Abstract
Objective
To analyse the frequency and characteristics of the Janus kinase 2 (JAK2) V617F mutation in patients with cerebral venous sinus thrombosis (CVST) with thrombocytosis.
Methods
The study enrolled CVST patients with thrombocytosis that had undergone JAK2 V617F mutation detection to determine the frequency of the JAK2 V617F mutation in this cohort. Correlations between patient demographics, whole blood cell counts, targeted sequencing results and JAK2 V617F mutation status were determined.
Results
A total of 23 patients were enrolled in the study: 11 (47.8%) with the JAK2 V617F mutation and 12 (52.2%) without the JAK2 V617F mutation. The mean platelet count was significantly higher in patients with the JAK2 V617F mutation than in patients without the mutation (478.1 ± 107.4 × 109/l versus 374.4 ± 54.1 × 109/l, respectively). There were no significant differences in age, sex, white blood cell count or haemoglobin level between the two groups. Other than single nucleotide polymorphisms, no hot-spot mutations associated with myeloid tumours other than the JAK2 V617F mutation were detected in four CVST patients that underwent targeted sequencing.
Conclusion
The JAK2 V617F mutation was frequently detected in CVST patients with thrombocytosis and it was associated with higher platelet counts.
Introduction
Cerebral venous sinus thrombosis (CVST) is a rare cerebrovascular disease that can afflict young people. 1 Hereditary or acquired thrombogenic diseases are one of the primary causes of CVST in younger patients. 2 Substitution of a valine residue with a phenylalanine residue at position 617 in the Janus kinase 2 (JAK2) gene, resulting in the JAK2 V617F mutation, is among the most frequently detected mutations in blood cells during aging.3,4This mutation triggers constitutive activation of the JAK2 gene, which causes aberrant engagement of downstream signalling pathways and results in excessive proliferation of myeloid cells (e.g. erythrocytosis or thrombocytosis).3,4 Hence, the JAK2 V617F mutation is considered a high-risk factor for arteriovenous thrombosis. 5 Research suggests that the JAK2 V617F mutation might exist in patients with CVST. 6 However, the frequency and characteristics of the JAK2 V617F transformation in CVST patients with thrombocytosis remain unclear.
This current study analysed the frequency of the JAK2 V617F mutation in CVST patients with thrombocytosis and investigated the relationships between this mutation and other patient characteristics, including age, sex, white blood cell count, haemoglobin level, platelet count and co-occurrence of other mutations, to provide a theoretical basis for the aetiological study of CVST patients.
Patients and methods
Study population
This study enrolled patients with CVST with thrombocytosis that had undergone JAK2 V617F mutation detection in the Department of Haematology, Xuanwu Hospital, Capital Medical University, Beijing, China between August 2018 and August 2020. The inclusion criteria were as follows: (i) patients were diagnosed with CVST; (ii) patients had thrombocytosis of unknown cause with platelet counts >300 × 109/l. The exclusion criterion was as follows: (i) secondary thrombocytosis of known cause.
This study was approved by the Institutional Review Board of Xuanwu Hospital, Capital Medical University, Beijing, China. Written informed consent was obtained from the patients or the patient’s parent, carer or legal guardian.
Real-time quantitative PCR and targeted next-generation sequencing analyses
Real-time quantitative polymerase chain reaction (RT–qPCR) targeting the JAK2 V617F mutation and/or targeted next-generation sequencing was performed for all patients. Samples of peripheral blood collected in tubes containing ethylenediaminetetra-acetic acid or bone marrow samples were provided by all patients. DNA was extracted from the samples using a rapid genomic DNA extraction kit for whole blood according to the manufacturer's instructions (DP1802; BioTeke Corporation, Beijing, China) and stored at –20°C. For the RT–qPCR, a commercial JAK2 V617F mutation detection kit (Yuanqi Ltd., Shanghai, China) was used to determine the JAK2 V617F mutation status using a quantitative real-time PCR cycler (Rotor-Gene Q; QIAGEN, Hilden, Germany). The cycling programme was as follows: 42°C for 5 min and 94°C for 3 min, followed by 45 cycles of 94°C for 15 s and 60°C for 60 s. Fluorescence signals were collected at 60°C in the second step of the PCR cycle.
Targeted next-generation sequencing was used to determine variations in DNA sequences from patients based on a panel of 34 genes commonly involved in myeloid neoplasms: ASXL1, BCOR, BCORL1, CALR, CBL, CSF3R, IDH1, IDH2, JAK2, KRAS, MPL, NRAS, SF3B1, SH2B3, SRSF2, TET2, TP53, U2AF1, EZH2, SETBP1, ETV6, DNMT3A, ZRSR2, PHF6, FLT3, RUNX1, CEBPA, PIGA, WT1, PDGFRA, KIT, NPM1, GATA2 and KMT2A. A PCR-based method was used to undertake target enrichment (KAPA Library Amplification Kits; Roche Diagnostics, Mannheim, Germany) and the mean sequencing depth was > 1500×. Variants were identified using Samtools software version 1.3 and the variant allele frequency was calculated as mutation reads/coverage reads.7,8
Data analyses
The relationships between JAK2 V617F mutation status, age, sex, white blood cell count, platelet count, haemoglobin level and sequencing results were calculated. The patient cohort was divided into two groups based on JAK2 V617F mutation status: a mutation and a non-mutation group. Differences in age, white blood cell count, haemoglobin level and platelet count between patients with and without the JAK2 V617F mutation were determined. According to standards and guidelines for the interpretation and reporting of sequence variants in cancer, sequence variants were divided into four categories: strong clinical significance, potential clinical significance, unknown clinical significance and benign or likely benign variants. 9
Statistical analyses
All statistical analyses were performed using GraphPad Prism 8 (Graphpad Software Inc., San Diego, CA, USA). Continuous data are presented as mean ± SD and were compared using Student’s t-test. Categorical data are presented as n of patients (%) and compared using χ2-test and Fisher’s exact test. A P-value < 0.05 were considered statistically significant.
Results
This study enrolled 23 patients with CVST with thrombocytosis: 11 (47.8%) with the JAK2 V617F mutation (mutation group) and 12 (52.2%) without the JAK2 V617F mutation (non-mutation group). Of the 11 patients with the JAK2 V617F mutation, four were male and seven were female (Table 1). Of the 12 patients without the JAK2 V617F mutation, three were male and nine were female. The mean ± SD ages of onset in the mutation and non-mutation groups were 37.18 ± 15.84 years and 27.50 ± 8.94 years, respectively. There were no significant differences between the age or sex distribution between the two groups (Figures 1a and 1b).
Demographic and haematological characteristics of patients (n = 11) with cerebral venous sinus thrombosis with thrombocytosis that had the Janus kinase 2 (JAK2) V617F mutation.

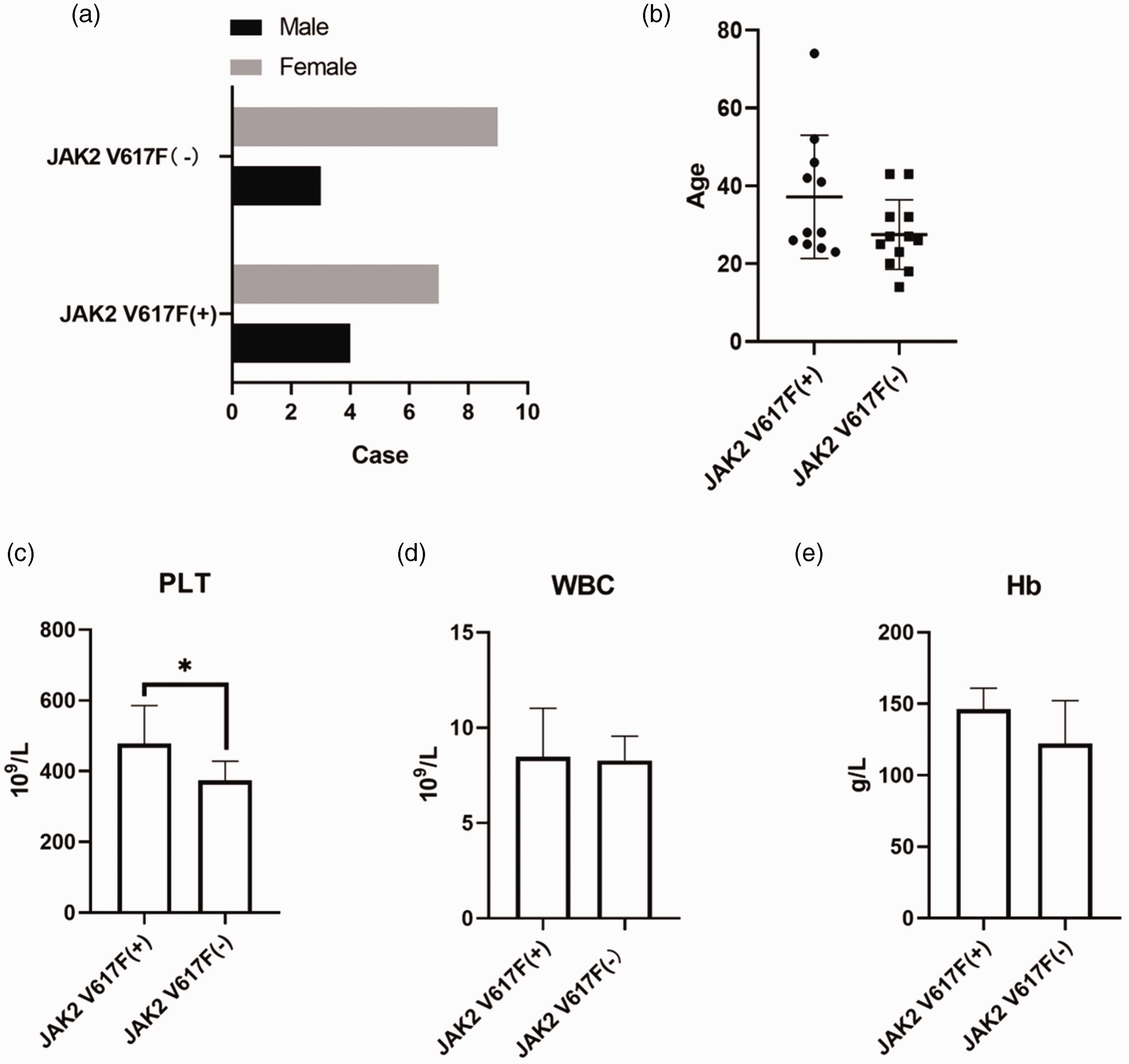
Comparison of the demographic and haematological characteristics of patients with cerebral venous sinus thrombosis with or without the Janus kinase 2 (JAK2) V617F mutation: (a) sex distribution; (b) age distribution with the central black horizontal lines being the mean and the error bars being the SD; (c) platelet count (PLT); (d) white blood cell (WBC) count; (e) haemoglobin (Hb) levels. For C, D and E, data are presented as mean ± SD. JAK2 V617F(–), without the mutation; JAK2 V617F(+), with the mutation. *P < 0.05 between-group comparison using Student’s t-test.
Patients with the JAK2 V617F mutation had a significantly higher mean ± SD platelet count than those without the mutation (478.1 ± 107.4 × 109/l versus 374.4 ± 54.1 × 109/l, respectively; P = 0.013; Figure 1c). There were no significant differences in white blood cell counts or haemoglobin levels between the two groups (Figures 1d and 1e).
Sequence variants in four patients with CVST that underwent targeted sequencing are shown in Table 2. With the exception of the JAK V617F mutation, no other hot-spot somatic mutations related to myeloid tumours were detected in these four patients. However, GATA2, TET2, SH2B3 and ASXL1 single nucleotide polymorphisms (SNPs) were observed. All four patients had GATA2 SNPs: rs2335052 (three of four patients) and rs78245253 (one of four patients).
Sequence variants in four patients with cerebral venous sinus thrombosis with thrombocytosis that had the Janus kinase 2 (JAK2) V617F mutation and underwent targeted sequencing.

SNP, single nucleotide polymorphism.
Discussion
This current study reports the frequency of the JAK2 V617F mutation in CVST patients with thrombocytosis. The current study demonstrated that CVST patients with the JAK2 V617F mutation had significantly higher platelet counts compared with those without the JAK2 V617F mutation, but they did not have other hot-spot somatic mutations associated with myeloid tumours. These current results showed that the JAK2 V617F mutation occurs frequently in CVST patients with thrombocytosis, potentially providing a theoretical basis to help guide clinical investigations of the aetiology of thrombocytosis.
Therapies for CVST mainly address the aetiology and symptoms, hence clarification of the aetiology is necessary for the effective clinical management of patients with CVST.10,11 Venous thrombosis occurs under low shear flow and often requires rich fibrin, activated platelets and a large amount of red blood. 12 Risk factors associated with thrombosis complications include advanced age and a history of thrombosis, as well as the traditional cardiovascular and venous thromboembolism risk factors. 12 Hence, for CVST patients with thrombocytosis, it is essential to determine whether the thrombocytosis is primary or reactive. The JAK2 V617F mutation frequently occurs in patients with essential thrombocythaemia and polycythaemia vera and it has become one of the diagnostic criteria for both conditions. 13 Research has reported that the JAK2 V617F mutation in CVST patients could increase the risk of thrombotic complications, 14 although the frequency and characteristics of the JAK2 V617F mutation in these patients were unclear. In this current cohort, approximately half (47.8%) of the CVST patients with thrombocytosis had the JAK2 V617F mutation, suggesting that this mutation is prevalent among these patients. To the best of my knowledge, this is the first report of the frequency of the JAK2 V617F mutation in CVST patients with thrombocytosis. These current results indicate that attention should be paid to the possibility of myeloproliferative neoplasms in CVST patients. In addition to the mutation being a risk factor for CVST, determining a patient’s JAK2 V617F mutation status may improve the visual prognosis of young patients when there is sagittal sinus or sigmoid sinus involvement, thrombocytosis or hyper haemoglobin present. 6
To further explore the characteristics of the JAK2 V617F mutation in CVST patients, the current study analysed the relationship between the JAK2 V617F mutation status and age, sex, white blood cell count, haemoglobin level and platelet count. The mean platelet count of patients with the JAK2 V617F mutation was 478 ×109/l, while that of the CVST patients without the JAK2 V617F mutation was lower than 400 × 109/l. However, the JAK2 V617F mutation status was not significantly correlated with white blood cell count or the haemoglobin level. Previous research has found that the JAK2 V617F mutation can lead to increased platelet counts and abnormal platelet function, 15 which may explain why CVST patients with the JAK2 V617F mutation have a higher platelet count. Hence, in clinical practice, physicians should be alert to CVST patients with higher platelet counts (> 400 × 109/l). Early detection of the JAK2 V617F mutation may facilitate the clinical management of these patients. It is noteworthy that the JAK2 V617F mutation is not the only molecular genetic change in essential thrombocythaemia and polycythaemia vera, so other known or unknown gene mutations should also be considered. 16
Previous research has found that additional mutations exist in patients with myeloproliferative neoplasms (MPN), 17 although no additional somatic mutations were detected in a subset of four patients with the JAK2 V617F mutation in the current study. Possible explanations for this discrepancy are: (i) the mean age of the CVST patients was significantly lower than that of non-CVST patients with MPN and there may be a lack of age-related clonal haematopoiesis; 10 (ii) the JAK2 V617F mutation is a phenotype-driven mutation in CVST patients, therefore, other somatic or germline mutations associated with susceptibility may not have been found; (iii) the sample size was limited because CVST is a rare disease and it is difficult to evaluate a large number of CVST patients with thrombocytosis. Although this current study found that all four patients did harbour SNPs in other genes, especially for GATA2 rs2335052, it is difficult to make a generalized conclusion from the limited number of patients in this current cohort. Further studies with large patient cohorts should be performed to further determine the role of the JAK2 V617F mutation in CVST patients with thrombocytosis.
In conclusion, the JAK2 V617F mutation was observed in a large proportion of CVST patients with thrombocytosis. These patients usually had higher platelet counts but lacked other hot-spot somatic mutations. This study may provide a potential theoretical basis for the aetiology and clinical management of CVST patients with thrombocytosis.
