Abstract
Much attention has been paid to renal hemangioblastoma, but there are still challenges in its differential diagnosis. Three cases (2 men, 1 woman; age: 40–56 years) presented with renal tumors. The tumors were surrounded by a thick fibrous capsule, well-demarcated from the surrounding renal parenchyma, and composed of sheets or nests of polygonal to short spindle-shaped tumor cells with a rich capillary network. In cases 1 and 3, the large polygonal tumor cells contained abundant pale or eosinophilic cytoplasm, and some possessed intracytoplasmic lipid vacuoles. In case 2, tumor cells were characterized by a uniform size, mild, clear, or lightly stained cytoplasm, and typical "clear cell" appearance. Immunohistochemistry revealed that the polygonal stromal cells were strongly and diffusely positive for α-inhibin, neuron-specific enolase (NSE), S100 protein, and vimentin. Cluster of differentiation (CD)10 and paired box gene (PAX)8 were positive, while epithelial membrane antigen (EMA) and cytokeratin (CK) were focally positive in case 3. CD34 and CD31 outlined the contours and distribution of the vascular networks. Renal hemangioblastoma is rare and prone to misdiagnosis; more attention should be paid to the morphological features and reasonable application of immunohistochemistry in the diagnosis of hemangioblastoma.
Background
Hemangioblastoma, a benign tumor of unknown histogenesis, mainly occurs in the central nervous system (CNS), such as in the cerebellum, brainstem, and medulla oblongata. In many cases, the pathogenesis of hemangioblastoma is associated with von Hippel–Lindau (VHL) disease, which is an autosomal dominantly-inherited neoplastic disorder with germline mutations in the
The differential diagnosis of sporadic renal hemangioblastoma is a major clinical challenge because this disease is similar to malignancies, such as renal cell carcinoma (RCC). In this case report, we present three cases of sporadic renal hemangioblastoma without clinical evidence of VHL disease. We also performed a literature review to further illustrate the clinicopathologic features and differential diagnosis of this rare renal tumor.
Case presentation
Material and methods
Tumor samples were obtained from all cases after nephrectomy. Then, the samples were fixed in 10% buffered formalin, followed by embedding in paraffin. The sections (4 μm) were stained with hematoxylin and eosin, and the histological features of the tumors were evaluated by two experienced pathologists. Immunohistochemical (IHC) staining was performed with the Ventana BenchMark XT automated IHC stainer (Roche, Basel, Switzerland). Sections treated with phosphate buffered saline (PBS) served as negative controls, and the positive controls were the specific tissues, according to the manufacturer’s instructions. Antibody information is provided in Table 1. The reporting of this study conforms to the CARE guidelines. 3
Antibody information in our and case of renal hemangioblastoma.

CK, cytokeratin; EMA, epithelial membrane antigen; CD10, cluster of differentiation 10; CgA, human chromogranin A; NSE, neuron-specific enolase; PAX8, paired box gene 8; SMA, smooth muscle actin; Dako, DAKO Diagnostics, Mississauga, Canada; Novocastra Laboratories, Novocastra Laboratories Ltd., Newcastle upon Tyne, UK; Proteintech Group, Proteintech Group Inc., Chicago, IL, USA.
Results
Clinical history of the three cases in this study

Computed tomography (CT) and macroscopic findings. (a) CT image showing a heterogeneous soft tissue mass at the mid-lateral pole of the right kidney in case 1. (b) The cut surface of the tumor was a grayish-red to grayish-brown color. The tumor was solid and tough, with no necrosis or cystic degeneration. (c) A round mass was seen at the middle pole of the right kidney in case 2, with a clear boundary and significant enhancement. (d) The tumor was solid and tough, and the cut surface was a grayish-red and grayish-brown color with a small grayish-white area in the center. No necrosis or cystic changes were observed. (e) CT image in case 3 showing a rounded mass in the dorsal right kidney. The lesion had a clear boundary, protruded extrarenally and pelvically, and had low-density necrosis. (f) A well-circumscribed, soft grayish-brown tumor is visible in the middle of the kidney.
Microscopic features
In all cases, the tumors were surrounded by a thick fibrous capsule and were well-demarcated from the surrounding renal parenchyma (Figure 2a). Tumors were composed of sheets or nests of polygonal to short spindle-shaped tumor cells and a rich capillary network. In case 1 and case 3, the large polygonal tumor cells contained abundant pale or eosinophilic cytoplasm, and some of the cells contained intracytoplasmic lipid vacuoles. In case 1, the cellular morphology was diverse, with varying nuclear size. Some cells showed atypical features with no karyokinesis (Figure 2b and 2c). However, tumor cells were characterized by uniform size; mild, clear, or lightly stained cytoplasm; and typical “clear cell” appearance in case 2 (Figure 2d). Additionally, in case 2, stromal cells were scattered in small nests among the vascular networks. No mitosis and necrosis were found in all cases. Significant stromal cell hyaline degeneration was seen in case 3 as well as psammoma-like calcification (Figure 2e and 2f). In all three cases, there was edema and hemosiderin deposition in the interstitium.

Histological features. (a) Case 2. The tumor is surrounded by a thick fibrous capsule and is well-demarcated from the surrounding renal parenchyma under low magnification (×5). (b) The large polygonal tumor cells contain abundant pale or eosinophilic cytoplasm. Some of the cells contain intracytoplasmic lipid vacuoles (case 3) under high magnification. (c) Cell morphology, nuclear size, and atypical features of some cells in case 1 under high magnification. (d) Tumor cells are characterized by uniform size with mild, clear, or lightly stained cytoplasm and typical "clear cell" appearance (case 2) under high magnification. (e) Significant stromal cell hyaline degeneration in case 3 under high magnification. (f) Psammoma-like calcification in case 3 under high magnification.
Immunohistochemistry
The polygonal stromal cells demonstrated strong and diffuse positivity for α-inhibin (Figure 3a), neuron-specific enolase (NSE) (Figure 3b), S100 protein (Figure 3c), and vimentin. Cluster of differentiation (CD)10 (Figure 3d) and paired box gene (PAX)8 (Figure 3e) expression was positive in case 3. Additionally, epithelial membrane antigen (EMA) and cytokeratin (CK) were focally positive in case 3. The neoplastic cells uniformly showed negative reactivity for immunohistochemical antibodies to CK8/18, HMB45, MelanA, human chromogranin A (CgA), Syn, smooth muscle actin (SMA), desmin, and CD56. CD34 and CD31 outlined the contours and distribution of the vascular networks in the tumors (Figure 3f). Ki67 showed a proliferative index of less than 3% (Table 2).

Immunohistochemical findings. (a) Tumor cells demonstrating diffuse and strong reactivity for α-inhibin in case 1 under high magnification. (b) Diffusely and strongly positive reaction against neuron-specific enolase (NSE) in case 1 under high magnification. (c) S100 positive expression in case 2 under high magnification. (d) Diffusely positive reaction against cluster of differentiation (CD)10 in case 3 under high magnification. (e) Tumor cells positive for PAX8 in case 3 under high magnification. (f) CD34 highlighted in the endothelial cells lining the rich vascular network in case 2 under high magnification.
Summary of the results for each antibody for all three cases.
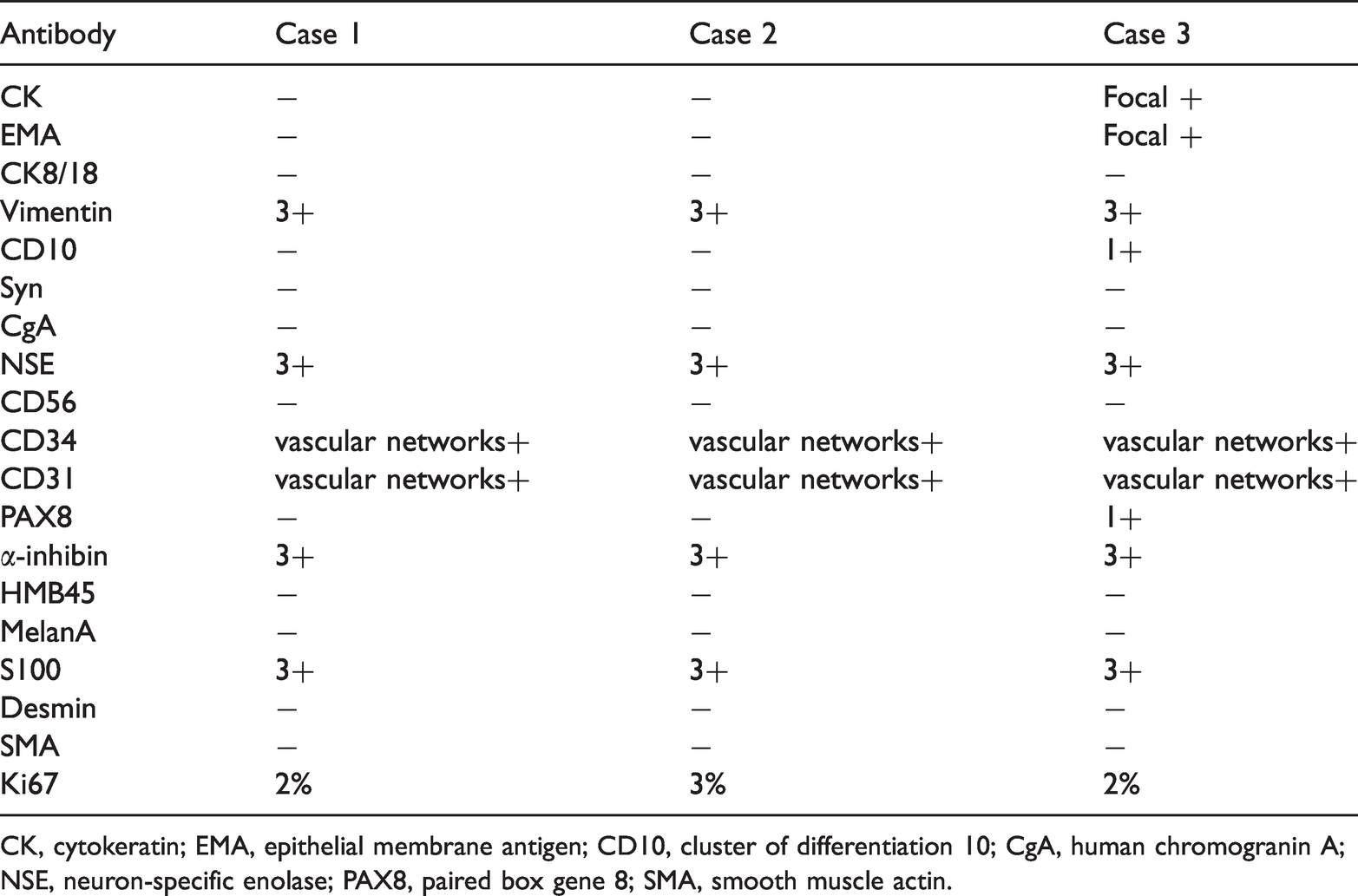
CK, cytokeratin; EMA, epithelial membrane antigen; CD10, cluster of differentiation 10; CgA, human chromogranin A; NSE, neuron-specific enolase; PAX8, paired box gene 8; SMA, smooth muscle actin.
Discussion
Hemangioblastoma is often sporadic and is partly related to VHL disease. Sporadic hemangioblastoma mainly occurs in the cerebellum and typically presents as cystic masses. Multiple hemangioblastomas can also involve the brainstem, spinal cord, nerve roots, and retina, which are closely connected with VHL disease. Recently, increased attention has been paid to extraneural hemangioblastomas.2,4,5 To our knowledge, there have been 24 cases of sporadic renal hemangioblastomas reported in the English literature.4–17 The clinical characteristics of previously reported cases, with those of the three cases in the current study, are summarized in Table 3. Twenty-one of 24 cases in the literature as well as our 3 cases had complete clinical data. The tumors in 19 cases (70.3%) were located in the right kidney, and most patients were men (17/26, 65.4%). Among the male cases, 14 (53.8%) were aged <50 years (range: 16–71 years; mean age: 45.19 years), and the tumor size ranged from 1.2 cm to 15 cm (mean size: 4.2 cm). Sporadic renal hemangioblastomas was more likely to occur in the right kidney. It appears that this disease was more likely in young adults, but more patients are required to confirm this phenomenon. There were no significant clinical symptoms among the reported cases, and occasional symptoms, such as lumbar pain or hematuria were observed. CNS hemangioblastoma could secrete erythropoietin, which then resulted in secondary polycythemia. In one previous study, a case with renal hemangioblastoma showed erythrocytosis, which indicated that renal hemangioblastoma may have the capacity to secrete erythropoietin. 7 However, more studies are required. CT showed a clear, round, or quasi-round low-density mass in the renal parenchyma, which showed uneven enhancement. These masses could easily be misdiagnosed as clear cell RCC or angiomyolipoma. Interestingly, Bisceglia et al. 2 found that most cases reported in the literature were from the Far East, and the authors considered that there might be an unknown genetic feature common to members of a particular ethnic or racial group.
Clinical characteristics of the 27 reported cases of renal hemangioblastoma.

R, right; L, left; M, male; F, female; NED, no evidence of disease; VHL, von Hippel–Lindau disease.
Macroscopically, most tumors appear as well-defined masses surrounded by a thick fibrous capsule; only two cases presented with areas of poorly marginated growth or with the tumor breaking through the fibrous capsule.6,11 In contrast to CNS hemangioblastoma, renal tumors generally presented as a solid mass, occasionally with small focal cystic changes.7,15 The cut surface often appeared grayish-white, grayish-yellow, grayish-brown, or reddish-brown to grayish-yellow in color, and focal hemorrhage and necrosis were observed incidentally. 13
Histologically, renal hemangioblastomas were well-demarcated from the surrounding renal parenchyma, and some cases showed fibrous pseudocapsules. Similar to CNS hemangioblastoma, renal hemangioblastoma was composed of large, polygonal vacuolated stromal cells with a rich capillary network. According to the abundance of stromal cells, tumors were divided into cellular and reticular subtypes. Tumor cells showed no atypia, presenting with round nuclei with fine chromatin, cytoplasmic microvacuoles, and rich in lipids. Eosinophilic hyaline globules were found both within the tumor cell cytoplasm and within the stroma in two cases.7,12 Atypical cells and cells with rhabdoid features presented in some tumors, which may be mistaken for malignant tumors.13,17 In addition, a few cells exhibiting mitotic activity, and focal necrosis, were also documented.11,13 Except for the classical rich capillary network, dilated thick-walled vessels that are similar to the thick-walled veins in angiomyolipoma can be observed in renal hemangioblastoma, and stromal edema, fibrosis, and hyalinization were also common findings. Focal calcification of the fibrous stroma and psammoma body-like calcifications were described in some studies.9,12,15 In the current study, tumor cells were distributed in sheets or lobules, with abundant eosinophilic cytoplasm, and some had lipid vacuoles (case 1 and case 3). In some areas, atypical cells could be seen in case 1. In case 2, the stromal cells were scattered in small nests among the vascular networks, and cells had transparent cytoplasm. Significant hyaline degeneration of stromal cells was seen in case 3, with psammoma-like calcification. Edema and hemosiderin deposition was observed in the interstitium in all three cases.
According to the literature review, more than 20 antibodies were used in the differential diagnosis of renal hemangioblastomas, namely NSE, S100, α-inhibin, vimentin, anticytokeratin antibodies AE1/AE3, EMA, PAX8, PAX2, CAIX, CD10, CK7, CK8/18, calretinin, HMB45, MelanA, CgA, Syn, SMA, desmin, merozoite surface antigen (MSA), calponin, GLUT-1, brachyury, CD34, and CD31. NSE, S100, vimentin, and α-inhibin were strongly positive in all reported renal hemangioblastomas, similar to the expression in CNS hemangioblastomas.8,11 In contrast, the expression of CK7, CK8/18, calretinin, HMB45, MelanA, CgA, Syn, and desmin was negative in all cases. CD34 and CD31 outlined the contours and distribution of the vascular networks in the tumors. However, renal hemangioblastomas showed distinct immuno-profiles from CNS tumors. RCC markers (i.e., PAX8, PAX2, CAIX, and CD10) were diffusely expressed, or at least focally expressed, in renal hemangioblastoms.6,11–13,16,17 Regarding the abnormal immunohistochemical expression in renal hemangioblastomas, several authors speculated that the possible explanation was that these tumors were derived from pluripotent mesenchymal cells and acquired site-specific markers of their parental organs during tumorigenesis.12,13,15 Additionally, AE1/AE3 and EMA were also locally expressed in renal hemangioblastomas.7,12,13,16 Similar to previous cases, all cases in our study were positive for NSE, S100, α-inhibin, and vimentin. In addition, one case was positive for PAX8 and CD10 and focally positive for EMA. CK8/18, HMB45, MelanA, CgA, Syn, SMA, desmin, and CD56 were negative. Expression of GLUT1 in CNS hemangioblastoma has been reported to be helpful in differentiating hemangioblastoma from metastatic RCC. Unlike RCC staining, there is strong endothelial staining in most hemangioblastomas. 18 However, these findings are controversial because there are doubts regarding the low specificity and the tendency for high background staining. 19 Recently, expression of brachyury has been described in cerebellar hemangioblastoma tumor cells.19,20 Tirabosco et al. 19 reported nuclear expression of brachyury in stromal cells in 14 CNS cases. However, Doyle and Fletcher 5 reported no expression of brachyury in extraneural tumors.
VHL disease is an autosomal dominant hereditary disorder leading to clear cell tumors in various organs, such as CNS hemangioblastoma, pancreatic microcystadenoma, pheochromocytoma, epididymal papillary cystadenoma, endolymphatic sac tumor of the inner ear, and renal clear cell carcinoma. The
Renal hemangioblastoma is rare and is most likely to be misdiagnosed as clear cell RCC because it is characterized by a rich vascular network and epithelioid cells with clear or eosinophilic cytoplasm. Regarding the organizational structure, sheet-like or compact architecture was a common feature of hemangioblastomas in previous cases. In contrast, clear cell RCC is more likely to be characterized by tubular, alveolar, nests, or papillary structures, and clinically, these differences might be clues for the differential diagnosis of hemangioblastomas. Additionally, immunohistochemistry plays a key role in distinguishing clear cell RCC from hemangioblastoma. NSE, S100, and α-inhibin were expressed in almost all hemangioblastomas, while there was no expression in clear cell RCC. Furthermore, clear cell RCC is usually positive for AE1/AE3, PAX8, PAX2, CD10, and carbohydrate antigen (CA)9. However, two cases of clear cell RCC with hemangioblastoma-like features were recently reported.21,22 Notably, some RCC markers were positive in hemangioblastoma, as mentioned; therefore, a panel of antibodies was required for the differential diagnosis. Another often confused major tumor is epithelioid angiomyolipoma (AML), in which the diagnosis could be based on the presence of large thick-walled veins, spindle-shaped smooth muscle bundles, and various proportions of mature adipose tissue. Immunohistochemically, AML stains positively for HMB45 and MelanA and negative for α-inhibin, NSE, and S100 in contrast to hemangioblastoma. Other differential diagnosis are adrenocortical adenocarcinoma and paraganglioma, which can be distinguished by careful morphological observation and appropriate immunohistochemistry.
Conclusion
Renal hemangioblastoma is a rare benign tumor. We reported three cases of renal hemangioblastoma, with a complete literature review. Clinically, hemangioblastoma occurs in middle-aged and younger people. Micromorphologically, hemangioblastoma shares similarities with clear cell RCC and AML. Genetically, renal hemangioblastoma appears to have no relationship to
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211027774 - Supplemental material for Sporadic hemangioblastoma of the kidney: a clinicopathologic study of three cases and a literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605211027774 for Sporadic hemangioblastoma of the kidney: a clinicopathologic study of three cases and a literature review by Yanmei Xu, Xuehua Ma, Yong Ma, Juan Li, Renya Zhang and Xiaomei Li in Journal of International Medical Research
Footnotes
Ethics statement
The study protocols were approved by the Ethics Committee of the Tai’an Central Hospital (approval number: 2021-05-10). Each patient provided written informed consent for publication of this article and any accompanying images.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Authors' contributions
YX wrote the manuscript. XL revised the manuscript. XM and YM performed the data analysis. JL and RZ collected the data. All authors read and approved the final manuscript.
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
