diabetic mice. The mechanism seems closely linked to regulating the balance of S1PRs in the heart. However, there are certain limitations in this study, such as the lack of in-depth functional assessment of vascular homeostasis, unclear molecular mechanisms, and lack of information on the particular cell type by which S1P regulates myocardial vascular homeostasis in diabetes. In future research, we will further explore the specific mechanisms of S1P in the protection of the heart in diabetes. In addition, due to limited experimental conditions, the analysis of heart samples in this study was not conducted according to the left ventricle, right ventricle, and atrial division. This may have some influence on the results. In future studies, we will expand the sample size and conduct statistical analysis according to the cardiac cavity partition.
In conclusion, our experiment suggested that S1P treatment relieved diabetic symptoms and may restore myocardial vascular homeostasis by regulating the balance of S1PRs in the hearts of diabetic mice. These findings indicated that S1P/S1PRs are potential therapeutic targets for diabetic myocardial injury.
Footnotes
Acknowledgements
We are very grateful to Wuhan Science and Technology Bureau and Wuhan Fourth Hospital for their kind support toward this work.
Author contributions
HY conceived and designed the study. HY and GJ wrote and revised the manuscript. GJ, HZ, and QC performed the experiments. LL and GJ performed data analysis. All authors have read and approved the final manuscript.
Data availability statement
The raw data are publicly available upon request. The corresponding author can be contacted at
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was funded by the knowledge innovation project of Wuhan Science and Technology Bureau (Grant Number 2022020801020562).
