Abstract
Breast cancer is the most diagnosed cancer among women worldwide. Cyclin dependent kinase 4/6 inhibitors (ribociclib, palbociclib, and abemaciclib) modulate endocrine resistance and are widely used treatment for patients with advanced-stage hormone receptor-positive, human epidermal growth factor receptor 2-negative breast cancer. Reports of both venous and arterial thromboembolic events, as a complication of cyclin dependent kinase 4/6 inhibitors, are increasingly recognized, but none involved cerebral venous sinus. We herein report on a 44-year-old female patient who initially presented with an early-stage breast cancer treated with surgery, chemotherapy, radiation therapy and finished 5 years of tamoxifen uneventfully. Eight years after her initial diagnosis, she relapsed with a solitary brain lesion which was resected and treated with radiation therapy, and was then started on aromatase inhibitors. Few months later, she progressed with biopsy-proven cervical and mediastinal lymph node metastasis. She was then switched to fulvestrant and ribociclib; both were well-tolerated. However, few weeks later she presented with increasing headache and mild dizziness. Imaging studies showed right lateral sinus acute non-occlusive thrombosis with no parenchymal changes. Patient was anticoagulated with low molecular weight heparin and follow-up visits showed stable disease with no bleeding.
Introduction
Breast cancer continues to be the most commonly diagnosed cancer worldwide. 1 Cancer patients, in general, and those with advanced-stage disease, in particular, are at higher risk for thromboembolic events (TEE); both venous and arterial.2,3 The tumor itself, especially adenocarcinomas, 4 certain chemotherapeutic agents, 5 and hormonal therapy, 6 in addition to major surgical interventions contribute most to this risk. 7 Additionally, patient-related factors including advanced age, smoking, obesity and underlying comorbidities may enhance this risk, too. 8 TEE represent the second most common cause of death in cancer patients and those who survive the event may have worse outcome compared to other cancer patients without TEE. 9
Most of these TEE are venous, in the form of deep vein thrombosis (DVT) and pulmonary embolism (PE). Cerebral venous sinus thrombosis (CVST), however, is relatively rare, but may lead to serious consequences if diagnosis and treatment are delayed. 10 Cancer-related CVST is inadequately addressed, and diagnosis is usually delayed. 11 Obstruction of cerebral venous drainage may lead to increased intracranial pressure, 12 and patients may present with headache, focal neurological deficits such as hemiparesis and hemisensory disturbances, seizures, impaired level of consciousness and papilledema. 13 The superior sagittal sinus, and less commonly the transverse and the sigmoid sinuses are the most affected parts.
The CDK 4/6 (Cyclin Dependent Kinase) Inhibitors are a new class of targeted therapy that have changed the therapeutic landscape of advanced-stage breast cancer. 14 Three such inhibitors; palbociclib, ribociclib, and abemaciclib target the proliferation of tumor cells by inhibiting progression through the cell cycle. These drugs selectively inhibit the binding of CDK4/6 to the ATP-binding pocket in the protein kinases, hence blocking the phosphorylation of Retinoblastoma (Rb) protein. 15 These drugs are combined with AI or fulvestrant, and are particularly used for hormone receptor-positive (HR+), HER2-negative metastatic breast cancer (MBC). 16 CDK 4/6 inhibitors, with aromatase inhibitors (AI) or fulvestrant, have improved progression-free survival, maintained the quality of life, and some have even improved overall survival for patients with MBC.17–19 Nevertheless, their use is associated with several adverse events; many are commonly encountered such as neutropenia, leukopenia, thrombocytopenia, and anemia. 20
A limited number of patients receiving different types of CDK 4/6 inhibitors, reported by major clinical trials like the PALOMAs 21 , the MONALEESA 22 and the MONARCHs, 23 encountered a variety of TEE, including DVT, PE, subclavian vein thrombosis, and vena cava thrombosis. 24 However, CVST was not reported in this setting before.
In this report, we describe the occurrence of CVST in a patient with MBC treated with ribociclib and fulvestrant. Though we cannot directly link the CVST to ribociclib, our aim is to highlight this under-recognized diagnosis to alert physicians to its existence.
After identifying our case, we reviewed all existing meta-analyses, reviews, case reports, and clinical trials on the possible adverse events associated with the use of CDK4/6 inhibitors, plus AI or fulvestrant. The literature review was conducted utilizing PubMed, with the following keywords: CDK4/6 inhibitors, ribociclib, palbociclib, abemaciclib, CVST, thrombosis, breast cancer, and hematological adverse events.
Case presentation
A 44-year-old female patient was diagnosed with an early-stage breast cancer in 2013. She underwent left modified radical mastectomy and axillary dissection; pathology revealed a T1, N2 and grade-2 invasive ductal carcinoma. Estrogen (ER) and progesterone (PR) receptors were both positive, while HER2 was negative. She was treated with adjuvant combination chemotherapy consisting of 5-fluorouracil (FU), epirubicin and cyclophosphamide, followed by docetaxel (T). Following chemotherapy, she was given radiation therapy and was started on tamoxifen. Eight years later, she presented to the emergency department with headache, recurrent vomiting and two episodes of seizures with loss of consciousness. A brain magnetic resonance imaging (MRI) showed a 3.8 cm contrast enhancing mass in the posterior aspect of the right temporal lobe with marked perilesional edema creating a mass effect on the right lateral ventricle (Figure 1). She underwent navigation-guided right occipitotemporal craniotomy and gross total resection of occipitotemporal tumor. The pathology of the extracted tumor was metastatic poorly differentiated adenocarcinoma consistent with the known primary tumor; HR-positive and HER2-negative. Following the surgery, she completed fractionated stereotactic radiotherapy (FSRT) as 30 Gy in 5 fractions to the surgical cavity which was completed in May 2020, and was started on single-agent letrozole. In July 2020, she underwent follow-up brain MRI (8 weeks after resection and FSRT), and showed postoperative sequel with no evidence of residual disease with patent venous sinuses. Three months later, a follow-up CT scan showed bilateral deep cervical and right posterior triangle lymph nodes enlargement with no evidence of local recurrence or distant visceral metastasis. Lymph node biopsy confirmed metastatic carcinoma consistent with the patient’s known primary tumor. She was then started on ribociclib and fulvestrant.
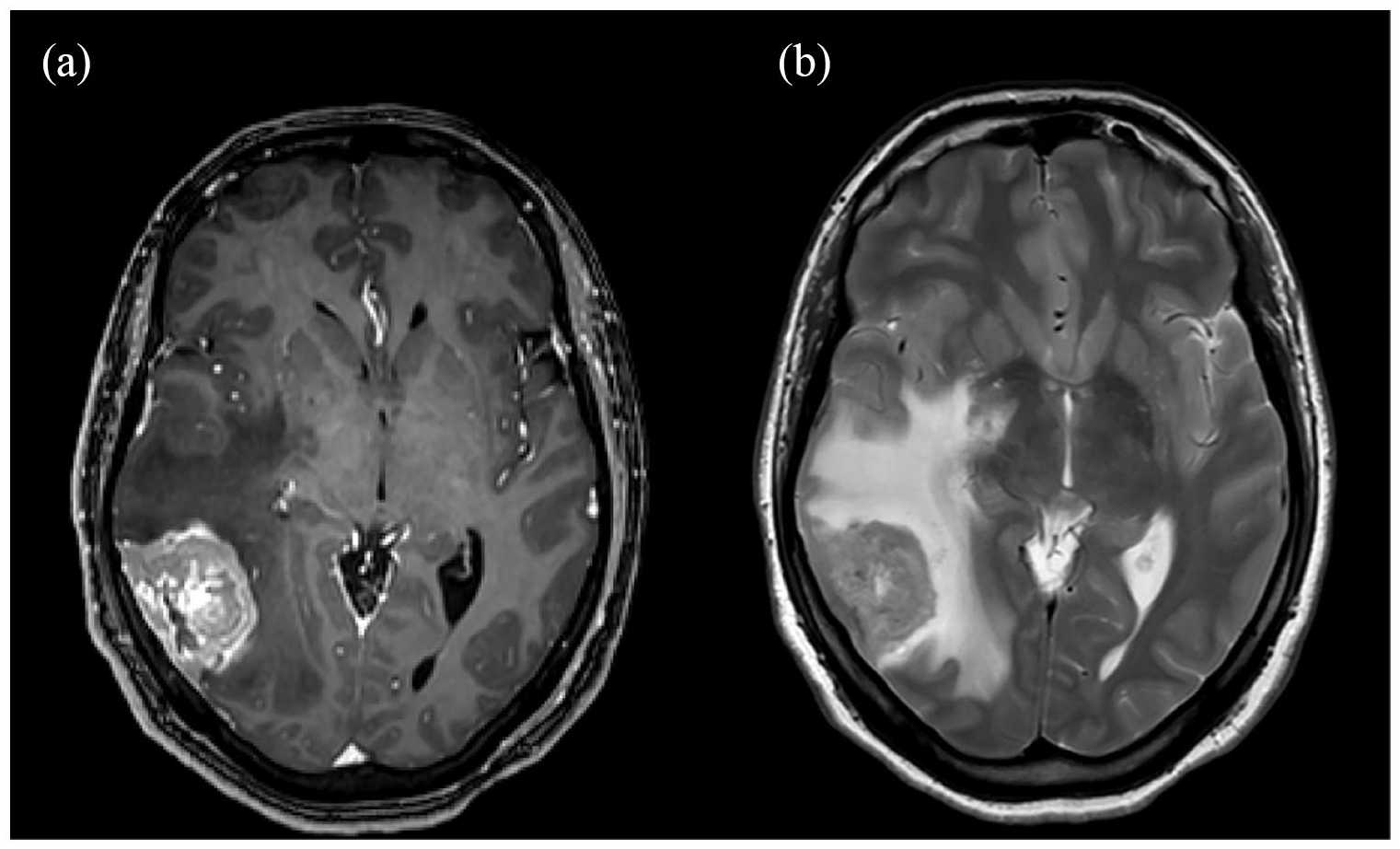
Axial brain MRI T1 (a) and T2WI (b), demonstrate a dural-based heterogeneously enhancing mass lesion in the right temporo-occipital region with marked surrounding vasogenic edema, consistent with brain metastasis.
Another follow-up brain MRI was performed in January 2021 and showed no recurrence of the brain lesion and patent dural venous sinuses without evidence of thrombosis (Figure 2). However, in April 2021, she started to have increasing headache; a subsequent brain MRI showed features of right lateral sinus acute non-occlusive thrombosis with no parenchymal changes (Figures 3 and 4). Laboratory work up, including full chemistry panel and complete blood count were all unremarkable with Hemoglobin of 12.4 g/dL, platelet count of 304 × 109/L and white blood count of 3.8 × 109/L; therefore, she was started on anticoagulation with low molecular weight heparin. Follow-up visits showed stable disease with no bleeding.

(a and b) Axial T1 post-contrast images show patent dural venous sinuses without evidence of thrombosis.

(a and b) Axial T1 precontrast demonstrate hyperintense signal in the right transverse and sigmoid sinuses, and the proximal right internal jugular vein.
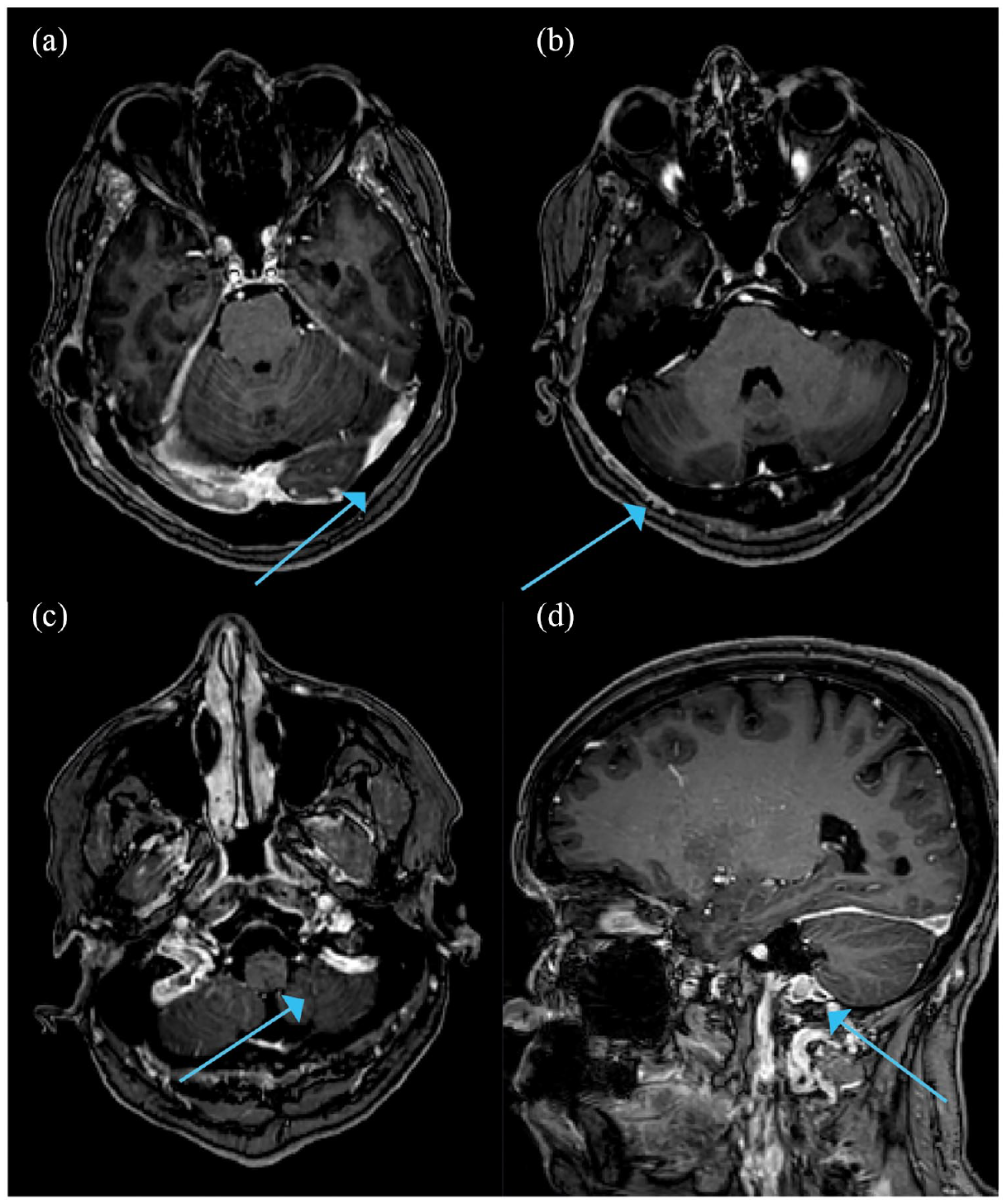
(a–c) axial T1 post-contrast images. (d) Sagittal T1 post-contrast image. There is a non-occlusive filling defect in the right transverse and sigmoid sinuses with extension to the proximal right internal jugular vein, consistent with non-occlusive right lateral sinus thrombosis.
Literature review
The association of cancer with TEE is not debatable. 2 Though breast cancer is not among the known high-risk cancers for TEE, even when patients present with advanced stage disease, endocrine therapy (ET), commonly utilized in HR-positive patients is well known for its thrombotic predisposition. 25 The association of TEE with the newly introduced CDK4/6 inhibitors is not adequately addressed. We had recently reported our own real-world experience using ribociclib in a total of 305 patients, all with metastatic disease; TEE were confirmed on only 6 (1.97%) patients, a rate similar to what we usually encounter with similar patients not on ribociclib. 26 A meta-analysis reviewed the association of CDK4/6 inhibitors with TEE in over 4000 patients enrolled on eight controlled trials, in which patients were randomized to receive ET plus a CDK4/6 inhibitor (palbociclib, ribociclib, or abemaciclib) or ET and placebo; venous TEE were reported in 56 (2%) in the CDK4/6 inhibitor plus ET arm, compared to only 10 (0.5%) in the control arm. Most of the patients who developed venous thromboembolic events were on abemaciclib and not on ribociclib or palbociclib. 27
There is no established clinical evidence confirming the efficacy of CDK4/6i within the central nervous system. In the context of clinical trials, the PALOMA-2 and 3 studies included only seven patients with brain metastases treated with palbociclib and similar number of patients were enrolled on the MONALEESA-3 trial, which was the only ribociclib clinical trial in the MONALEESA program that permitted recruitment of patients with brain metastases. However, none of the MONARCHE 1, 2, or 3 trials enrolled patients with brain metastases. Notably, data from a phase 2 trial indicated an intracranial objective response rate (ORR) of 5.2% and an intracranial clinical benefit rate of 24%. It is worth mentioning that the study did not meet its primary endpoint, which aimed for an intracranial ORR of ⩾15%. 28 In our center, we offer palbociclib for patients with contraindications for ribociclib; however, abemaciclib is not included in our formulary.
CVST comprises 0.5%–1% of all strokes and about 0.3%–4% of the complications in patients with cancer, 29 yet early diagnosis is vital to prevent outcomes like hydrocephalus, intracranial hypertension, and seizures. 30 Another reason why such a complication can go unrecognized is the non-specific clinical presentation of CVST, where it can range from mild headaches to nausea, vomiting, seizures, reduced consciousness and focal neurological deficits or even coma; added to this is the non-specific findings on brain CT scan commonly used when such patients present to emergency departments. When diagnosed, physicians should review other known etiologies causing CVST in cancer patients, like the use of oral contraceptives, puerperium, hypercoagulability, primary or metastatic intracranial tumors obstructing the venous drainage, or even mastoid infections. The association of the three CDK 4/6-inhibitors with CVST was raised as their use may potentially increase the risk of venous thromboembolism. Early and prompt monitoring with adequate follow-up visits to manage the possible side effects remains vital and is consequently the best way to make use of such innovative treatment.
Though CVST is relatively rare, and similarly so with breast cancer patients treated with CDK4/6 inhibitors, our case report should alert physicians about this clinical event that should be differentiated from the more common brain metastasis. Early recognition and early initiation of anticoagulation should help prevent associated complications. 31
Conclusions
With the expanded use of the innovative CDK4/6 inhibitors in patients with advanced-stage breast cancer, physicians should be aware of the rarely encountered thromboembolic event associated with these drugs. Given the rarity of CVST, our case report should be an eye-opener to alert physicians about this complication for timely and appropriate anticoagulation.
Footnotes
Acknowledgements
The authors would like to acknowledge the efforts of our medical photographers, Mr. Ameen Harb and Mr. Ramzi Abu-Khader.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Given the anonymous nature of the described patient and lack of any identifier, our institution does not require ethical approval for reporting of case series.
Informed consent
Written informed consent for publication of clinical details and related images was obtained from the patient.
