Abstract
Objective
Prognostic indicators in acute coronary syndrome (ACS) would aid in decision-making and identifying high-risk patients. The systemic immune-inflammation index (SII) has good prognostic value in many diseases; however, its use has not been reported for ACS. We aimed to determine the associations between the SII and outcomes in patients with ACS, with adjustment for confounders.
Methods
In this retrospective cohort study, we used the MIMIC-III (Multiparameter Intelligent Monitoring in Intensive Care) database and the eICU Collaborative Research Database. The primary outcome was 30-day mortality. Cox regression analysis was performed to determine the relationship between the SII and patient outcomes, and we conducted subgroup analysis and smooth curve fitting.
Results
We identified 4699 patients with ACS: 1741 women and 2949 men, mean age 82.8±29.7 years, and mean SII 72.58±12.9. For 30-day all-cause mortality, the unadjusted hazard ratio (HR) (95% confidence interval [CI]) of SII <69.4 and SII >88.8 were 1.25 (1.04, 1.50) and 1.38 (1.15, 1.65), respectively. With SII >88.8, this association remained significant after adjustment for numerous potential confounders: HR 1.27 (1.06, 1.52). A similar relationship was observed for 90-day and 1-year all-cause mortality.
Conclusions
SII is a promising prognostic indicator for unselected patients with ACS. This finding needs to be confirmed in prospective studies.
Keywords
Introduction
Acute coronary syndrome (ACS) is rapidly becoming a global public health concern. 1 Despite advances in understanding of the mechanisms in ACS, approximately 40% of patients who experience a coronary event will die within 5 years; the risk of death is 5 to 6 times higher in people who experience recurrent events. 2 Because of the high morbidity and mortality in the population with ACS, identification of prognostic indicators would assist medical decision-making and help identify patients at high risk of developing ACS.
Chronic low-grade inflammation has an important role in the biology of cardiovascular diseases.3–5 The Global Use of Strategies to Open Occluded Arteries Trial IV (GUSTO-IV) showed C-reactive protein level to be an independent predictor of outcome in ACS. 6 The Canakinumab Anti-inflammatory Thrombosis Outcomes Study verified that anti-inflammatory therapy targeting the interleukin-1β innate immune pathway led to a significantly lower rate of recurrent cardiovascular events. 7 The systemic immune-inflammation index (SII) is a cost-effective biomarker, easily obtained from complete blood counts because it is based on neutrophil, lymphocyte, and platelet counts.8–10 The SII was initially used to assess the prognosis of patients with cancer; 11 currently, it is used as a biomarker for the evaluation of inflammation status. Epidemiological studies have found that in some disease, the SII is a stronger prognostic index than systemic inflammation response biomarkers, such as the neutrophil–lymphocyte ratio, platelet–lymphocyte ratio, and monocyte–lymphocyte ratio.12,13
To the best of our best knowledge, no studies have examined whether the SII can predict prognosis in patients with ACS. Therefore, we used the Multiparameter Intelligent Monitoring in Intensive Care (MIMIC) 14 database to evaluate the association of the SII with outcomes in patients with ACS.
Methods
Data source
The study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 15 This was a retrospective cohort study using the MIMIC-III database (version 1.4) 14 and the eICU Collaborative Research Database. 16 The application for data use was approved by the Massachusetts Institute of Technology and our institutional review board. Definitions of type of ACS were based on International Classification of Diseases Ninth Revision (ICD-9) codes.17,18 The inclusion criterion was a diagnosis of ACS. The exclusion criteria were 1) stay in the intensive care unit (ICU) of less than 48 hours; 2) no neutrophil, lymphocyte, or platelet counts within the first 24 hours after admission; and 3) any cancer or malignancy.
Assessment of SII
Lymphocyte, neutrophil, and platelet counts were evaluated using automated hematology instruments and were expressed as ×109 cells/mm3. The SII was calculated as platelet count × neutrophil count/lymphocyte count. 18 Study participants were subdivided into three groups according to SII tertiles.
Study variables and outcomes
We recorded the following: age, sex, race, body mass index (BMI), vital signs, laboratory characteristics, comorbidities, and index scores (described below). Vital signs included heart rate, oxygen saturation (SPO2), systolic blood pressure (SBP), and diastolic blood pressure (DBP). Comorbidities included hypertension, stroke, diabetes mellitus (DM), coronary heart disease (CHD), chronic heart failure (CHF), alcohol abuse, and depression. The 30-item Elixhauser Comorbidity Index (Elixhauser-30) was calculated to identify comorbidities at the time of admission. 19 Laboratory characteristics included neutrophil counts, lymphocyte counts, erythrocyte counts, hemoglobin levels, platelet counts, blood urea nitrogen (BUN) levels, serum sodium levels, serum potassium levels, serum chloride levels, prothrombin time, and international normalized ratio over the first 24 hours in the ICU. The Simplified Acute Physiology Score II (SAPS II), 20 Sequential Organ Failure Assessment (SOFA),20,,21 and Glasgow Coma Scale (GCS) 22 scores were also recorded.
The primary outcome in our study was 30-day mortality, and the secondary outcomes were 90-day and 1-year mortality. The date of admission for eligible participants was the start date for follow-up, and all participants were followed for at least 1 year. The date of death was obtained from Social Security death index records.
Ethics
MIMIC-III obtained approval from the Institutional Review Boards of the Beth Israel Deaconess Medical Center and the Massachusetts Institute of Technology. To gain access to this database, we completed online courses required by the National Institutes of Health and completed all tests related to protecting human research participants. Our institutional ethics review committee waived the need for approval of the study protocol and informed consent because this study involved a retrospective analysis using a public database.
Statistical analysis
To assess the relationship between the SII and outcomes, the statistical analysis process was divided into three steps. Continuous data are expressed as mean±standard deviation (SD), and categorical variables are expressed as frequency or percentage. Continuous and categorical variables were compared among tertiles of SII, using analysis of variance. Next, Cox regression analysis was performed to assess the relationship between the SII and outcomes in patients with ACS. Hazard ratios (HRs) with 95% confidence intervals (CIs) were calculated. For the SII at admission, 69.4–88.8 was selected as the reference range for outcome comparison. In Model 1, no covariates were adjusted; in Model 2, only age, sex and race were adjusted. Model 3 included all factors in model 2 were adjusted, plus BMI, heart rate, SPO2, hemoglobin level, lymphocyte count, leucocyte count, neutrophil count, serum glucose level, chronic conditions, and scores for the GCS, SOFA, and Elixhauser-30. Third, we used a weighted generalized additive model (GAM) and smooth curve fitting to identify nonlinearity of the SII and outcome of patients with ACS. The point of inflection was identified by applying a recursive algorithm. On both sides of the inflection point, a model of weighted two-piecewise linear regression was applied.
For data analysis, we used R software (The R Project for Statistical Computing, Vienna, Austria). All two-sided P values <0.05 were considered statistically significant.
Results
Patient characteristics
We identified 4699 patients with ACS who met our inclusion criteria. Eligible participants included 1741 women and 2949 men, with mean age 82.8±29.7 years and mean SII 72.58±12.9. Participants were divided into tertiles according to SII <69.4 (N=1563); 69.4 to 88.8 (N=1563); and >88.8 (N=1564) (Table 1). Among SII groups, the distributions according to BMI, SBP, SPO2, and heart rate were similar. Compared with the SII <69.4 and 69.4 to 88.8 groups, participants with a higher SII were more likely to be young, male, and to have a greater number of chronic conditions (CHD, DM, and Elixhauser-30). These groups had higher levels of platelet counts, neutrophils, BUN, and serum glucose.
Baseline characteristics of the study population.

Data are presented as mean±standard deviation or n (%).
SII, systemic immune-inflammation index; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; SPO2, percutaneous oxygen saturation; CHD, coronary heart disease; CHF, chronic heart failure; DM, diabetes mellitus; WBC, white blood cell; BUN, blood urea nitrogen; PT, prothrombin time; INR international normalized ratio; SAPS II, Simplified Acute Physiology Score II; SOFA, Sequential Organ Failure Assessment; GCS Glasgow Coma Scale.
Association between SII and outcomes
We constructed various models to assess independent effects of the SII and outcome of patients with ACS, after adjusting for other potential confounders. Table 2 displays the effect sizes (HR) and 95% CIs. For 30-day all-cause mortality, in an unadjusted model, the HR (95% CI) of SII <69.4 and SII >88.8 were 1.25 (1.04, 1.50) and 1.38 (1.15, 1.65), respectively, compared with the reference group. With SII >88.8, this association remained significant after adjustment for age, sex, race (white, black, other), BMI (normal weight, overweight, obese), heart rate, SPO2, hemoglobin, lymphocytes, leucocytes, neutrophils, serum glucose, chronic conditions (hypertension, CHF, DM CHD, and alcohol abuse), and scores for the GCS, SOFA, and Elixhauser-30 (HR 1.27; 95% CI 1.06, 1.52; P=0.0107; Table 2). Similar relationships were observed for 90-day and 1-year all-cause mortality.
Association between SII and mortality in patients with ACS.
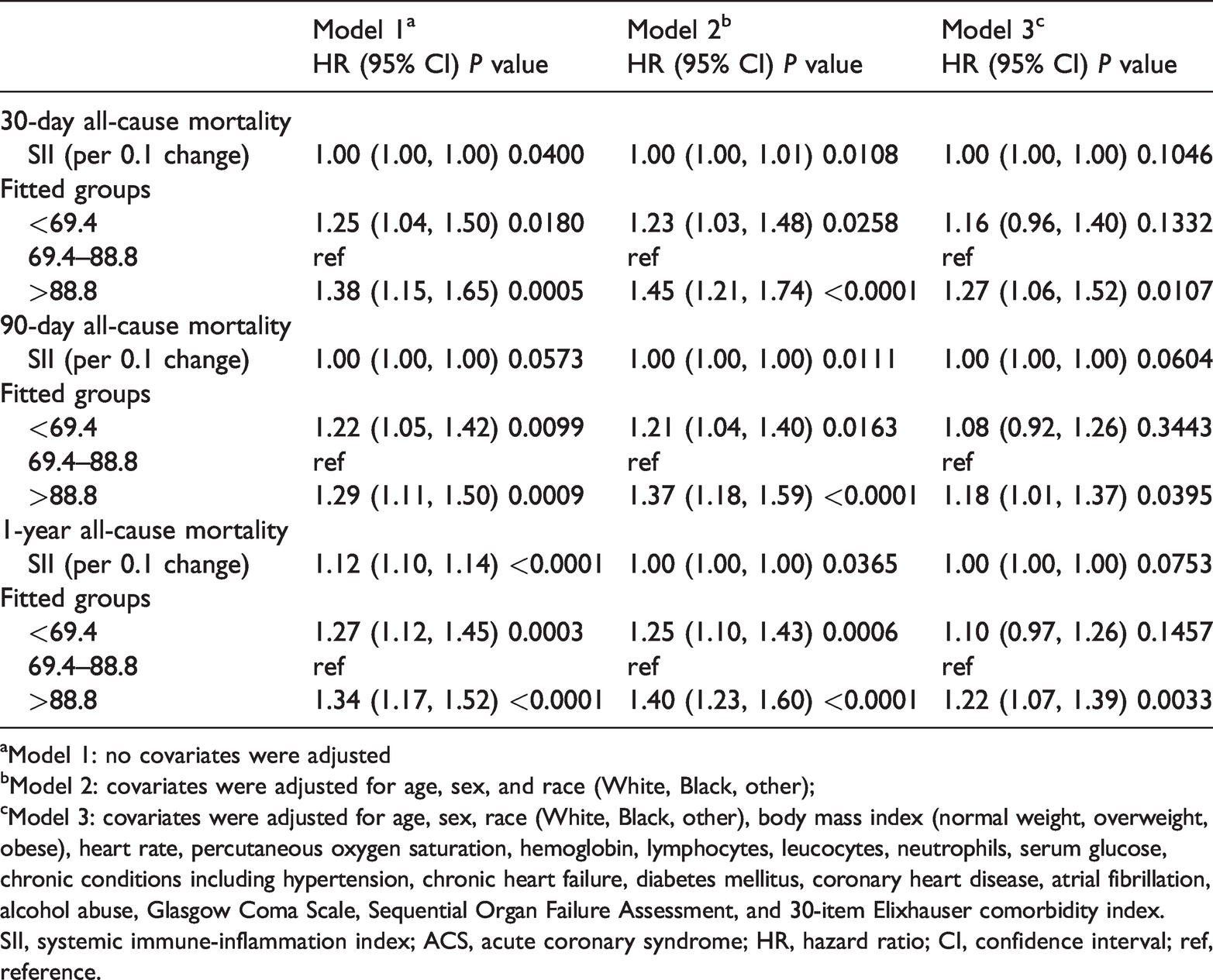
aModel 1: no covariates were adjusted
bModel 2: covariates were adjusted for age, sex, and race (White, Black, other);
cModel 3: covariates were adjusted for age, sex, race (White, Black, other), body mass index (normal weight, overweight, obese), heart rate, percutaneous oxygen saturation, hemoglobin, lymphocytes, leucocytes, neutrophils, serum glucose, chronic conditions including hypertension, chronic heart failure, diabetes mellitus, coronary heart disease, atrial fibrillation, alcohol abuse, Glasgow Coma Scale, Sequential Organ Failure Assessment, and 30-item Elixhauser comorbidity index.
SII, systemic immune-inflammation index; ACS, acute coronary syndrome; HR, hazard ratio; CI, confidence interval; ref, reference.
To assess nonlinearity of the SII and 30-day all-cause mortality in patients with ACS, we performed smooth curve fitting using the penalized spline method (Figure 1). After adjusting for covariates, nonlinear relationships were observed. Because of the limitations of classification analysis, a two-piecewise linear regression model was established, and the inflection point of the SII was 69.33. To the left of the inflection point (SII ≤ 69.33), the HR (95% CI) was 0.98 (0.97, 0.99; P<0.0001). To the right of the inflection point, there was a correlation between the SII and outcome of ACS (HR 1.03, 95% CI 1.02, 1.04, P<0.001; Table 3). For 90-day and 1-year all-cause mortality, we also performed smooth curve fitting and the same relationship was observed.

Correlation between systemic immune-inflammation index (SII) and 1-year all-cause mortality, using the penalized spline method.
Threshold and saturation effect analysis of SII in patients with ACS.

aAdjusted for age, sex, and race (White, Black, other)
SII, systemic immune-inflammation index; ACS, acute coronary syndrome; HR, hazard ratio; CI, confidence interval; NLR, neutrophil–lymphocyte ratio.
Discussion
To the best of our knowledge, ours is the first study to explore the relationship between the SII and short-term and long-term mortality in patients with ACS. The SII is a promising prognostic biomarker, based on neutrophil, lymphocyte, and platelet counts. Our results indicated that the SII was associated with short- and long-term mortality in patients with ACS. After adjusting for potential confounding factors, the association remained. We observed a U-shaped relationship between SII levels and logit-transformed short- and long-term mortality in patients with ACS.
Chronic systemic inflammation promotes cardiovascular disease (CVD) by inducing the expression of endothelial adhesion molecules and chemokines; these in turn promote leukocyte adherence and infiltration. 23 Studies suggest that the SII has high prognostic value in patients with cancer.24,,25 The SII level is helpful in demonstrating inflammation activation and could serve as a reproducible biomarker of systemic inflammatory activity. 26
The accumulation of metabolites and tissue cell fragments during ischemia can trigger acute inflammatory reactions. Evidence is accumulating that apart from the inflammatory function of neutrophils, they also promote intravascular thrombus formation. Additionally, lymphocytes are involved in the immune response as an important subtype of the leucocyte family. Many studies have found a negative association between lymphocytes and outcomes of CVD. Furthermore, platelets are involved in the early stage of atherosclerosis. Activated platelets increase leucocyte activity via direct receptor–ligand interactions, which increases the inflammatory burden inside atherosclerotic plaques.
In short, the pathogenesis of ACS is associated with ischemia-reperfusion injury and systemic inflammatory responses. On the basis of these theories, it appears appropriate to consider the SII an indicator that comprehensively reflects disease characteristics in patients with ACS.
Strengths and limitations
There are several strengths in the present study. We included a large cohort of patients, and we used multiple regression to adjust for several potential confounders, without producing problems of overfitting. The GAM model was used to assess the nonlinear relationship between the SII and outcomes of patients with ACS. A saturation effect was also found.
There are also some study limitations to consider. First, because of the retrospective observational study design, causality cannot be established. Prospective studies are needed to resolve this issue. Second, data were extracted from one blood test only. Third, the SII is easy to obtain in clinical practice; nevertheless, the MIMIC-III database lacked neutrophil, lymphocyte, and platelet counts, which may have led to selection bias.
Conclusion
We provided the first evidence that SII elevation is associated with increased risk of short- and long-term mortality in patients with ACS. This finding should be confirmed in prospective studies.
