Abstract
Objectives:
The incidence of stroke-related restless legs syndrome (RLS) has been reported to be high but varies regionally. Therefore, this study aimed to investigate the incidence and some of the factors related to restless legs syndrome after stroke onset in patients in Vietnam.
Methods:
Data were collected from a total of 423 patients who had stroke, including 283 ischemic and 140 hemorrhagic strokes, which were confirmed by magnetic resonance imaging within 7 days after the onset of symptoms, at the Department of Stroke, Military Hospital 103 from September 2023 to April 2024. Restless legs syndrome was diagnosed 1 month after the stroke onset according to the criteria of the International Restless Legs Syndrome Study Group.
Results:
Restless legs syndrome was diagnosed in 59 patients (11.6%), including 37 (13.1%) who had ischemic stroke and 12 (8.6%) who had hemorrhagic stroke. An increased rate of restless legs syndrome was observed in stroke patients with brain lesions in the thalamus and lentiform nucleus; those who smoked; and those with a family history of restless legs syndrome.
Conclusions:
This study found that the incidence of restless legs syndrome after stroke was 11.6% overall and 13.1% and 8.6% after ischemic and hemorrhagic strokes, respectively. Lesion in the thalamus and lentiform nucleus, smoking history, and family history of restless legs syndrome were the predictors of restless legs syndrome after stroke onset.
Introduction
Stroke is defined as an abrupt neurological outburst caused by a sudden interruption of the blood supply to the brain secondary to blockage (ischemic stroke) or rupture (hemorrhagic stroke) of blood vessels. 1 It remains the second leading cause of death and a major cause of disability worldwide. 2 The neurological manifestations of stroke have been reported to be diverse, depending on the type of stroke and the affected area of the brain. 3 Even after discharge from the hospital, patients who had a stroke might face several physical or mental health issues that can affect their quality of life. 4
Restless legs syndrome (RLS) is a sensorimotor disorder rather than purely a motor disorder. It is characterized by an urge to move the legs and often associated with unpleasant paresthesias. 5 The worldwide prevalence of RLS was estimated to be ~3% in the general adult population 6 and was more pronounced in patients with stroke.7,8 In a meta-analysis of 13 studies on stroke-related RLS from 2009 to 2020, Wang et al. reported that incidence of stroke-related RLS ranged from 2.3% to 15.1%. 8 However, the reported incidence of stroke-related RLS had been mainly based on cases of ischemic stroke,7,9 –12 with few studies with a small number of patients with hemorrhagic stroke.7,13 In a study by Chandan et al. on 58 patients with stroke (47 ischemic and 11 hemorrhagic) in India, the incidence of stroke-related RLS was 10.3%. 13 In another study in India by Gupta et al. on 346 patients with stroke (139 hemorrhagic and 207 ischemic), including 35 patients who had RLS before stroke, the incidence of RLS after stroke onset was 2.3%. 7 However, neither of these two studies reported the incidence of poststroke-related RLS.7,13 In addition, the proportion of stroke-related RLS varies regionally, 8 and RLS has been reported to be both a risk and a consequence of ischemic and hemorrhagic strokes. 7 Suggesting, identifying the proportion of RLS cases, and recognizing the factors related to RLS after the onset of acute stroke are required for each country to better manage the quality of life of patients after stroke.
Vietnam is a developing country that has an increasing prevalence of stroke.2,14 However, to date, there had been no study on the rates and related factors of stroke-related RLS in Vietnam. Therefore, we conducted this study to investigate the incidence and factors related to RLS after ischemic and hemorrhagic strokes in Vietnam.
Methods
Study participants
This cross-sectional prospective study was carried out on a total of 1446 patients who had stroke and were diagnosed at the Department of Stroke, Military Hospital 103, Hanoi, Vietnam, from September 2023 to April 2024. All patients were evaluated by brain magnetic resonance imaging (MRI) with diffusion-weighted imaging (DWI) at 1.5 T (Siemens, Erlangen, Germany) within 7 days after the onset of symptoms to confirm acute ischemic or hemorrhagic stroke. Patients were excluded for the following reasons: (i) a prior medical diagnosis of RLS before stroke; (ii) cognitive impairment or disability in acquisition to their mother language at the time of examination; (iii) muscle pain, arthritis on the feet, lower extremity deep vein thrombosis, or peripheral neuropathy; (iv) use of neuroleptic, sedative, anti-seizure, or anti-extrapyramidal syndrome medications for other diseases; or (v) a medical history of stroke, other brain diseases, or evidence of previous brain lesions on brain MRI or computed tomography screening. Of these, 519 patients (35.9%) were included in the present study. A follow-up study was conducted at 1 month after the onset of stroke to evaluate for the presence of RLS. At that time, 90 patients refused to participate in this research, and six patients could not be contacted. The final number of patients for data analysis was 423 (140 patients with hemorrhagic stroke and 283 patients with ischemic stroke; Figure 1).

A flowchart illustrating the number of stroke patients.
Information on age, sex, preexisting conditions (heart and vascular diseases, metabolic disease, renal failure, and other diseases), family history of RLS (parents, siblings, or offspring), smoking status, and consumption of caffeine and alcohol were collected. Physical examination parameters, including weight, height, and body mass index (BMI), were collected.
Written informed consent was obtained from the patients or legally authorized representative for participants who were unable to provide informed consent themselves such as impaired mobility or inability to read or write at the time of enrollment. All patient details were deidentified. Furthermore, this study was conducted in accordance with the guidelines developed and approved by the Vietnam Military Medical University.
Brain MRI
The location of the ischemic or hemorrhagic brain lesions in all patients was detected by an experienced neuroradiologist on head MRI screening within 7 days from the initial symptoms using DWI, T2-weighted imaging, T1-weighted imaging, fluid-attenuated inversion recovery, and T2* gradient echo along with susceptibility weighted imaging.
Clinical assessments
The clinical stroke grade and consciousness of the patients upon admission to the hospital were assessed by the National Institute of Health Stroke Scale (NIHSS) 15 and the Glasgow Coma Scale. 16 At follow-up 1 month after stroke onset, the patients were evaluated for clinical manifestations of RLS using the following criteria of the International Restless Legs Syndrome Study Group 17 : (1) an urge to move the legs, which is usually but not always accompanied by or felt to be caused by uncomfortable and unpleasant sensations in the legs; (2) an urge to move the legs and any accompanying unpleasant sensations that begin or worsen during periods of rest or inactivity, such as lying down or sitting; (3) an urge to move the legs and any accompanying unpleasant sensations that are partially or totally relieved by movement, such as walking or stretching, at least as long as the activity continues; (4) an urge to move the legs and any accompanying unpleasant sensations during rest or inactivity, which only occur or are worse in the evening or night than during the day; and (5) occurrences of the abovementioned features are not solely accounted for as primary symptoms of another medical or behavioral condition (e.g., myalgia, venous stasis, leg edema, arthritis, leg cramps, positional discomfort, or habitual foot tapping). At this time, neurologic disability was also evaluated using the Modified Rankin Scale (mRS). 18
Data analysis
IBM SPSS version 21.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. The patients were divided into the RLS and non-RLS groups. The Shapiro–Wilk test was used to check for the normality of the distribution of variables, and appropriate methods were applied for statistical analysis. To investigate the association between stroke severity and degree of disability at 1 month after the onset of stroke and RLS, the patients were divided into three groups according to the scores on the NIHSS (0–3, 4–15, and 16–42) or mRS (0–1, 2–3, and 4–5). The variables were compared between the RLS and non-RLS groups using an independent sample t-test for continuous parameters and a chi-square test for categorical parameters. The relationships of the presence or absence of RLS-related stroke with the type of stroke, stroke severity based on the NIHSS score group, and degree of disability based on the mRS score group were also analyzed using the chi-square test. Finally, to define the factors that were independently associated with RLS in patients with stroke, a multivariate logistic regression analysis was performed for the variables that showed a significant association in the univariate model. Statistical significance was considered when p < 0.05.
Results
Characteristics of the participants
Table 1 shows the characteristics, including the age, proportion of men, family history of RLS, BMI, smoking status, alcohol consumption, caffeine intake, and preexisting condition, of RLS and non-RLS groups. Compared with the non-RLS group, the RLS group was older (61.6 versus 60.9 years); had higher BMI; and had higher proportions of patients who had a family history of RLS, as well as those who smoked and consumed alcohol and caffeine. The differences between the groups were significant for family history of RLS and smoking status (p < 0.05 for both). On the other hand, the proportion of men tended to be lower in the RLS group than that in the non-RLS group. There were no significant differences in the rates of preexisting medical conditions, including hypertension, diabetes, lipid disorders, and renal failure, between the RLS and non-RLS groups (Table 1).
Characteristics of the participants (N = 423).
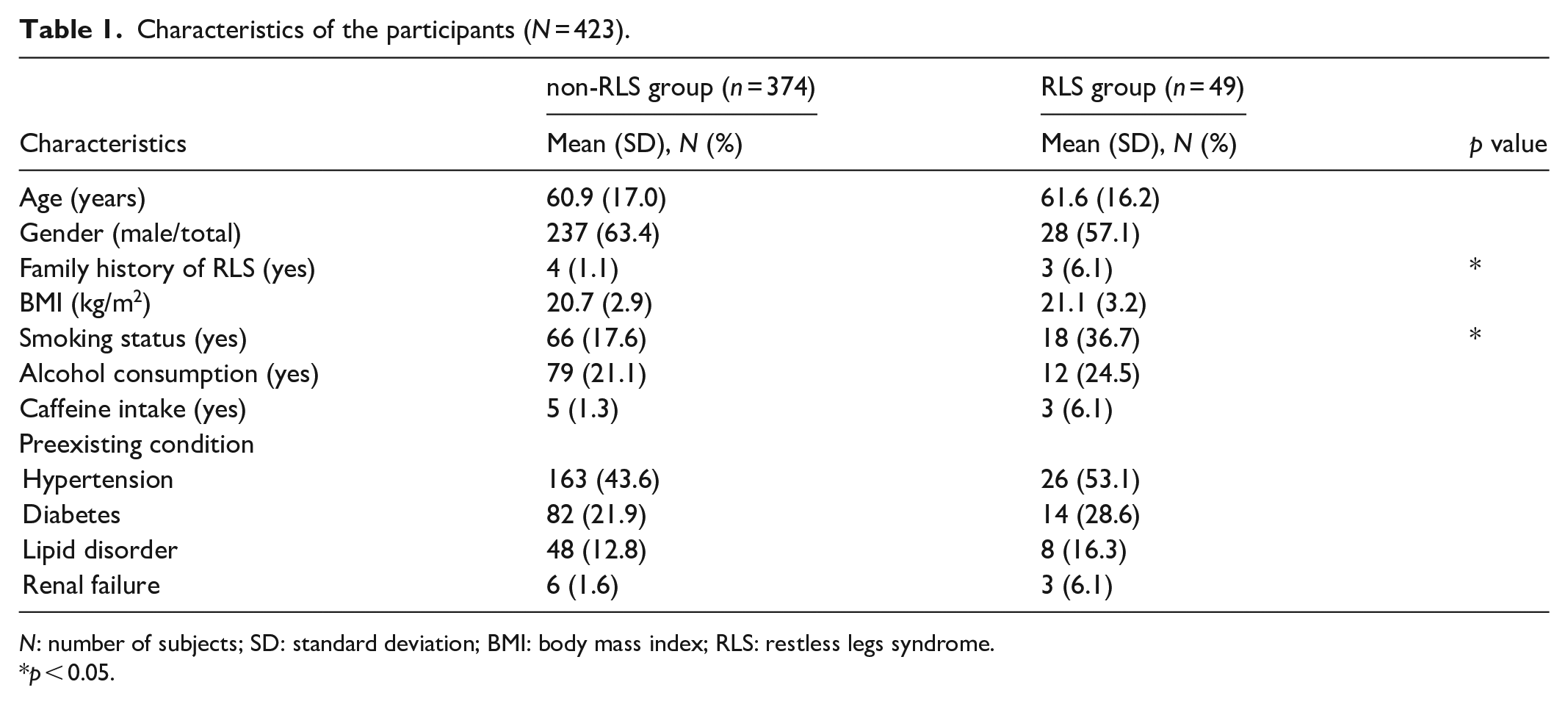
N: number of subjects; SD: standard deviation; BMI: body mass index; RLS: restless legs syndrome.
p < 0.05.
RLS incidence and association with clinical features
In this study, the number of patients who had hemorrhagic and ischemic strokes was 140 and 283 patients, respectively. Among them, 49 patients (11.6%) had RLS after ischemic (n = 37, 13.1%) and hemorrhagic (n = 12, 8.6%) strokes. In addition, a total of 171 patients had cortical strokes with infarcts or hemorrhages located in the cortical areas including frontal regions (26 patients), parietal regions (38 patients), occipital regions (33 patients), temporal regions (34 patients), and insular regions (40 patients), while 252 patients showed subcortical strokes with lesion in lentiform nucleus (34 patients), thalamus (24 patients), internal capsule (51 patients), caudate nucleus (22 patients), corpus callosum (19 patients), brainstem (42 patients), and cerebellum (50 patients). In addition, 15 (9.6%) and 34 (16.3%) patients showed RLS after cortical and subcortical strokes, respectively. The non-RLS and RLS groups had no significant differences in the type of stroke, brain location lesions (cortical and subcortical regions), NIHSS severity at the day of hospital admission, and degree of disability/dependence at 1 month after the stroke (p > 0.05; Table 2). However, the mean NIHSS score on the day of admission to hospital was significantly higher in the RLS group than in the non-RLS group (p < 0.05), but there was no significant difference in the mean mRS scores at 1 month after the onset of stroke between the RLS and non-RLS groups (Figure 2).
Relationships between clinical features and RLS after stroke onset.

N: number of subjects; RLS: restless legs syndrome; mRS: Modified Rankin Scale; NIHSS: National Institute of Health Stroke Scale.
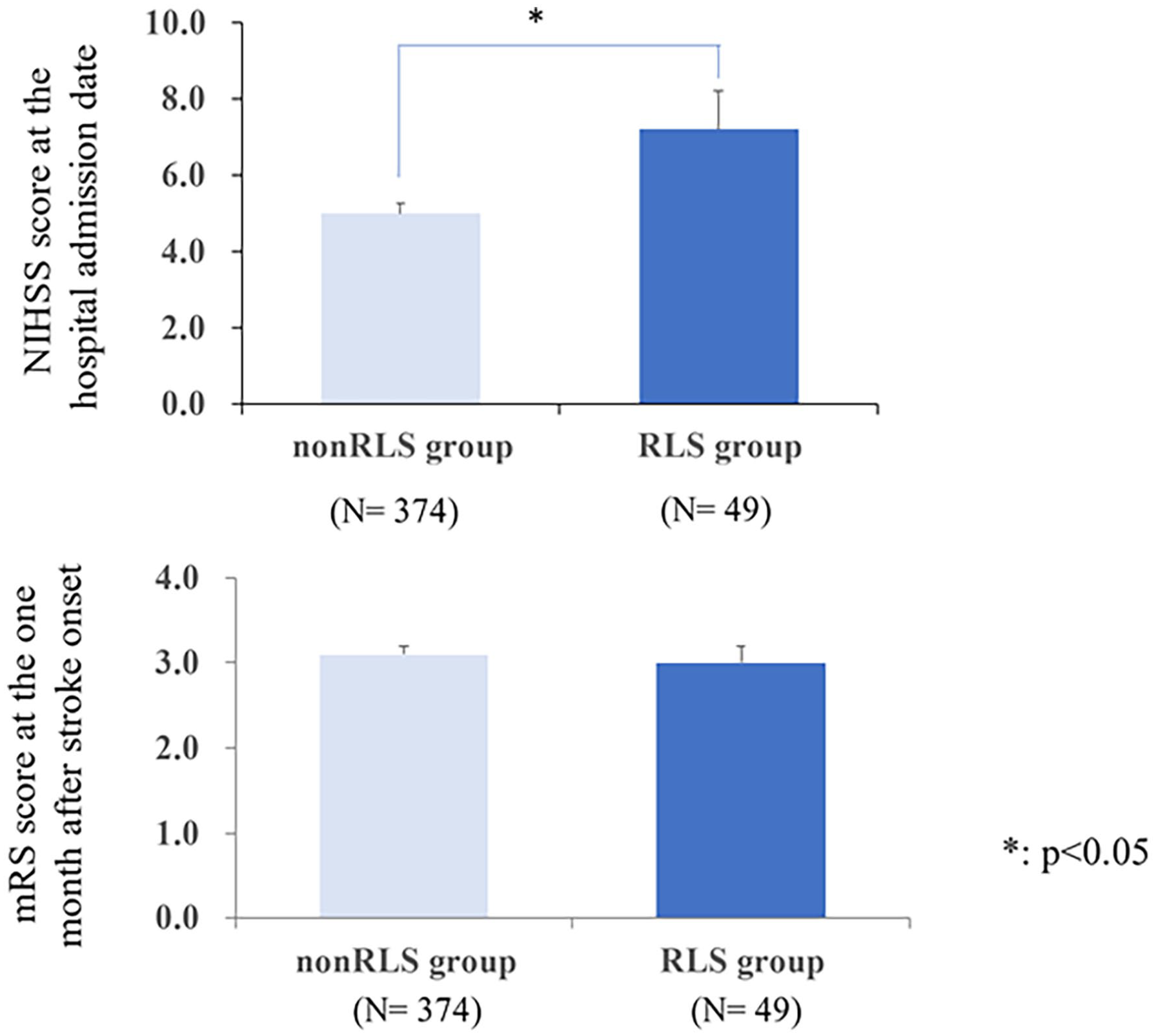
Comparison of the mean NIHSS and mRS scores between the RLS and non-RLS groups.
Relationship between brain lesions and RLS
Table 3 shows the relationships between the brain area lesions and RLS-related stroke. The percentage of patients with lesions in the lentiform nucleus, thalamus, caudate nucleus, and corpus callosum was significantly higher in the RLS group than in the non-RLS group (p < 0.05). There was no significant difference between the RLS and non-RLS groups in the percentage of lesions in the frontal, parietal, occipital, temporal, and insular lobes; internal capsule; midbrain; and cerebellum (Table 3).
Relationships between brain area lesion and of RLS after stroke onset.
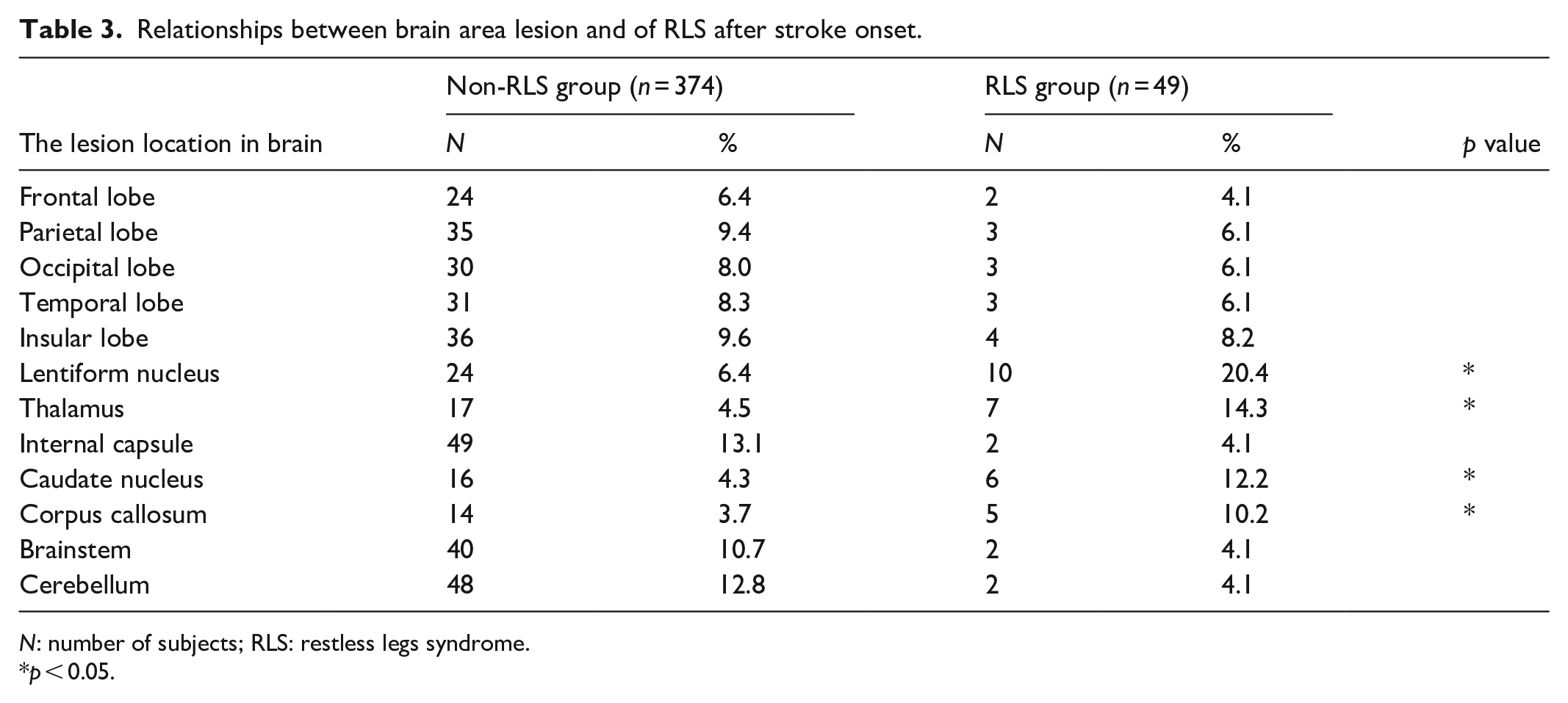
N: number of subjects; RLS: restless legs syndrome.
p < 0.05.
Potential risk factors associated with RLS after stroke onset
The results of the multivariate logistic regression analysis of the factors (i.e., smoking status; family history of RLS; lesion in the lentiform nucleus, thalamus, caudate nucleus, and corpus callosum; and mean NIHSS score at admission) that significantly affected the development of RLS on univariate analyses are presented in Table 4. Smoking status, family history of RLS, and lesions in the lentiform nucleus and thalamus were the independent factors related to RLS. Figure 3 shows the lesion in the thalamus (ischemic stroke) and putamen (hemorrhagic stroke) in patients with RLS (Figure 3). There were no significant differences between the RLS and non-RLS groups in terms of the mean NIHSS score and lesions in the caudate nucleus and corpus callosum (p > 0.05; Table 4)
Multivariate logistic regression analysis of the potential risk factors for RLS after stroke onset.
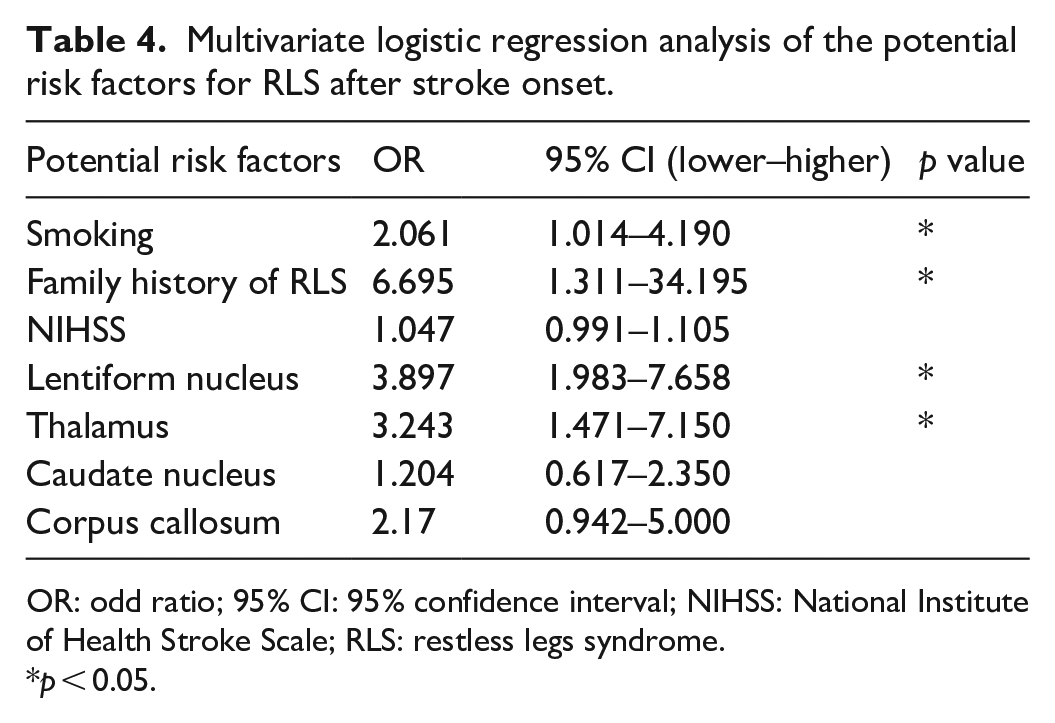
OR: odd ratio; 95% CI: 95% confidence interval; NIHSS: National Institute of Health Stroke Scale; RLS: restless legs syndrome.
p < 0.05.

Stroke lesions in the brain area on MRI. (a) Lesion in thalamus showed on MRI–DWI. (b) Lesion in thalamus showed on MRI–ADC image. (c) Lesion on putamen showed on MRI–DWI. (d) Lesion on thalamus indicated on MRI–ADC image.
Discussion
In this study, we found that the incidence of RLS at 1 month after the onset of stroke was 11.6% overall and 13.1% and 8.6% after ischemic and hemorrhagic strokes, respectively. The previously reported incidence/prevalence rates of RLS after stroke onset varied by region, with a range of 5%–15%, and were mainly based on cases of ischemic stroke. 8 In China, Tuo et al. found poststroke-related RLS in 13 of 244 (5.33%) consecutive patients with acute lacunar infarction. 11 In another study in China, Wu et al. reported RLS in 49 of 376 patients (13.03%) with acute cerebral infarction. 19 In Korean, Lee et al. reported that 17 of 137 patients (12.4%) were diagnosed as RLS after ischemic stroke. 10 In a study in Slovakia, 23 of 152 patients (15.1%) were diagnosed as RLS after the onset of acute ischemic stroke. 20 Meanwhile, there had been only a few studies on RLS related to hemorrhagic stroke. In a study in India, Chandan et al. reported that of 58 patients (10.3%) who had ischemic (n = 47) and hemorrhagic (n = 11) strokes, RLS developed in six patients, of which only one had hemorrhagic stroke-related RLS. 13 In another study in India, Gupta et al. collected data from 346 patients, including 139 with hemorrhagic stroke and 207 with ischemic stroke. The authors reported that 43 patients (10.1%), including 31 with ischemic stroke and 12 with hemorrhagic stroke, were diagnosed with RLS after stroke onset; however, among the 12 patients who had hemorrhagic stroke, RLS was diagnosed before the onset of stroke in 11 patients and after the stroke in only one patient. 7 Taken together, the results of the present study added data on the prevalence of stroke-related RLS, particularly hemorrhagic stroke.
Since it was reported that subcortical stroke is more strongly associated with RLS than cortical stroke.7,8 Gupta et al. reported that there were 29 patients (22.83%) in a total of 127 patients with subcortical strokes who reported RLS, while only 2.74% (6 of 210) of those with cortical-based strokes. 7 In this study, we found that the rate of RLS in patients after subcortical and cortical strokes was 16.3% and 9.6%, respectively. However, there was no difference between the proportion of RLS between subcortical and cortical strokes in this study. To explain it, we suppose that the number of stroke patients in this study is small. Also, stroke-related RLS has been reported to be associated with the topography of stroke,7,8 which may affect the rate of RLS in patients after cortical and subcortical strokes. Moreover, in the current study, we found that patients who had lesions in the lentiform nucleus, thalamus, caudate nucleus, and corpus callosum had a relatively high incidence of RLS. However, only lesions in the lentiform nucleus and thalamus were found to be the independent factors associated with RLS after stroke onset. Woo et al. reported that lesions in the corona radiata and adjacent basal ganglia were the most common sites of bilateral RLS after stroke. 21 Gupta et al. found that 82.8% of cases with stroke-related RLS had lesions in the subcortical area, including the basal ganglia, corona radiata, and centrum semiovale. 7 Lee et al. found that 16 of 17 patients with poststroke-related RLS had lesions in the subcortical areas. 10 Moreover, disruption of network loops rather than a specific nucleus has been proposed to be the mechanism of the variable anatomy in stroke-related RLS. 22 In addition, the thalamus was suggested to play a pivotal role in the generation of RLS symptoms. Stroke-induced changes in the thalamic nucleus volume and pontocerebellar fibers have been reported to be involved in the pathophysiology of RLS.23,24 Furthermore, injury to the thalamus was reported to interrupt sensory and motor networks and affect the control of dopamine secretion. 25 In a study on RLS in patients with lenticulostriate infarct-related RLS, Ruppert et al. reported that hyperdopaminergism, which was indicated by increased expression of dopamine precursors in response to stroke-induced neuronal loss, may contribute to RLS pathogenesis. 26 Taken together, further studies on the changes in the network loop in the subcortical areas in patients with stroke-related RLS, particularly the functional connectivity in the thalamic and dopaminergic pathways, are required.
Genetic factors have been suggested to be involved in the pathogenesis of RLS. 27 An increased risk of RLS was reported in individuals who have siblings or parents with RLS. 28 In a study on 54 patients with RLS, Ondo and Jankovic reported that 92% had a family history of RLS. 29 In the current study, we also found that the family history of RLS was an independent predictor of stroke-related RLS and accounted for 6.1% of stroke-related RLS. Similarly, Gupta et al. reported that 2 out of 35 patients (5.7%) with stroke-related RLS had a family history of RLS. 7 Moreover, Freeman et al. reported that RLS and cerebrovascular events, including strokes, may have a common cause, such as shared genetic predisposition. 30 Therefore, the role of genes in the mechanism of RLS after stroke onset needs to be clarified.
In this study, we found that smoking was independently associated with RLS after stroke onset. Although there was no association between smoking and RLS after stroke in the previous studies by Wu et al., 19 Gupta et al., 7 and Shiina et al., 31 smoking cigarettes has been reported to be linked with RLS in cerebrovascular events. 32 Bayard et al. reported that smoking was assumed to aggravate RLS conditions. 33 In addition, RLS might be a result of poor lifestyle, including obesity, physical inactivity, and smoking. 34 These findings indicated that cigarette smoking may be associated with poor lifestyle and could increase the rates of RLS after stroke.
Our findings showed that the initial NIHSS scores were significantly higher in the RLS group than in the non-RLS group, but this difference did not reach significance on multivariate logistic regression. Similarly, Wu et al. reported higher initial NIHSS scores in the RLS group than in the non-RLS group, but poststroke RLS was not associated with NIHSS. 19 On the other hand, Han et al. reported that RLS after acute ischemic stroke was associated with the initial NIHSS score, but it was not related to the brain lesion location. 35 These differences might be explained by the different populations between our study and the study by Han et al. Moreover, the differences in the brain lesions affected by stroke might have partly contributed to the different associations with the initial NIHSS scores. 36 In addition, in a longitudinal study by Medeiros et al., patients with acute stroke and RLS had worse clinical outcomes after 3 and 12 months. 37 In a prospective study, Boulos et al. demonstrated that patients with RLS had worse quality of life within 14 days after stroke or transient ischemic attack, and this negative impact persisted on follow-up assessment after 2 and 6 months. 4 This finding indicated the need for further studies to follow up on these patients and continuously investigate the negative influence of poststroke-related RLS.
Limitations
Our findings should be interpreted in the context of certain limitations. The calculation and justification of the sample size are limited in this study. The number of participants in the present study is relatively small, particularly when patients were categorized into non-RLS and RLS groups. Also, an appropriate control group was not established to compare the frequency and potential risk factors of RLS among patients with ischemic and hemorrhagic strokes. In the present study, we included all patients with or without RLS after stroke during the first month after stroke. However, we did not collect data on the onset of the RLS, its evolution, as well as asymmetrical or symmetrical symptoms. In addition, previous studies have suggested that various factors related to RLS such as genetics, iron deficiency, disturbances in the dopaminergic system, and patients with undiagnosed RLS symptoms existing prior to stroke8,38 were not measured in this study. Therefore, further large-scale studies that include this information should be performed in the future.
Conclusions
The incidence of RLS after stroke onset was 11.6% overall and 13.1% and 8.6% after ischemic and hemorrhagic strokes, respectively. This incidence was relatively high in patients with lesions in the lentiform nucleus, thalamus, caudate nucleus, and corpus callosum. Lesions in the lentiform nucleus and thalamus, smoking habit, and family history of RLS were the independent factors associated with RLS after stroke onset. Follow-up of these patients to continuously investigate the negative influence of poststroke-related RLS is necessary in future studies.
Footnotes
Acknowledgements
Ethical considerations
This study was conducted according to the ethical standards of the Institutional Ethics Committee and per the guidelines developed and approved by the Vietnam Military Medical University (no. 3389/QĐ-HVQY, August 17, 2023). The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from the patients or a legally authorized representative for patients who were unable to provide informed consent themselves such as impaired mobility or inability to read or write at the time of enrollment.
Author contributions
Do Xuan Tinh: conceptualization, methodology, data curation, formal analysis, validation, visualization, writing – original draft, writing – review and editing, investigation. Dinh Viet Hung: software, methodology, visualization, formal analysis, writing – original draft, writing – review and editing, supervisor. Do Duc Thuan: conceptualization, data curation, methodology, writing – original draft, data curation. Pham Ngoc Thao: formal analysis, writing – original draft, writing – review and editing. Nguyen Duc Thuan: data curation. Nguyen Trong Nghia: data curation. Huynh Ngoc Lang: data curation. Tran Minh Tuan: formal analysis. Dang Phuc Duc: data curation. Dang Minh Duc: data curation. Phan The Ha: data curation. Nguyen Dang Cuong: data curation. Nguyen Dang Hai: data analysis. All authors have read and approved the final submitted article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability Statement
The datasets in the current study are available from the corresponding author upon reasonable requests.
