Abstract
Objective
To identify serum protein markers in midtrimester that predict preterm delivery.
Methods
A retrospective case–control study randomly selected patients that experienced spontaneous preterm birth and healthy control patients that experienced a normal delivery at term. A proteomic analysis was undertaken using the data-independent acquisition method.
Results
A total of 30 singleton pregnant women were randomly selected from 12 800 pregnant women: 15 women had a spontaneous preterm birth (group Y) and 15 age- and body mass index-matched women gave birth at term (group D). All of the patients provided serum at 15–20 weeks of gestation. A total of 39 differentially expressed proteins were identified. Compared with group D, 24 proteins were upregulated and 15 were downregulated in the preterm group Y. Using Kyoto Encyclopedia of Genes and Genomes pathway enrichment, the 24 upregulated proteins were significantly enriched in the complement and coagulation cascade pathways. Search Tool for the Retrieval of Interacting Genes Furthermore (STRING) analysis showed that apolipoprotein A-II (apoA-II) and alpha-2-antiplasmin (α2-AP), two upregulated proteins, were key nodes in the STRING protein–protein network.
Conclusions
These findings suggest that apoA-II and α2-AP might be new markers for predicting preterm delivery in the midtrimester.
Introduction
In obstetrics, delivery before 37 weeks is defined as a premature birth. 1 The incidence of preterm birth is 11% worldwide. 2 In 2014, the preterm birth rate was 6.9% in China. 3 Preterm birth is a major cause of perinatal death. 4 Preterm delivery is a high-risk factor for postpartum depression. 4 Although cervical tube length measurement and fibronectin screening have been widely used, they still cannot predict premature delivery accurately, and the perinatal outcome has not been improved. 5 A Chinese study has shown that exposure to traffic fumes and environmental factors during pregnancy, especially early pregnancy, increases the risk of premature birth. 6 The aetiology of preterm birth is complex, with it being caused by the interaction between environmental, genetic and individual factors. 6 A previous study showed that increased levels of hypoxia inducible factor-1 α, ferritin and interleukin-6 could be used as biomarkers for preterm delivery. 7 Placental α 1-microglobulin and fetal fibronectin tests were used to predict preterm delivery. 8 Cervical α-actinin-4 increased in patients with preterm delivery. 9 However, none of these markers can predict premature delivery in the early stage of pregnancy. Therefore, this study aimed to identify serum markers that can predict preterm delivery at an earlier stage of pregnancy.
Patients and methods
Study population
This retrospective case–control study randomly selected singleton pregnant women from all of the pregnant women that delivered in the Department of Obstetrics, Hangzhou Women’s Hospital, Hangzhou, Zhejiang Province, China between January 2018 and January 2019 using a random number method. The inclusion criteria were as follows: (i) mid-term of pregnancy blood sample; (ii) delivered before 37 weeks; (iii) without high risk factors. The exclusion criteria were as follows: (i) twins and multiple pregnancy; (ii) pregnancy induced hypertension; (iii) preeclampsia; (iv) other medical diseases such as diabetes mellitus; (v) severe heart, liver kidney diseases; (vi) smoking; (vii) assisted pregnancies; (viii) incomplete information. The preterm group (group Y) consisted of women that had experienced a spontaneous preterm delivery that were within the normal range for body mass index (BMI), aged 25–30 years old and had no pregnancy complications. The control group (group D) of healthy pregnant women that delivered at term was randomly selected at the same follow-up visit and were matched to group Y for their age and BMI. All patient data were anonymized.
This study was approved by the Ethics Committee of Hangzhou Women’s Hospital on 5 September 2016 (approval no. [2016] R & D HLS (001) – 07). Written informed consent was obtained from all patients.
Sample collection
All pregnant women in China must have their blood analysed at 15–20 weeks of gestation. Whole blood was collected in a vacuum tube without anticoagulants and then centrifuged at room temperature using a DT5-6B medical centrifuge (Beijing Modern Beili Centrifuge Co., Ltd., Beijing, China) at 1000 g for 10 min to prepare 3–5 ml of serum. The serum samples were stored at –80°C until use. Before use, the serum was thawed and underwent the following processes before it could be used for data-independent acquisition (DIA) analysis: denaturation, reduction, alkylation, termination of alkylation, LysC enzymolysis, urea dilution and trypsin enzymolysis, acidification, desalination and freeze-drying. The following reagents were used: UltraPure™ Tris hydrochloride (Invitrogen, Carlsbad, CA, USA); ammonium bicarbonate (Sigma-Aldrich, St Louis, MO, USA); trifluoroacetic acid (Sigma-Aldrich); dithiothreitol (Sigma-Aldrich); iodoacetamide (Sigma-Aldrich); lysyl endopeptidase, MS grade (Wako Pure Chemical Industries, Osaka, Japan); sequencing grade modified trypsin (Promega, Madison, WI, USA); SOLAμ HRP 96 well plate (Thermo Fisher Scientific, Rockford, IL, USA).
Proteomics by data-independent acquisition
Data-independent acquisition quantitative proteomics analysis was used to identify differentially expressed proteins between the two groups of patients. The analysis was undertaken using an ultra-high performance liquid chromatography system (Thermo Scientific™ UltiMate™ 3000 RSLC Nano System; Thermo Fisher Scientific) in tandem with Orbitrap Fusion Lumos™ quadrupole-linear ion trap-electrostatic field orbital well high resolution mass spectrometry (Thermo Fisher Scientific) and data acquisition software (Xcalibur™ Software version 4.3; Thermo Fisher Scientific). The chromatographic separation conditions were as follows: high performance liquid chromatography; double column mode: trap column, analysis column; mobile phase: A: 0.1% formic acid water, B: 0.1% formic acid acetonitrile; gradient: 3%–8% B in 6 min, 8%–30% B in 102 min, 30%–100% B in 8 min, 100%–100% B in 4 min; flow rate: 300 nl/min. The parameters of the mass spectrometry were as follows: spray voltage: 2.1 KV; capillary temperature: 300°C; S-lens: 50%; impact energy: 32% high energy collision dissociation; scanning range of parent ion: 350–1200 M/Z; scanning range of sub ion: start from M/Z 200. Gene Ontology (GO) annotation and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway were used to analyse protein enrichment and function. Protein–protein interaction networks were investigated using the Search Tool for the Retrieval of Interacting Genes (STRING) database (http://string-db.org). STRING diagrams were used to illustrate the relationships between differentially expressed proteins.
Statistical analyses
The data collected by DIA were processed using Spectromant X software (Spectronaut 13; Biognosys AG, Schlieren, Switzerland). The protein signal strength information calculated by the Spectromant X software was imported into the Perseus software platform for graphical processing and statistical analysis (Max-Planck-Institute of Biochemistry, Martinsried, Germany). The comparison between the two patient groups was performed using a Student’s t-test. A P-value < 0.05 was considered statistically significant. The list of differentially expressed proteins found by the statistical analysis was imported into protein GO and KEGG function annotation (Thermo Fisher Scientific).
Results
This retrospective case–control study randomly selected 30 singleton pregnant women from 12 800 pregnant women that delivered between January 2018 and January 2019: 15 women that had a spontaneous preterm birth formed group Y and 15 age- and BMI-matched women that gave birth at term formed group D.
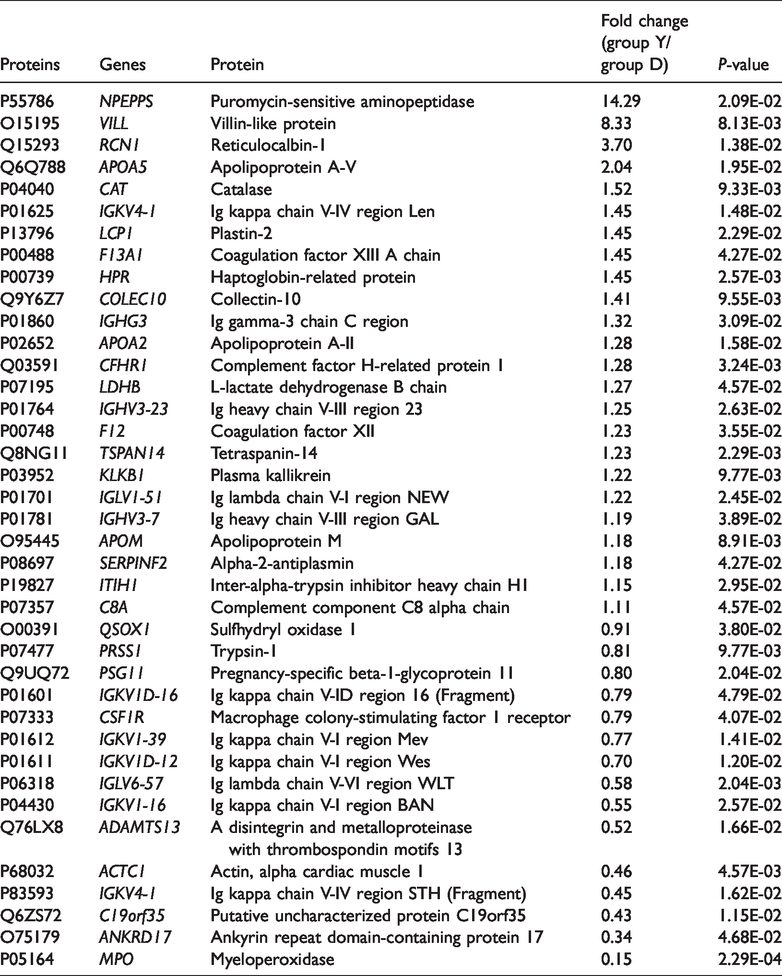
A total of 460 proteins were quantified. A volcano plot shows that 24 proteins were upregulated and 15 proteins were downregulated in the preterm delivery group Y compared with the healthy control group D (Figure 1). A total of 39 differentially expressed proteins were screened, including serum apolipoprotein A-II (apoA-II) and alpha-2-antiplasmin (α2-AP), pregnancy-specific beta-1-glycoprotein 11 and other important pregnancy-associated proteins (Table 1).

Volcano plot showing upregulated proteins (red dots) and downregulated proteins (blue dots) that were identified when comparing the preterm delivery group Y and the healthy control group D. The colour version of this figure is available at: http://imr.sagepub.com.
The characteristics of the differentially expressed proteins (n = 39) identified by comparing the preterm delivery group Y and the healthy control group D.
The KEGG analysis showed that 24 upregulated proteins were significantly enriched in the complement and coagulation cascade pathways (Table 2). Protein–protein interaction networks were investigated using the STRING database (a database of known and predicted protein interactions). It showed that serpin family F member 2 (SERPINF2), coagulation factor XIII A chain (F13A1), coagulation factor XII (F12), plasma kallikrein (KLKB1), complement component C8 alpha chain (C8A) and inter-alpha-trypsin inhibitor heavy chain H1 (ITIH1) were interacting with each other. In addition, apolipoprotein A-II (APOA2), apolipoprotein M (APOM) and apolipoprotein A-V (APOA5) were interacting with each other (Figure 2).
Analysis of upregulated KEGG pathways of differentially expressed genes.

KEGG, Kyoto Encyclopedia of Genes and Genomes.
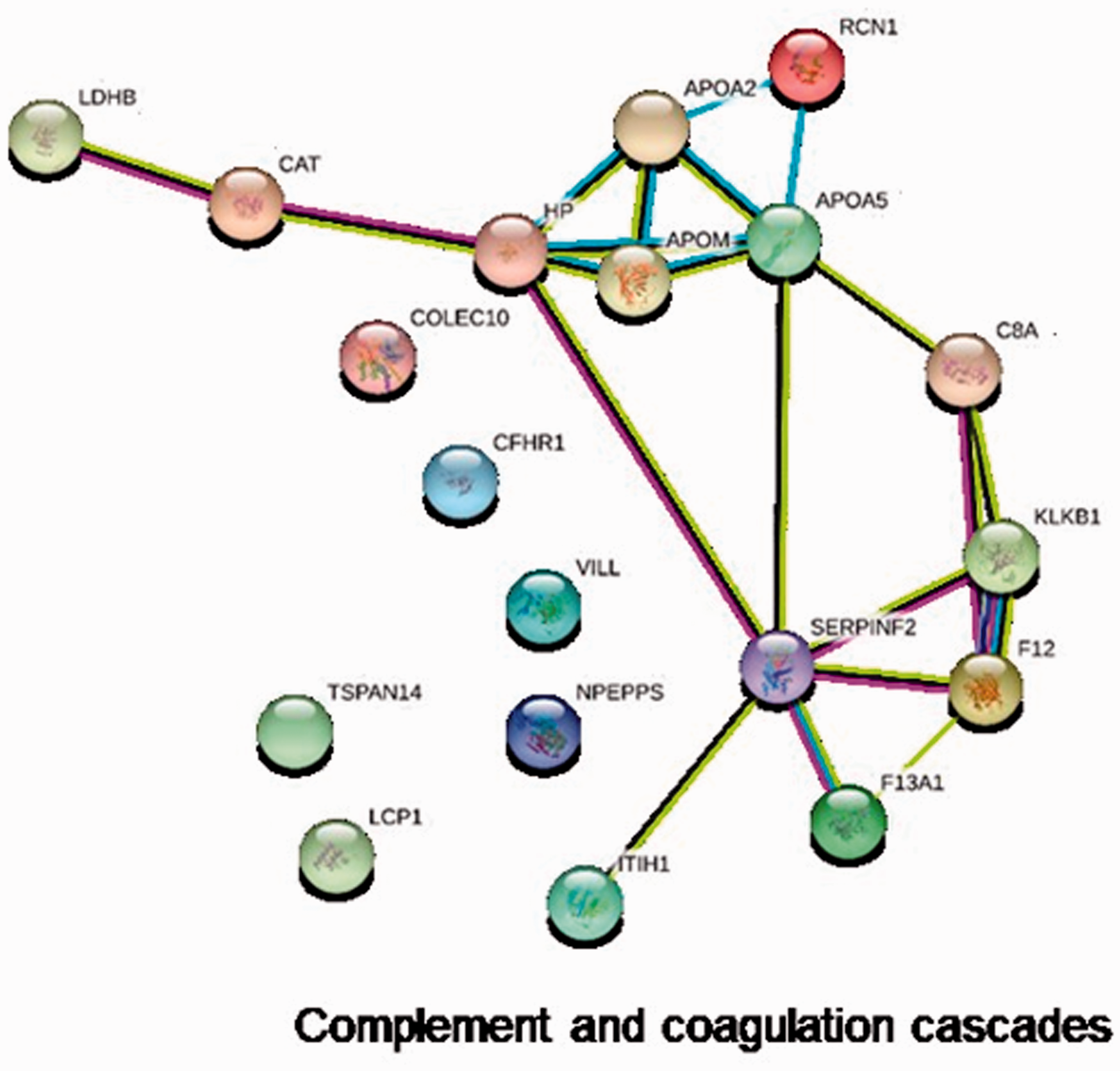
A Search Tool for the Retrieval of Interacting Genes (STRING) diagram showing the protein–protein networks involving the complement and coagulation pathways that were identified by comparing the preterm delivery group Y and the healthy control group D. The colour version of this figure is available at: http://imr.sagepub.com.
Discussion
At present, established research has demonstrated that there were four kinds of pathophysiological mechanisms associated with preterm birth: chorioamnionitis, premature contraction pathway, decidual haemorrhage and susceptibility to environmental toxins. 10 Most spontaneous preterm birth is caused by dominant or subclinical infection of the upper genital tract that results in a host anti-inflammatory response. 11 The destruction of the cervical epithelial barrier and an inflammatory response are important trigger factors for cervical remodelling, which is an important step in preterm delivery. 11 At present, transvaginal cervical tube length measurement is the best method to screen for the risk of premature delivery in pregnancy, but its application is limited, so it is necessary to find better screening indicators. 12
The differentially expressed proteins identified in this present study were mainly from the complement and coagulation cascade pathways. Previous research suggested that the mean platelet volume can predict the early-onset of neonatal sepsis in preterm infants. 13 Another study suggested that mean platelet volume was a biomarker of inflammation. 14 Therefore, it could be inferred that there is a relationship between coagulation mechanisms and the inflammatory response. This was consistent with the results of the current study. When compared with the healthy control group D, the levels of the apoA-II and α2-AP proteins were increased in the preterm group Y. The main physiological changes during pregnancy are inflammation and hyperlipidaemia. 15 Maternal triglyceride and cholesterol levels increase during pregnancy, indicating that both are particularly important for pregnancy and fetal growth. 16 Apolipoproteins are composed of a variety of triglyceride transporters, which can cause hyperlipidaemia in pregnancy by regulating maternal lipid metabolism. 16 High blood lipid levels are related to inflammation and obstetric complications. 16 The relative levels of apoA-II increased slightly with pregnancy duration. 15 The modified apoA-II subtype was significantly higher in the preterm group than in the term group (P = 0.02). 15 It is believed that apoA-II plays an important role in the synthesis, composition and function of high-density lipoprotein cholesterol (HDL-C). 17 An in vitro study demonstrated that apoA-II could form a dimer with apoE, which then affected the ability of ApoE to bind to HDL-C molecules. 17 The relationship between the apoE genotypes of mothers and small for gestational age infants suggests that low maternal circulating cholesterol levels reduce fetal growth. 18 Leptin is a typical adipokine secreted by adipocytes and research has shown that the leptin level is decreased in preterm infants. 19 All of these findings suggest that lipid metabolism is involved in the pathogenesis of preterm birth. 20 The STRING diagram (Figure 2) shows that apoA-II interacts with plasma kallikrein, coagulation factor XIII A chain, coagulation factor XII, complement component C8 alpha chain and other important coagulation and immune factors involved in preterm birth. Therefore, considering that apoA-II is closely associated with preterm birth and was increased in the preterm group Y, it may be a biomarker for predicting preterm birth.
The enzyme inhibitor α2-AP is a member of the serpin family and it is synthesized in the human liver. 21 The gene that encodes α2-AP, SERPINF2, is located on chromosome 17. 22 α2-AP is one of the fibrinolysis inhibitors. 23 Patients with decreased α2-AP are more likely to bleed. 24 Hyperfibrinolytic diseases are related to a high incidence of obstetric complications such as abortion and premature delivery. 24 A previous study showed that in patients with preeclampsia, due to the imbalance of oestrogen and testosterone levels, α2-AP secretion was increased. 25 The plasmin inhibitors are α2-AP and α 2-macroglobulin (α2-m). 23 α2-AP is the main physiological inhibitor of fibrinolytic enzymes. 23 When the formation of plasmin is excessive relative to α2-AP, the plasmin is neutralized by α2-m. 23 This defect can lead to the presence of a high level of fibrinolytic enzymes, resulting in high fibrinolysis and bleeding. 24 However, a study of patients with acquired α2-AP deficiency shows that clinical hyperfibrinolysis only occurs at the level of plasma α2-AP lower than 60%. 26 It is not clear whether women with a heterozygous deficiency of α2-AP have a high incidence of obstetric complications. 24 It has been found that α2-AP can induce macrophages to produce proinflammatory cytokines through extracellular signal regulated kinase1/2 and c-Jun N-terminal kinase pathways. 27 This current study demonstrated that α2-AP was upregulated in the preterm group Y compared with the healthy control group D. These findings suggest that there might be a correlation between the upregulation of α2-AP and the incidence of preterm birth, but the specific mechanism requires further research to determine if α2-AP is a predictive marker of preterm birth.
This current study had several limitations. First, this was a retrospective study. Secondly, the number of patients in each group was limited. Larger prospective studies are needed in the future.
In conclusion, apoA-II and α2-AP serum protein levels were upregulated in the preterm group midtrimester compared with the healthy control group. Differentially expressed genes were significantly enriched in the complement and coagulation cascade pathways. In addition, apoA-II and α2-AP were key nodes in the STRING protein–protein network, interacting with other proteins that might be associated with preterm birth. Following further larger prospective studies, apoA-II and α2-AP might become new markers for predicting preterm delivery in the midtrimester, which might facilitate the prevention of preterm birth.
Footnotes
Acknowledgements
We thank all members of the research team for their hard work.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study received a grant from the Zhejiang Medical and Health Research and Technology Project (no. 2017KY55).
