Abstract
Objective
Endoscopic submucosal excavation (ESE) has been established as an effective method for removal of gastric submucosal tumors (SMTs). The aim of the present study was to explore risk factors for technical difficulties in ESE.
Methods
In this retrospective study, we collected clinical data from patients who underwent ESE for gastric SMTs. Difficult ESE was defined as a procedure time ≥90 minutes, piecemeal resection, and/or occurrence of major adverse events. Univariate and multivariate analyses were performed to explore the risk factors for a difficult ESE.
Results
ESE was successfully performed in 96.5% (195/202) of patients from April 2011 to December 2019. The average tumor size was 17.41 mm, and en bloc resection was achieved in 97.4% of patients (190/195). Five patients (2.56%, 5/195) had complications, including two with delayed bleeding, two with fever, and one with chest pain accompanying ST-T changes in an electrocardiogram. Twenty-four patients (11.88%, 24/202) had a difficult ESE. Logistic analysis showed that outgrowth behavior and an inexperienced surgeon were risk factors for a difficult ESE.
Conclusion
ESE may be safe and effective to treat patients with gastric SMTs. Outgrowth behavior and an inexperienced surgeon were risk factors for a difficult ESE.
Keywords
Introduction
Most patients with gastric submucosal tumors (SMTs) are asymptomatic or present with nonspecific symptoms such as abdominal pain, 1 , 2 and SMTs are occasionally found during esophagogastroduodenoscopy (EGD). For small, asymptomatic gastric SMTs, periodic surveillance using EGD and/or endoscopic ultrasonography (EUS) is usually recommended. 3 However, patients usually feel stress, and some patients cannot adhere to the surveillance strategy. Surveillance is also associated with repeated endoscopic procedures and the risk of a delayed diagnosis of malignancy. Moreover, most of the gastric SMTs are gastrointestinal stromal tumors (GISTs), which have a malignant potential even when they are small. 4 , 5 Therefore, it is necessary to remove these SMTs.
Current strategies to resect a gastric SMT include surgical and endoscopic resection.2,6 Endoscopic resection has several advantages over surgical methods, such as it is minimally invasive, associated with a shorter hospital stay, and has a lower cost.6–9 Available endoscopic methods include endoscopic submucosal dissection (ESD), endoscopic submucosal excavation (ESE), endoscopic full-thickness resection (EFTR), and submucosal tunneling endoscopic resection (STER). 10 , 11 ESD is only recommended for SMTs that originate from the superficial muscularis propria (MP) layer. Moreover, perforation frequently occurs during an ESD procedure for SMTs that are located in deeper layers. 12 In addition, the ESD procedure takes a longer time than ESE and EFTR for deep lesions. EFTR requires full-thickness resection and endoscopic closure of the defect using metallic clips, 13 which further expands the depth of the ESE treatment and broadens the indications for endoscopic treatment. However, this technique has risks including intraperitoneal contamination with gastric contents and intraperitoneal dissemination of tumor cells. STER is used for SMTs that are generally less than 3.5 cm in diameter because larger SMTs are difficult to remove, and this technique is usually used usually for gastric SMTs that are located at selected sites. 14 ESE is a modified ESD technique that has been used effectively to treat gastric SMTs. ESE was first reported by Jeong et al., 15 and this procedure showed a high complete resection rate and an acceptable complication rate. However, ESE is technically difficult, and little is known about the risk factors that are associated with difficult ESEs for gastric SMTs. In this retrospective study, we assessed the safety and efficacy of ESE for gastric SMTs and explored the risk factors for the occurrence of technical difficulties.
Patients and methods
Patients
This single-center, retrospective study was conducted in China. The study was approved by the ethics committee at the Second Xiangya Hospital, Central South University, Changsha, China (No. LYF2020129). All the participants provided written informed consent before treatment. The inclusion criteria for the study were as follows: (a) presence of gastric SMTs originating from the MP layer, which was confirmed by EGD, EUS, and/or computerized tomography (CT), and a preoperative cross-sectional diameter (tumor size) of no more than 5 cm measured using EUS/CT; (b) EUS showing no high-risk features of malignancy, such as internal heterogeneity or heterogeneous enhancement; (c) no signs of metastasis or invasion outside the gastrointestinal tract during CT examination; and (d) patient consented to undergo an ESE procedure at our hospital. Patients who could not tolerate anesthesia and those with severe cardiopulmonary disease or blood coagulation disorders (international normalized ratio >2.0, platelet count <100,000/mm3) were excluded from the study. Two hundred two consecutive patients were enrolled into the study between April 2011 and December 2019. Their demographic information, tumor size, procedure-related parameters, complications, length of hospital stay, and follow-up data were retrospectively collected and recorded. The reporting of this study conforms to the STROBE statement, 16 and we have de-identified all patient details.
A regular tumor shape was defined as an oval or globular shape, and tumors with other shapes were considered to have an irregular shape. Outgrowth behavior was defined as >50% of the cross-sectional diameter of the tumor that was located outside the gastric lining, as revealed by EUS and/or CT. 17 An experienced surgeon was defined as having treated at least 100 patients with of gastrointestinal ESD before performing ESE.
Equipment for ESE
ESE was performed under general anesthesia through tracheal intubation. A carbon dioxide insufflator (UCR; Olympus, Tokyo, Japan) was used. Other equipment and accessories included a high-frequency generator (VIO 200D; ERBE, Tübingen, Germany), an argon plasma coagulation unit (APC300; ERBE), a hybrid knife (ERBE), a dual knife (KD-650Q; Olympus), an insulation-tip knife (KD611L, IT2; Olympus), an injection needle (NM-4L-1; Olympus), and hemostatic clips (HX-600-90; Olympus).
ESE procedure
ESE was performed as previously reported and is briefly described as follows: 18 (a) the tumor location was marked; (b) a submucosal injection was administered and the mucosal and submucosal layers around the lesion were precut; (c) the tumor was exposed using the ESD technique; (d) the submucosal and muscular tissue around the tumor capsule were dissected and peeled away from the tumor; and (e) the mucosal incision or muscular injury was closed using metallic clips with or without nylon loops after careful hemostasis. Figures 1 and 2 depict the ESE procedure for SMTs that are located in the gastric corpus and fundus. A difficult ESE was defined as a procedure time ≥90 minutes, piecemeal resection, and/or the occurrence of major adverse events, such as major bleeding and perforation.
Case illustration of ESE for a SMT located in the gastric corpus.

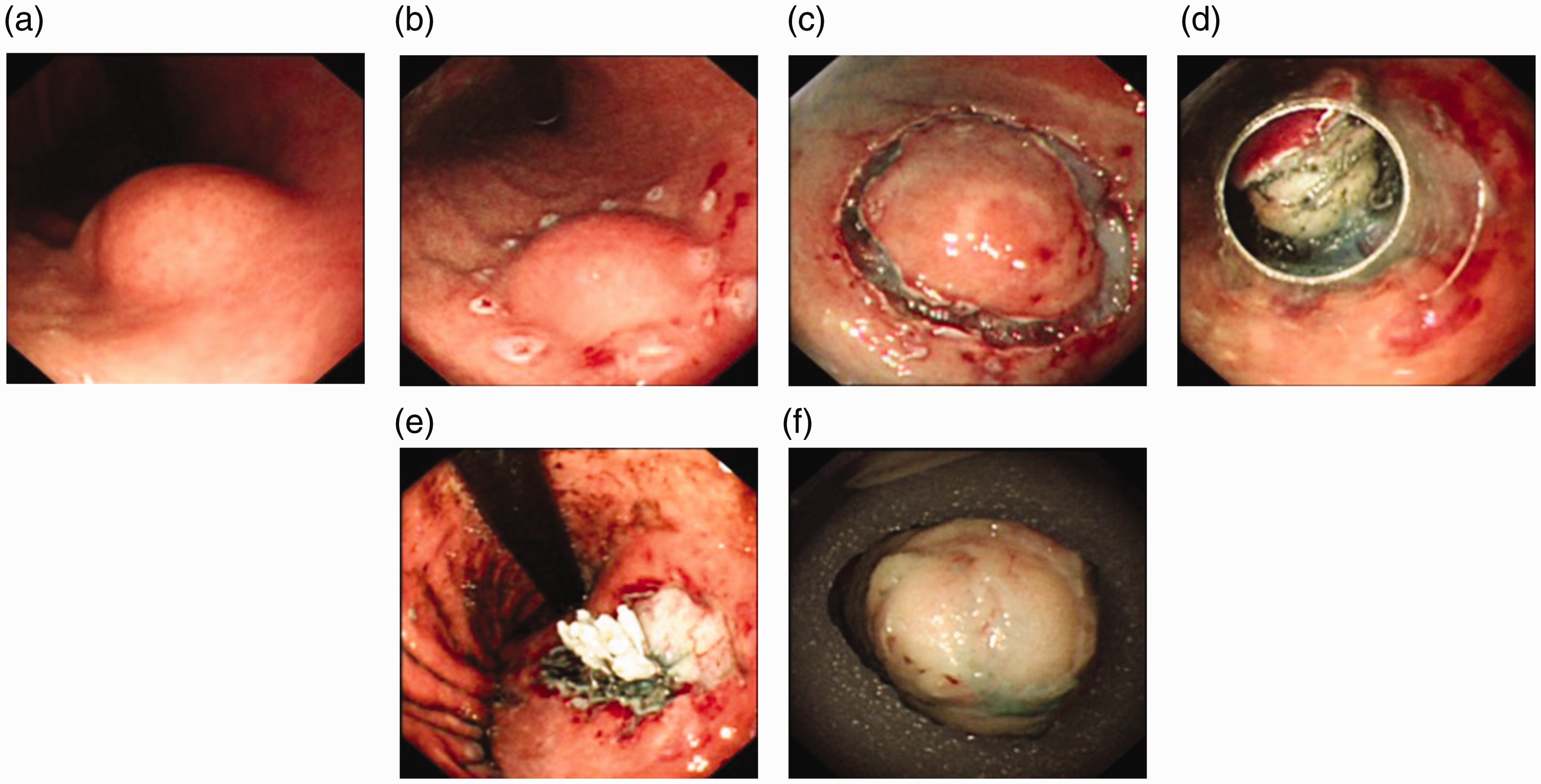
Case illustration of ESE for a SMT located in the gastric fundus. (a) SMT located in the gastric fundus, (b) Precutting the mucosal and submucosal layers around the lesion, (c) Tumor removal, (d) The wound surface after ESE, (e) Closing the wound surface using metallic clips and (f) The resected SMT.
Postoperative management
The postoperative management was performed as previously reported. 19 All the patients were kept nil per oral for 24 hours and then on a liquid diet for 3 to 5 days, followed by a gradual return to a normal diet within 2 weeks. An intravenous proton pump inhibitor (PPI) and antibiotics were used for 3 days. Patients with SMTs in the gastric fundus were asked to stay in a semireclining position for 3 days after ESE.
Pathological evaluation
The specimen was fixed, embedded with paraffin, and then sectioned. Hematoxylin and eosin and immunohistochemical staining (e.g. for SMA, Ki67, CD34, CD117, S-100, desmin, and Dog-1) were performed to determine the SMT characteristics. En bloc resection was defined as an intact fibrous capsule on the resected tumor, and the absence of any remnant of tumor observed on endoscopy.
Follow-up
Periodic follow-up was performed at an out-patient clinic. Surveillance endoscopy was performed at 3, 6, and 12 months and annually thereafter to observe wound healing and to check for any residual tumor. White-light endoscopy was usually performed, and EUS was performed for patients with suspected tumor residual or recurrence.
Statistical analysis
SPSS 21.0 software (IBM Corp., Armonk, NY, USA) was used for data analysis. Continuous variables were presented as the mean ± standard deviation. The Chi-square test or Fisher’s exact test were used for univariate analyses, and logistic regression (“Enter” method) was used for multivariate analyses. A two-tailed P value <0.05 was considered to be statistically significant in all cases.
Results
Clinical characteristics and therapeutic outcomes
Two hundred two patients were consecutively scheduled for ESE from April 2011 to December 2019. Seven of these patients underwent failed ESE (where ESE was started but the procedure was aborted and converted to laparoscopic surgery because of adverse events such as bleeding), but they all underwent laparoscopic surgery. Several patients underwent conversion surgery, as follows: one patient because of massive intraoperative bleeding, one patient because of suspicious malignancy, one because the SMT had grown into the abdominal cavity and was beyond the reach of the endoscope, and the other four patients showed extraluminal growth. All these seven patients had irregularly shaped tumors and showed outgrowth behavior, and six of them had a tumor with a diameter ≥30 mm. Among the 195 patients who completed ESE, the mean age was 48.77 years (range 22–74 years), and the male-to-female ratio was 65:130. There were 195 SMTs that were removed by ESE, the mean surgical time was 39.23 minutes (range 15–210 minutes), and the mean tumor diameter was 17.41 mm (range 5–70 mm). Five patients (2.56%) were not able to undergo en bloc resection; among them, four patients had an excessive tumor diameter, and one patient had fragile tissue. All the five SMTs had a diameter of ≥30 mm, and four of them had an irregular shape. Among the 202 SMTs, six were located at the cardia, 125 in the gastric fundus, 62 in the gastric body, and 9 in the antrum. Among the 202 patients, 128 were diagnosed with gastrointestinal stromal tumors (GISTs) pathologically, which included 58 leiomyomas, four neurofibromas, four schwannomas, three lipomas, three patients with a heterotopic pancreas, and one splenosis. The 128 GISTs were classified on the basis of the National Comprehensive Cancer Network guidelines, 20 and included 83 that were very low risk, 42 that were low risk, and three that were moderate risk. No recurrence was noted during a mean follow-up of 54 months, except for one patient who had developed another gastric SMT adjacent to the primary site at the 3-year follow-up.
Complications
Five patients (2.56%, 5/195) had complications, including two patients with delayed bleeding, two with fever, and one with chest pain that was accompanied by ST-T changes in the ECG. For the two patients with delayed bleeding, both of them had hematemesis, and one of these patients underwent successful conservative treatment, while the other patient underwent endoscopic treatment. For the two patients with fever, only one of them had a temperature higher than 38.5°C, and none of them had abdominal cavity effusion on abdominal ultrasound. Their symptoms resolved 1 to 3 days after ESE after treatment using a PPI and antibiotics. One patient had chest pain that was accompanied by transient ST-T changes on the ECG, but their serum creatine kinase (CK), CK-MB, and troponin test results were normal, and the pain resolved within 2 days.
Long procedure time
Eleven patients had a procedure time of ≥90 minutes. A univariate analysis revealed that irregular shape, tumor outgrowth, and the surgeon’s experience level were risk factors for a long procedure time (P < 0.05 for all; Table 1). A multivariate analysis revealed that outgrowth and an inexperienced surgeon were risk factors for a long procedure time (P = 0.016, P = 0.002, respectively; Table 1).
Univariate and multivariate analyses of risk factors for a long procedure time.

GISTs, gastrointestinal stromal tumors; OR, odds ratio; 95%CI, 95% confidence interval.
Difficult ESE
Twenty-four patients (11.88%, 24/202) had a difficult ESE (Table 2). A univariate analysis revealed that tumor size, irregular shape, outgrowth, surgeon experience, and pathology were risk factors for a difficult ESE (P < 0.05 for all; Table 3), while a multivariate analysis revealed that outgrowth and an inexperienced surgeon were risk factors for a difficult ESE (P = 0.001, P = 0.007, respectively; Table 3).
Detailed information about the 24 patients who were defined as having a difficult ESE.

ESE, endoscopic submucosal excavation; F, female; M, male; GIST, gastrointestinal stromal tumor; –, no data.
Univariate and multivariate analyses of the risk factor for a difficult ESE.

ESE, endoscopic submucosal excavation; GISTs, gastrointestinal stromal tumors; OR, odds ratio; 95%CI, 95% confidence interval.
Discussion
In 2011 (the first ESE was performed in 2006), Jeong et al. 15 first reported the ESE method (which was called endoscopic enucleation) to treat patients with gastric SMTs, and 60 of 65 gastric SMTs were removed by ESE. Thereafter, an increasing number of clinical studies have confirmed the safety and efficacy of ESE for treating gastric SMTs, with a reported success rate of 92.3% to 100%, an en bloc rate of 77.8% to 100%, and a complication rate of 0% to 23.7%.21–25 In the present study, ESE was successfully performed in 96.5% of the patients, the en bloc rate was 97.4%, and the complication rate was 2.56%, which was comparable with the above studies. This indicates that ESE is a safe and effective method for removal of gastric SMTs.
ESE has several advantages over other endoscopic methods for gastric SMTs. Compared with multiband mucosectomy and ESD, ESE can allow more tumors to be resected in en bloc and increase the complete resection rate for SMTs that are ≥15 mm. 25 Although the complete resection and complications rates are comparable between EFTR and ESE, ESE had a shorter fasting time, shorter hospital stay, and lower cost.26,27 Although STER reduced the potential rate of air leakage and perforation, ESE was associated with a shorter surgical time, and it is preferable for SMTs that are ≥40 mm or those that are located at sites for which it is difficult to create a submucosal tunnel.18,28
Although ESE is a feasible technique for gastric SMTs, it is a relatively complicated procedure, and technical difficulties sometimes occur even for experienced surgeons, which ultimately prolongs the procedure time or leads to procedure-related complications. In the present study, we defined a difficult ESE for the first time as a procedure time that was ≥90 minutes, included piecemeal resection, or had major complications that occurred. On the basis of this definition, 11.88% of the patients had a difficult ESE. Further analyses revealed that outgrowth behavior and an inexperienced surgeon were risk factors for a difficult ESE. Jeong et al. 15 found that the complete resection and complication rates of ESE were related to the location, and lesions located at the fundus had a higher rate of complications and a lower rate of complete resection. Li et al. 26 found that tumor size was a significant contributor to a longer procedure time. Additionally, our previous study on EFTR revealed that a tumor size of ≥30 mm and a tumor located in the gastric corpus are risk factors for technical difficulties. 17 In the present study, all five patients who had a failed en bloc resection had a tumor diameter of ≥30 mm. Thus, tumor size was a risk factor for a difficult ESE, as shown by the univariate analysis, but it was not a risk factor in the multivariate analysis. This may be because of the relatively small proportion (23/202) of patients with a tumor diameter of ≥30 mm. Du et al. 28 found that an irregular tumor shape was a risk factor for piecemeal resection. In the present study, four of the five patients with incomplete resection had an irregular tumor shape, and an irregular tumor shape was a risk factor for both a long procedure time and a difficult ESE in the univariate analysis, but they were not risk factors in the multivariate analysis. Outgrowth behavior and an inexperienced surgeon were demonstrated to be risk factors for both a long procedure time and a difficult ESE. It is easy to understand that outgrowth behavior is a risk factor because excessive dissection or even conversion to EFTR is necessary for en bloc resection of the SMT, which usually requires a long time, and this was shown by the multivariate analysis of the risk factors for long procedure time. An inexperienced surgeon usually needs more time to complete the procedure and manage potential complications, and thus, this was also a risk factor for a difficult ESE. This result suggested that ESE should be performed by an experienced surgeon. We recommend that a minimum of 100 upper gastrointestinal ESD procedures should be completed before ESE is performed.
The present study had some limitations. First, this was a single-center, retrospective study. Second, the sample size was relatively small, with only 202 patients enrolled, and thus, a prospective, large-scale study is required to verify the conclusion. The present study showed that ESE is a safe and effective method to treat patients with gastric SMTs. Outgrowth behavior and an inexperienced surgeon were risk factors for a difficult ESE.
Footnotes
Acknowledgements
We would like to thank the staff from the Department of Pathology for their pathologic diagnosis of the SMTs, and the staff from the Department of Statistics at Central South University for their kind help with the statistical analysis.
Author contributions
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
