Abstract
Objective
Stress urinary incontinence (SUI) is prevalent worldwide, particularly among elderly women. Although electroacupuncture (EA) has been accepted by many patients and physicians in Asia, its efficacy for SUI has not been evaluated scientifically and systematically. We aimed to conduct a systematic evaluation of the efficacy and safety of EA treatment for women with SUI.
Methods
We retrieved publications up to February 2019 from seven databases. Randomized controlled trials for women with SUI treated by EA were included. Therapeutic effect, 1-hour urine leakage and International Consultation on Incontinence Questionnaire – Short Form (ICIQ-SF) scores were the primary outcomes. The Cochrane Collection’s RevMan 5.3 software was used to pool data.
Results
The 15 included articles demonstrated that EA for SUI was effective (odds ratio [OR], confidence interval [CI] = 5.64, 4.19–7.59; Ι2 = 22%). ICIQ-SF scores increased (standard mean difference, CI = −0.48, −0.62 to −0.33; Ι2 = 32%) and 1-hour urine leakage decreased (OR, CI = −4.14, −4.96 to −3.33; Ι2 = 78%) in patients undergoing EA compared with those receiving sham EA, physical exercise or medication.
Conclusion
EA for women with SUI exhibited significant efficacy and safety for key outcomes.
Keywords
Introduction
Stress urinary incontinence (SUI) is defined by the International Continence Society as an involuntary loss of urine on physical exertion, sneezing or coughing. 1 Epidemiological studies have demonstrated that SUI is a common health problem worldwide, and is experienced by 23% to 45% of the female population. The prevalence in the United States is 24.8% 2 and that in China is 18.9%. 3 , 4
Although SUI is not life threatening, it negatively affects the physical health and social and psychological well-being of patients, causing reduced quality of life. Moreover, SUI is associated with complications, including diabetes, hyperlipidaemia and chronic kidney disease, which can be a burden for patients’ families. 5
A few effective therapies for SUI are available, including pelvic floor muscle training (PFMT), which is generally regarded as the first line treatment for SUI and is recommended by the American Urological Association; 3 however, PFMT must be practised for a long time and adherence rates are negatively correlated with time. 6 , 7 In addition, PFMT training techniques are difficult to master. Surgery can be effective for patients with severe SUI; however, the potential complications, which include pain, infection and dysuria following surgery, are unacceptable for patients with SUI. Hence, effective and safe treatments for patients with SUI are urgently required.
Acupuncture has been practised for over 2500 years in China, and includes a range of techniques, such as manual acupuncture (MA), scalp acupuncture, electroacupuncture (EA) and body acupuncture. EA is particularly recommended by the National Institutes of Health as a supplementary or alternative treatment for many diseases, including SUI. A 2017 randomized controlled trial (RCT) demonstrated a clinically significant reduction in urine leakage and improved quality of life in patients administered EA compared with those receiving sham EA treatment. 8 Several clinical studies have suggested that acupuncture, including EA, can effectively treat SUI by improving acetic-acid induced bladder irritation through inhibition of capsaicin sensitivity. 9 , 10 The results of one meta-analysis of studies of patients with urinary calculus showed that the acupuncture treatment group had a higher total effective rate and cure rate and a lower urinary calculi excretion rate than the drug treatment group. 11
Nevertheless, the status of current evidence means that the efficacy of acupuncture for SUI remains controversial. For example, a 2013 meta-analysis that investigated standard interventions, including MA, EA and other acupuncture methods, showed that acupuncture had an uncertain curative effect on SUI. 10 However, EA and MA are not interchangeable. Therefore, research must differentiate between the effects of these two methods; studies that mix MA and EA are not homogeneous. 10
There have been many clinical studies and reports on the effects of EA treatment in patients with SUI; however, meta-analyses evaluating the effectiveness and safety of this type of therapy are lacking. Here, we aimed to assess the efficacy and safety of EA for SUI and provide high quality evidence for its clinical therapeutic effects in this context. This study provides detailed evidence regarding the safety and efficacy of EA therapy for SUI.
Methods
This systematic review and meta-analysis is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement 12 and was registered in the International Prospective Register of Systematic Reviews (number: CRD42018112689).
Search strategy
We systematically searched databases for relevant studies published up to February 2019, including three international and four Chinese databases: PubMed, the Cochrane Library, Web of Science, China National Knowledge Infrastructure Database (CNKI), Chinese Biological Medicine Database (CBM), VIP (Database for Chinese Technical Periodicals) and WanFang Digital Periodicals Database (WFDP). The search terms used related to acupuncture (e.g. acupuncture, electroacupuncture, needle, needles) and SUI (e.g. stress urinary incontinence, stress incontinence). Only papers originally written in English or Chinese were considered. In addition, included papers were limited to clinical trials, RCTs and National Institutes of Health RCTs.
We used the following combined text for searches: ‘((acupuncture OR acup* OR electroacupuncture OR electroacup*) AND (stress urinary incontinence OR stress incontinence OR uroclepsia OR incontinentia urinae))’. Databases were searched for publications indexed between their inception and February 2019.
Inclusion criteria
Studies that met the following criteria were included.
Types of participants
All studies needed to include women who met the criteria for the standardized diagnosis of SUI; 12 within a trial, the control group had to have received the same baseline interventions as the EA group. Studies were excluded if they contained patients with serious systemic or neurologic disease or urinary system infection; patients who had undergone preoperative radiotherapy or chemotherapy; patients who had serious conditions, such as cardiovascular, liver, kidney and haematopoietic system disease (because they may have had an unusual medical history); or patients who refused EA treatment.
Types of studies
RCTs of EA as a treatment option for SUI were included. Nonrandomized trials, quasi-experimental studies, observational studies, animal studies, qualitative studies, letters, news articles, editorials and commentaries were also excluded. The titles and abstracts of the searched articles were read by a single primary researcher (JC) who had trained in literature searching and systematic reviewing for 3 years. If the articles were not written in English, we primarily reviewed them using their abstracts, which were translated into English prior to screening by a commercial service (if we experienced difficulty with language comprehension). Articles for potential inclusion were checked by two independent reviewers (ZY, JC).
Types of intervention/control groups
Treatment groups received EA; there was no limit on the needle material, choice of acupoints, treatment methods, needle retention time or course of treatment. Control groups received internationally recognized therapies, such as PFMT or Western medicine, or sham EA therapy.
Types of outcome measures
The primary outcome was the therapeutic effect. The secondary outcomes were scores on the International Consultation on Incontinence Questionnaire – Short Form (ICIQ-SF), 13 the change from baseline in the amount of urine leakage as measured by the 1-hour pad test, and the frequency and nature of adverse events.
Data extraction
The titles and abstracts of all citations identified in the search were imported into Endnote, Version X6 (Clarivate Analytics, Thomson Reuters, Canada) and duplicates removed. These citations were independently reviewed for eligibility by two reviewers (JC and ZY) and the full texts of ambiguous articles retrieved if a consensus was not reached. When agreement could not be reached, a third author (PC) was consulted.
Two reviewers independently extracted the data from each trial into Excel, using a standardized form, and entered the combined data into RevMan 5.3 (Copenhagen, Denmark: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). The following data were extracted from each selected study: first author, publication year, total number of participants, age of patients, disease course, details of intervention and control groups, outcome indicators and reported adverse events. When consensus could not be reached, a third author (PC) was consulted.
Assessment of risk of bias
Two reviewers (JC and ZY) independently assessed the methodological quality of each included trial. A third reviewer (PC) helped to resolve disagreements. RevMan 5.3 was used to assess the risk of bias of each study using the following items: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other bias. We scored each criterion as showing a ‘high’, ‘low’ or ‘unclear’ risk.
Statistical analysis
All statistical analyses were conducted using RevMan 5.3. Studies were combined according to the types of intervention and assessment of the therapeutic effect of EA. Standardized mean difference (SMD) and 95% confidence intervals (CI) were calculated in the meta-analysis if the data were continuous. Risk estimates (odds ratio [OR]) and 95% CI were calculated if the data were dichotomous. Heterogeneity between studies was evaluated using the χ2 (chi-squared) test (
Subgroup analysis
If there was significant heterogeneity among the included trials, we conducted subgroup analysis, based on patient age, severity of SUI, different EA acupoints and the course of treatment.
Sensitivity analysis
If factors influencing heterogeneity could not be identified using subgroup analysis, we conducted a sensitivity analysis using the leave-one-out approach. Publication bias was evaluated using funnel plot analysis if a sufficient number of trials (10 trials) were identified; it was not evaluated when there were fewer than 10 studies in a group.
Level of evidence
The Grading of Recommendations, Assessment, Development and Evaluations (GRADE) framework was used to assess the level of evidence and summarize each outcome. The level of evidence was classified as high, moderate, low or very low.
Results
Search results
Figure 1 shows details of the trial selection process. We retrieved 1150 articles, 125 from CNKI, 116 from WFDP, 50 from VIP, 77 from CBM, 306 from Web of Science, 299 from PubMed and 177 from the Cochrane Library. After initial screening of the abstracts and titles, 1022 studies were excluded because they were case reports, duplicates or meta-analyses; 128 articles were fully evaluated. Next, we excluded a further 89 articles, four of which compared the efficacy of EA with that of acupuncture, and 85 because they were of low quality, as evaluated using the Cochrane Handbook for Systematic Reviews of Interventions. The full texts of 39 articles were downloaded and assessed. Thirteen articles were excluded from further evaluation because they did not describe RCTs; 11 articles were excluded because they included duplicate data. Finally, 15 RCTs were analysed. 8 ,14–27 Details of this strategy and a flow chart of the analysis are presented in Figure 1.

Flow diagram of search results and study selection. CNKI: China National Knowledge. Infrastructure Database; WFPD: WanFang Digital Periodicals Database; VIP: Database for Chinese Technical Periodicals; CBM: Chinese Biological Medicine Database; EA: electroacupuncture; RCT: randomized controlled trial.
Characteristics of included RCTs
Table 1 shows the main study characteristics. A total of 1577 patients were included in the analysis: 790 in experimental groups and 787 in control groups. All included studies were published between 2004 and 2019. The age of included patients ranged from 26 to 84 years. All studies used EA with no other treatment interventions. Five studies compared EA with sham EA,8,14,20,23,24 with a pragmatic placebo needle used at sham acupoints. Three studies compared EA with oral midodrine hydrochloride, 16 , 19 , 22 a synthetic sympathomimetic amine that is structurally similar to methoxamine, a relatively long-acting α1-selective adrenergic agonist. Three included studies compared EA with PFMT, 18 , 21 , 26 which enhances the ability of the urinary tract sphincter to control urine and improves the symptoms of urinary incontinence by enhancing the function of the pelvic floor muscles. The other studies compared EA with traditional electrical stimulation, acupuncture alone or with MiNing (Chinese drug approval number: Z20026838), 15 , 17 , 25 , 27 which is a Chinese diuretic drug. Fourteen studies had treatment durations between 4 and 12 weeks. Only one article reported a treatment duration of less than 4 weeks (10 days). 25 Baseline characteristics and data were reported in each included study. Thirteen studies reported the therapeutic effect as the primary outcome, seven studies evaluated ICIQ-SF scores, two studies reported clinical symptom scores and two reported the frequency of urinary incontinence. Four studies reported adverse events. A summary of the studies is presented in Table 1.
Characteristics of the included studies.

SUI: stress urinary incontinence; E: experimental group; C: control group; EA: electroacupuncture; SD: standard deviation; PFMT: pelvic floor muscle training; IQOL: Incontinence Quality of Life Questionnaire; UDI: Urinary Distress Inventory; VAS: visual analogue scale: ALPP: abdominal leak point pressure; MUCP: maximum urethral closure pressure; MUP: major urinary protein; SFL: functional urethral length; Y: years; M: months; ICUD: International Consultation on Urological Diseases; EAM: European Association of Urology; ICI: International Continence Society.
Assessment of risk of bias
Two authors (JC and ZYZ) independently assessed the risk of bias for all included studies, using the Cochrane risk-of-bias tool, which evaluates random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting and other biases. As EA involves insertion of needles connected to low-voltage electricity, it is difficult to blind patients and physicians.
Thirteen papers described a proper method for, and details of, randomization, which was conducted using computerized randomization, a randomization list, the PROC PLAN procedure in SAS software (SAS Institute Inc., Cary, NC, USA) or sealed envelopes. Hence, these studies had a low risk of bias. Two studies were evaluated as having a high risk of bias, because they did not describe the randomization sequence (Figure 2).

Risk of bias graph.
Given the obvious nature of EA, most studies did not conduct blinding. Only five studies, in which sham EA was used as the control intervention, performed blinding and therefore had a low risk of bias. All other studies had a high risk or an unclear risk of bias for the blinding of outcome assessment, as insufficient information was provided to assess whether or not the experimenters were blinded (Figure 3).

Risk of bias summary.
Main findings
Therapeutic effects
Thirteen studies reported therapeutic effects, including the ability to control urination, improvement of symptoms of discomfort, lack of catheterization requirement and residual urine volume <100 mL. As total heterogeneity was large, we used a sensitivity analysis to determine the heterogeneity values following elimination of studies one by one. Finally, the heterogeneity was reduced by 22% after eliminating the study by Chen and Ma (2015)
18
(OR, CI = 5.64, 4.19–7.59;

Forest plot of pooled estimates of total therapeutic effect. CI: confidence interval.
ICIQ-SF scores
Of the 15 studies, 6 reported ICIQ-SF scores. Analysis of their combined results demonstrated statistical significance (SMD, CI = −0.61, −0.74 to −0.48;

Forest plot of pooled estimates of ICIQ-SF scores. CI: confidence interval.
1-hour pad test
Combined analysis of five studies reporting data from the 1-hour pad test indicated statistical significance (OR, CI = −4.14, −4.96 to −3.33;

Forest plot of pooled estimates for the 1-hour pad test. CI: confidence interval.
Adverse events
Only four studies reported adverse events. The study by He et al. (2016) 20 reported that, in the control group, 21 of 50 patients had adverse events: 11 experienced xerostomia, 4 headache and 6 dizziness. Bi (2007) 16 reported that in the control group, 11 patients experienced xerostomia, 7 dizziness, 4 headache and 1 patient experienced piloerection; however, there were no adverse events in the EA group. Xu et al. (2014) reported that 1 patient could not endure the pain in the EA group, and 2 patients developed haematomas. 24 Liu et al. (2017) 8 reported that 2 control patients experienced fatigue, 1 sharp pain, and 1 palpitations, whereas in the EA group, 4 patients developed subcutaneous haematoma and 1 experienced fatigue.
Publication bias
We use RevMan to evaluate the bias in the total therapeutic effect of EA. A total of 13 trials were included, all from China. The distribution of included studies was asymmetric on both sides of the funnel plot, indicating the presence of publication bias in the therapeutic effect of EA (Figure 7).
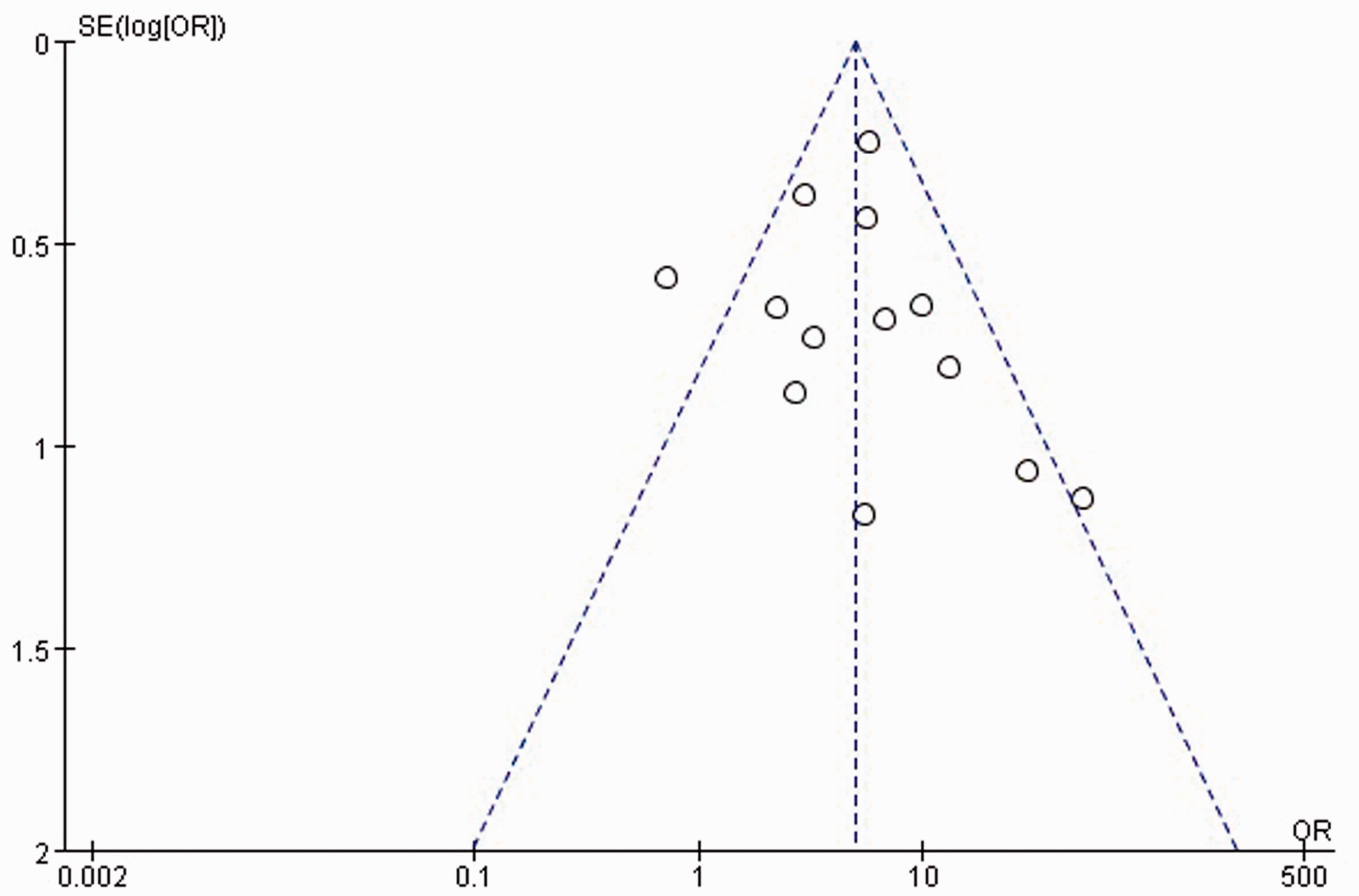
Funnel plot showing analysis of publication bias.
Discussion
Modern medicine considers the brain and spinal cord to be high-level nerve centres that control urination. The urinary sphincter can be controlled by neuromodulation of urinary sphincter activity. Studies have shown that the sacral spinal nerves S2 and S4 play an important role in the process of urination. 29 The detrusor activity of the bladder is dominated by the sacral nerves, including S4 and S2. EA may stimulate the skin or organ tissues, which are innervated by the sacral nerves. The nerve impulses then transfer to the striated muscles, which are innervated by S2 to S4. In addition, electrical stimulation of S3 is transmitted to the spinal cord, the nerve signals are transmitted to the thalamus, and then the thalamus integrates the impulses, which regulate bladder function. 30
One cause of SUI is an excessive or weak detrusor bladder function. The use of different EA frequencies (2 Hz and 100 Hz) can regulate the detrusor function to normal or near normal. 31 Additionally, some studies have shown that EA stimulation of the acupoint Huiyang (BL 35) can stimulate the sacral autonomic nervous system, thereby enhancing the strength of the pelvic floor muscles. 8 In addition, EA stimulates the perineal nerve, which regulates detrusor muscle function and controls urination. 32
Clarification of the effectiveness of EA compared with other conservative or drug therapies for patients with SUI is urgently required. Although many studies have shown that EA can have substantial therapeutic effects on SUI, its efficacy has not been confirmed scientifically. Here, we aimed to determine the effectiveness of EA by summarizing and evaluating its overall therapeutic effect and its influence on urine leakage and ICIQ-SF scores.
Thirteen studies comparing EA with other conservative or drug therapies reported a therapeutic effect of EA. All found statistically significant benefits of EA therapies, according to the duration of treatment (≤4 weeks and >4 weeks). Six studies used ICIQ-SF scores and five used 1-hour urine leakage as primary outcomes. Our analysis showed that EA was superior to other therapies or drugs in terms of therapeutic effect, 1-hour urine leakage and ICIQ-SF scores. Analysis of therapeutic effects suggested that EA was highly effective compared with control treatments, without any accompanying heterogeneity for the first subgroup (≤4 weeks), but with substantial heterogeneity for the second subgroup (>4 weeks). The data on 1-hour urine leakage revealed a clear and significant reduction of urine leakage, with considerable heterogeneity; however, subgroup and sensitivity analyses reduced heterogeneity to within the acceptable range. Analysis of improvements in ICIQ-SF scores showed that EA was significantly superior to control treatments and that heterogeneity was reduced significantly by removal of the study by Wang et al. (2016); however, we did not identify differences among studies according to treatment time, intervention or course of disease.
Although this meta-analysis demonstrates that EA can improve and ameliorate SUI, more investigations are needed. More importantly, sample sizes were small in the included studies, with no multicentre or multiblinded trials, and only four studies reported adverse events. In these, 49 control group patients and 7 EA group patients experienced adverse reactions, such as fatigue, sharp pain, palpitations and haematoma. We have yet to determine the mechanism by which EA ameliorates SUI; however, its effectiveness in relieving and improving symptoms has been confirmed by numerous studies and is widely accepted by clinicians and patients.
Conclusion
EA for women with SUI exhibited significant efficacy and safety for key outcomes, but additional large-scale, long-term RCTs with rigorous methodological quality are needed.
