Abstract
Objective
We examined whether antiplatelet therapy is safe for ischemic stroke patients with cerebral microbleed.
Methods
We retrospectively analyzed ischemic stroke patients admitted to our hospital from 2015 to 2018. Baseline information was extracted from the computerized database. Adverse events, including symptomatic cerebral hemorrhage, recurrent cerebral infarction, and death, were collected by phone.
Results
A total of 184 ischemic stroke patients were examined, including 106 with and 78 without cerebral microbleed. No patient experienced symptomatic cerebral hemorrhage after discharge. Patients with cerebral microbleed had a higher prevalence of hypertension (92% vs 74%) and suffered from more serious leukoaraiosis (3.0 ± 1.7 vs 1.3 ± 1.4 points on the Fazekas scale). Leukoaraiosis scores were correlated with the number of cerebral microbleeds (r = 0.42).
Conclusions
Antiplatelet therapy may be safe for ischemic stroke patients with cerebral microbleed. The risk-benefit ratio should be carefully evaluated before withholding antiplatelet therapy.
Introduction
A cerebral microbleed (CMB) is defined as a 2- to 10-mm focus of hypointensity that can be visualized as a round lesion on susceptibility-weighted imaging (SWI). 1 , 2 The prevalence of CMB is 40% to 57% in ischemic stroke patients. 3 Antiplatelet therapy plays an important role in secondary prevention of ischemic stroke. However, for ischemic stroke patients with CMB, use of antiplatelet therapy still remains controversial in clinical practice, resulting in a debate on its safety and benefit.
Previous studies have revealed that a CMB may increase the risk of cerebral hemorrhage during antiplatelet therapy. 4 , 5 These studies noted a trend toward a higher risk of future cerebral hemorrhage in patients with a greater number of CMBs and concluded that antiplatelet therapy may not be safe and should not be recommended for ischemic stroke patients with CMB. However, some studies have shown that CMB is not associated with the risk of cerebral hemorrhage and concluded that antiplatelet therapy should be recommended for ischemic stroke patients with CMB. 6 , 7 Therefore, the use of antiplatelet therapy is still a clinical dilemma for these patients.
Approximately 8% to 11% of patients experience recurrent cerebral infarction at the early stage of stroke. 8 , 9 More worryingly, the outcome of recurrent cerebral infarction is much worse than the symptoms at first onset. Because of this dilemma, additional research is urgently needed to determine whether antiplatelet therapy should be recommended for ischemic stroke patients with CMB. Therefore, we aim to assess the risk-benefit ratio of antiplatelet therapy in ischemic stroke patients with CMB.
Methods
Patient selection
This retrospective study included ischemic stroke patients admitted to our hospital from 2015 to 2018. Patients were divided into a CMB group and a non-CMB group according to the presence of CMB. The inclusion criteria for the CMB and non-CMB groups were as follows: (a) patients diagnosed with ischemic stroke; (b) magnetic resonance imaging (MRI) examinations were performed within 30 days after stroke onset; (c) patients received antiplatelet therapy (100 mg of aspirin or 75 mg of clopidogrel/day) after discharge; and (d) no complications that may increase the risk of cerebral hemorrhage. Diagnosis of ischemic stroke was based on neurologic decline, physical examination, and radiological results.
All patients underwent MRI examinations on a 1.5 T scanner (GE Healthcare, Piscataway, NJ, USA) during hospitalization. The presence of CMB was identified on SWI according to the following parameters: repetition time: 78 ms, echo time: 50 ms, flip angle: 15°, and slice thickness: 2 mm. Leukoaraiosis was common on MRI in ischemic stroke patients. The severity of leukoaraiosis was classified into three grades based on the total scores assessed using the Fazekas scale: 10 mild (1–2), moderate (3–4), and severe (5–6).
This study was approved by the ethics committee of Guangxi Jiangbin Hospital and was conducted in accordance with the ethical guidelines of the Declaration of Helsinki. Because of the retrospective nature of this study, the requirement for informed consent was waived.
Data collection
Baseline information and MRI data were extracted from a computerized database. For the collection of adverse events, all patients were contacted once by phone in 2019. Patients voluntarily provided information on whether they experienced symptomatic cerebral hemorrhage or recurrent cerebral infarction after discharge. The identification of adverse events was based on disease certificates provided by patients or their relatives.
Statistical analysis
Data analyses were performed using SPSS version 23.0 software (IBM Corp., Armonk, NY, USA). Continuous variables with a normal distribution are expressed as the mean ± standard deviation and were compared using Student t-test. Categorical variables were analyzed by the chi-square test. A value of P<0.05 was considered statistically significant.
Results
Clinical characteristics
A total of 184 patients were included in this study, including 106 in the CMB group and 78 in the non-CMB group (Figure 1). The clinical characteristics of the two groups are summarized in Table 1. Patients with CMB had a higher prevalence of hypertension (92% vs 74%, P<0.05) and suffered from more serious leukoaraiosis (3.0 ± 1.7 vs 1.3 ± 1.4 points, P<0.05).
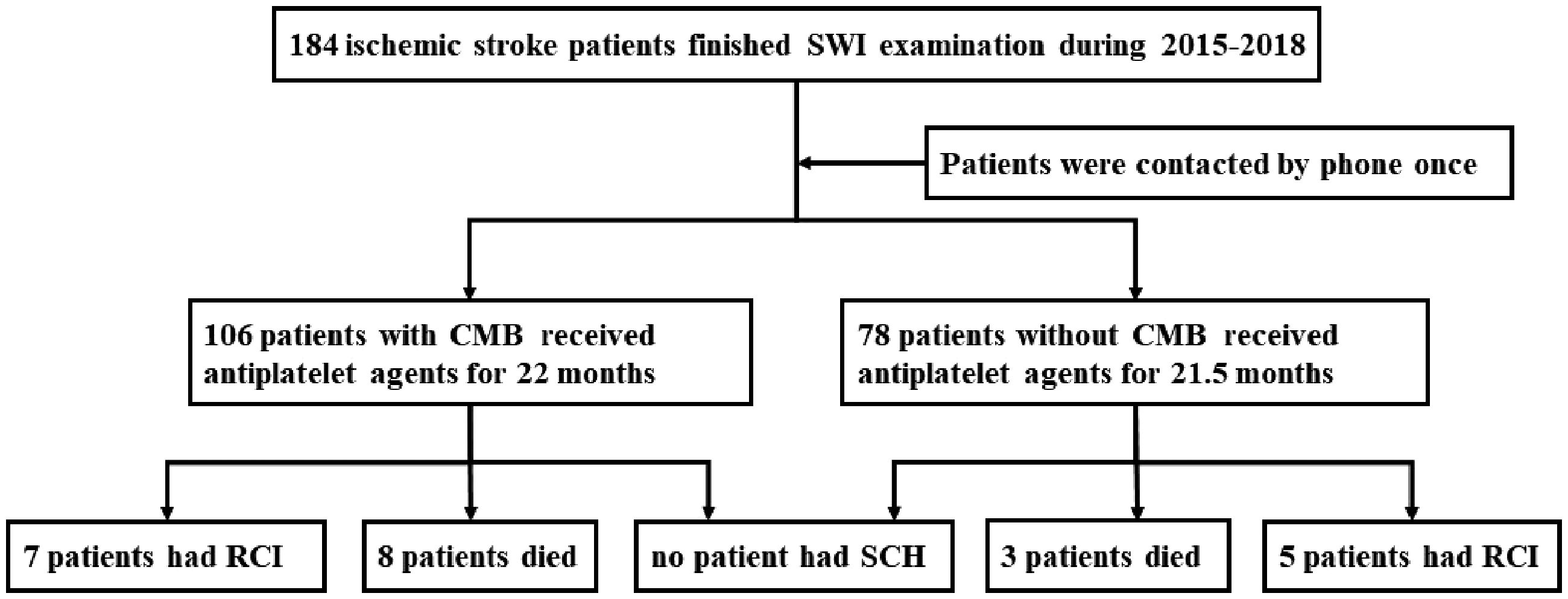
Study flow chart. CMB = cerebral microbleed, RCI = recurrent cerebral infarction, SCH = symptomatic cerebral hemorrhage, SWI = susceptibility-weighted imaging.
Clinical characteristics.

*P<0.05, CMB = cerebral microbleed, n = number of patients, SD = standard deviation.
Number of CMBs
The numbers of CMBs were significantly different in various cerebral regions. The percentages of CMBs in different cerebral regions were as follows: basal ganglia (23.1%), temporal lobe (18.2%), thalamus (12.6%), parietal lobe (10.2%), occipital lobe (9.9%), cerebellum (9.6%), brainstem (9.6%), and frontal lobe (6.8%).
Patients with a greater number of CMBs tended to suffer from more serious leukoaraiosis. A positive correlation was found between number of CMBs and leukoaraiosis scores (r = 0.42, P<0.05) (Figure 2).

Correlation between the number of CMBs and leukoaraiosis scores. CMB = cerebral microbleed, LA = leukoaraiosis.
Adverse events
No patient in the two groups experienced symptomatic cerebral hemorrhage after discharge. However, seven patients had recurrent cerebral infarction and eight patients died in the CMB group. Five patients had recurrent cerebral infarction and three died in the non-CMB group (Figure 1). The morbidity of recurrent cerebral infarction was 6.5% (12/184) and the mortality was 6.0% (11/184) in the two groups.
Discussion
In this study, no patient experienced symptomatic cerebral hemorrhage. However, even when using antiplatelet agents, 6.5% (12/184) of ischemic stroke patients still had recurrent cerebral infarction. In addition, leukoaraiosis scores were positively correlated with the number of CMBs.
Our study revealed that antiplatelet therapy may be safe for ischemic stroke patients with CMB. Although patients were receiving antiplatelet therapy, they still had a high risk of recurrent cerebral infarction during the early stage after stroke. Furthermore, our findings were consistent with previous studies. One study showed that dual antiplatelet therapy was beneficial after endovascular intervention in patients with CMB. 7 Another study revealed that for ischemic stroke patients taking antiplatelet agents, CMB did not increase the risk of cerebral hemorrhage. 11 Even for patients with more than four CMBs, antiplatelet therapy still should be continued in the first year after ischemic stroke. 6 In general, the benefit of antiplatelet therapy may outweigh the risk in ischemic stroke patients with CMB. Therefore, the presence of CMB should not influence the decision to administer antiplatelet therapy without further sufficient evidence.
However, some studies have revealed that antiplatelet therapy is inappropriate in ischemic stroke patients with CMB. One study showed that antiplatelet therapy should not be used in patients with five or more CMBs. 12 Another study found that the risk of cerebral hemorrhage increased greatly in patients with more than 10 CMBs. 13 However, no consensus exists regarding a threshold number of CMBs that may increase the risk of cerebral hemorrhage. Therefore, the risk-benefit ratio should be carefully evaluated before withholding antiplatelet therapy in patients with CMB.
The clinical significance of leukoaraiosis is not entirely clear. Our study revealed that leukoaraiosis may be a predictor of CMB. Some studies have found that leukoaraiosis may increase the risk of stroke events. 14,15 These findings suggest that leukoaraiosis may have potential clinical significance and emphasize the importance of leukoaraiosis prevention.
There are two explanations as to why no patient experienced symptomatic cerebral hemorrhage in this study. First, all patients received antiplatelet therapy but not anticoagulant therapy. Anticoagulants may increase the risk of cerebral hemorrhage. Second, this study had few patients with CMB in brain lobes, which may increase the risk of cerebral hemorrhage. 16
This study had several limitations. Partial data were obtained by phone interviews, and the accuracy depended on the cooperation of respondents. Moreover, because of the relatively small number of patients, the findings may be not fully representative.
Conclusion
Antiplatelet therapy may be safe for ischemic stroke patients with CMB. The risk-benefit ratio should be carefully evaluated before withholding antiplatelet therapy. Further prospective studies should be conducted to confirm these preliminary findings.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81860222 and No. 81960220) and the Science and Technology Plan Project of Guangxi (No. AB16380204).
