Abstract
Objective
To explore whether protective ileostomy is beneficial in preventing anastomotic leakage after anterior resection of rectal cancer.
Methods
A total of 347 patients underwent anterior resection of rectal cancer in our hospital. Ninety-five patients were treated with protective ileostomy (treatment group), and 252 patients were not (control group). The incidences of anastomotic leakage and permanent stoma were compared between the two groups.
Results
The overall incidences of anastomotic leakage were 6.32% (6/95) and 8.73% (22/252) in the treatment group and control group, respectively. In the cohort of patients who underwent neoadjuvant radiotherapy, the incidence of anastomotic leakage was 5.88% (2/34) and 12.0% (3/25) in the treatment group and control group, respectively. Logistic regression showed that the incidence of anastomotic leakage was not statistically significant. However, diabetes and the anastomotic height significantly affected the occurrence of anastomotic leakage. The permanent stoma rate was 6.42% (6/95) and 5.95% (15/252) in the treatment group and control group, respectively.
Conclusion
Protective ileostomy did not show a significant advantage in reducing the incidence of postoperative anastomotic leakage in patients with rectal cancer, and it may lead to a permanent stoma.
Keywords
Introduction
Rectal cancer is a common type of gastrointestinal cancer, and its incidence is increasing year by year. Surgery is the major treatment modality and is often supplemented by preoperative neoadjuvant radiotherapy and chemotherapy or postoperative adjuvant chemotherapy. By promoting and implementing neoadjuvant treatment, many patients with low rectal cancer retain their anal anatomy and function after the operation. Anterior resection of rectal cancer (Dixon method) is the major anal-preservation operation. 1 However, postoperative anastomotic leakage is a serious complication after such an operation. Studies have shown that the mortality rate after anastomotic leakage can be as high as 16%.2–4 Anastomotic leakage not only affects the patient’s postoperative recovery but also increases the length of hospital stay and medical expenses. Severe cases may require reoperation and can even significantly affect the patient’s long-term survival.5,6 Therefore, how to reduce the incidence of anastomotic leakage remains an important topic.
In recent years, the use of terminal ileostomy after anterior resection of rectal cancer to lower the incidence of anastomotic leakage has been increasing in clinical practice. However, whether protective ileostomy can effectively reduce the incidence of anastomotic leakage and whether it is worth being adopted as a routine surgical treatment for rectal cancer remain controversial. Some studies have suggested that protective ileostomy reduces the occurrence of anastomotic leakage, especially in patients with low rectal cancer and rectal cancer after neoadjuvant therapy.7–9 However, other reports have demonstrated that a protective stoma was unable to effectively reduce the incidence of anastomotic leakage and instead caused inconvenience to patients. In addition, the incidence of intestinal obstruction and other complications increased and the length of hospital stay was prolonged.10,11 Therefore, whether such patients should be routinely treated with a protective stoma is unclear.
In the present study, we retrospectively analyzed the clinical data of patients who underwent anterior resection of rectal cancer with or without protective ileostomy in our hospital to explore the significance of protective ileostomy in preventing anastomotic leakage and permanent stoma after anterior resection of rectal cancer. Our goal is to provide useful information on choosing individualized treatment options in patients with rectal cancer.
Methods
Clinical information
We reviewed the clinical data of all patients with rectal cancer who underwent anterior resection from June 2011 to July 2018 in our hospital. The inclusion criteria were an age of ≥18 years; postoperative pathological diagnosis in line with the diagnostic criteria of rectal cancer and no distant metastasis; feasible distance between the lower edge of cancer and the anal edge for anal-preserving surgery; no serious functional disorders or illness involving the heart, brain, digestive system, liver, kidney, or lungs; and no other malignant tumors. The exclusion criteria were stage IV cancer that could not be radically removed combined with a poor prognosis, other serious diseases and no indication for surgical treatment, serious infection or trauma, and serious nervous system diseases or mental disorders. The patients were divided into two groups: those who underwent protected ileostomy (treatment group) and those who did not (control group).
All patients underwent colonoscopy and pathological examination before the operation with a defined diagnosis of rectal cancer. In addition, a medical imaging examination was routinely performed to classify the American Joint Committee on Cancer preoperative clinical stage (I, II, or III). Rectal cancer resection was performed according to the principle of total mesorectal excision. A protective ileostomy was created at the terminal ileum, 15 to 20 cm proximal to the ileocecal valve in the lower right abdominal quadrant, with single- or double-lumen stoma that would be recovered 3 to 6 months postoperatively.
Classification and diagnosis of anastomotic leakage
In 2010, the International Rectal Cancer Research Group defined postoperative anastomotic leakage after surgical treatment of rectal cancer as an interruption and defect of the integrity of the intestinal wall at the site of anastomosis of the colorectum or coloanal canal, resulting in a connection between the internal and external compartments (including reconstruction of the rectal leakage at the suture site of the storage bag, such as a J-pouch) and a pelvic abscess near the anastomotic site. 12 Anastomotic leakage is further graded as follows. Grade A is defined as subclinical anastomotic leakage (also known as imaging anastomotic leakage) without clinical symptoms or a requirement for special treatment. Grade B is characterized by abdominal pain, fever, and purulent or fecal-like drainage from the anus, drainage tube, or vagina (rectovaginal fistula); an increased white blood cell count and C-reactive protein concentration; and anastomotic leakage requiring conservative treatment. Grade C is characterized by peritonitis, sepsis, and other clinical manifestations of Grade B anastomotic leakage requiring a secondary surgery. By combining the above standards, patients who met any one of the following four criteria were determined to have anastomotic leakage in the present study: the drainage tube near the pelvic anastomosis drained the intestinal contents or had a fecal odor, outflow of contrast agent was observed by digestive tract radiography, gas accumulation and intestinal wall discontinuity were found around the anastomosis site by computed tomography examination, or the anastomotic opening was confirmed by anal palpation using a finger. These findings were combined with other clinical evidence of anastomotic leakage, such as fever, chills, positive blood culture, or high leukocyte count.
Statistical methods
Data are expressed as mean ± standard deviation and were analyzed with SPSS version 19.0 statistical software (IBM Corp., Armonk, NY, USA). Because comparison of the patients’ ages in this study involved specific measurements, the
Ethics and consent
This study was a retrospective analysis and did not involve patient privacy concerns, disease exposure, or life-threatening conditions; therefore, no ethics approval was needed. Verbal consent was obtained from all patients in this study.
Results
In total, 347 patients qualified for this study after application of the inclusion and exclusion criteria. Among these patients, 95 underwent protected ileostomy (treatment group) and 252 did not (control group). The treatment group comprised 59 men and 36 women ranging in age from 33 to 89 years (mean, 64.2 ± 10.5 years). Seventy-eight patients underwent laparoscopy and 17 underwent laparotomy. The tumor stage was stage I in 31 patients, stage II in 33, and stage III in 31. The histological classification was highly differentiated adenocarcinoma in 17 patients, moderately differentiated adenocarcinoma in 68, poorly differentiated adenocarcinoma in 4, and mucinous adenocarcinoma in 6. The control group comprised 146 men and 106 women ranging in age from 35 to 82 years (mean, 61.7 ± 10.3 years). Laparoscopic surgery was performed in 189 patients, and laparotomy was performed in 66. The tumor stage was stage I in 58 patients, stage II in 101, and stage III in 83. The histological classification was highly differentiated adenocarcinoma in 48 patients, moderately differentiated adenocarcinoma in 175, poorly differentiated adenocarcinoma in 13, and mucinous adenocarcinoma in 16. There were no statistically significant differences in sex, age, tumor stage, or histological classification between the two groups, as shown in Table 1.
Clinical information.

Data are presented as mean ± standard deviation or number of patients.
NS, not significant.
All 347 patients successfully completed the treatment. Among all patients, 6 in the treatment group (6/95, 6.32%) and 22 in the control group (22/252, 8.73%) developed anastomotic leakage. In the stratified analysis, the treatment group and control group showed no significant differences in the proportions of .Grade A leakage (1/6 vs. 7/22 (16.67% vs. 31.83%)), Grade B leakage (4/6 vs. 12/22 (66.6% vs. 54.45%)), or Grade C leakage (1/6 vs. 3/22 (16.67% vs. 13.63%)). With neoadjuvant radiotherapy, the incidence of anastomotic leakage in the treatment group and control group was 5.88% (2/34) and 12.00% (3/25), respectively, as shown in Table 2.
Incidence and grades of anastomotic leakage.
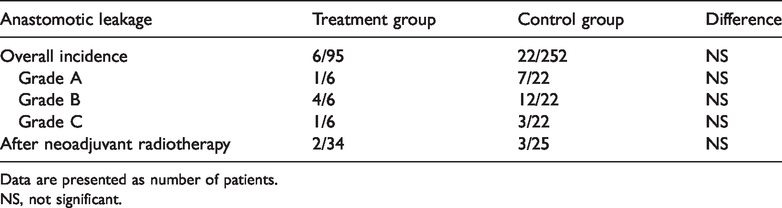
Data are presented as number of patients.
NS, not significant.
Because a univariate analysis may have been biased, we conducted a multivariate analysis (logistic regression) of the clinical data to rule out confounding effects of other factors. The results showed that sex, hypertension, utilization of neoadjuvant therapy, and protective terminal ileostomy were not associated with the incidence of anastomotic leakage. However, a history of diabetes and the anastomotic height seemed to contribute to the occurrence of anastomotic leakage (
Multivariate analysis of clinical parameters.

In the treatment group, the preventive ileostomy was recovered in 89 of 95 patients. The shortest duration until ileostomy recovery was 125 days, the longest was 294 days, and the mean was 221.6 ± 47.5 days. Compared with a protective stoma, a permanent stoma is no longer included in the abdominal cavity and serves as a permanent excretion route for the patient. Six patients in the treatment group failed to return; the proportion of a permanent stoma was 6/95 (6.42%). Among these patients, one underwent conversion to a permanent colostomy because of a recurrent anastomotic stoma, one discontinued treatment for physical reasons associated with advanced age, and four (including three with anastomotic leakage) developed postoperative anastomotic stenosis. Fifteen patients in the treatment group underwent permanent stoma creation for various reasons after anterior resection; the proportion of permanent stoma creation was 5.95% (15/252). Among these patients, 2 underwent permanent colostomy because of intractable diarrhea, 2 underwent emergency permanent colostomy because of severe anastomotic leakage, and 12 underwent permanent colostomy because of tumor recurrence and metastasis with intestinal obstruction (including 5 patients with anastomotic leakage). There were no significant differences in these clinical conditions between the two groups, as shown in Table 4.
Incidence of permanent stoma.

Data are presented as number of patients.
NS, not significant.
Discussion
Clinicians and patients are increasingly demanding higher quality of life after radical resection along with the development and revolution of surgical techniques for rectal cancer and the application of comprehensive treatment modalities. With the wide application of stapler technology and the promotion of total mesorectal excision for rectal cancer (especially the low rectal cancer), anal preservation is becoming increasingly more feasible through adoption of the Dixon method of anterior rectal resection, and the rate of anal preservation has been steadily increasing during the past few years. However, this technique also makes the operation more difficult and riskier. Anastomotic leakage is still a major postoperative complication that seriously affects patients’ quality of life 13 and increases the economic burden and family burden. Research has shown that the incidence of anastomotic leakage ranges from 1% to 21%.14,15 How to reduce this complication remains a persistent concern among surgeons. With the extensive development of laparoscopic surgical techniques, increased participation of young surgeons performing this operation, and clinicians’ increasing recognition of neoadjuvant radiotherapy and chemotherapy for rectal cancer, protective ileostomy has emerged and become increasingly popular. However, if the patient is not individually diagnosed and treated, the surgeon may choose to create a protective stoma at will and potentially perform this treatment as a routine modality in rectal cancer resection. This would undoubtedly increase the surgical trauma, cost of medical treatment, risk of secondary surgery, and even the need to convert from a protective to permanent stoma. Especially in countries where patients must bear part of the medical expenses, implementation of a second operation can result in an increased financial burden on the patient’s family after the operation. Therefore, this method of preventing anastomotic leakage is controversial.
The occurrence of anastomotic leakage is believed to be related to the following five factors: intestinal ischemia or a poor blood supply, neoadjuvant radiotherapy and chemotherapy, high anastomotic tension, poor intestinal preparation, and infection.16,17 In view of this, research has shown that a preventive stoma can cause most of the intestinal contents to be discharged through the stoma and thus achieve the effect of diversion, minimize the expansion effect of feces on the anastomotic opening, protect the blood supply of the anastomotic opening, avoid complications such as infection of the anastomotic opening, shorten the postoperative fasting time, and further decrease the incidence of anastomotic leakage. 18 In contrast, a protective stoma cannot effectively reduce the incidence of postoperative anastomotic leakage of rectal cancer. Instead, it may increase the incidence of other complications such as disuse enteritis, intestinal obstruction, and other conditions. 19 The incidence of anastomotic leakage in the present study was about 6% to 8%, and a protective ileostomy had no clear advantage in reducing the incidence of anastomotic leakage after rectal cancer surgery. Our in-depth analysis showed that regardless of the grade of anastomotic leakage (Grade A, B, or C) or whether a protective ostomy was performed after neoadjuvant therapy, a preventive ileotomy did not significantly reduce the incidence of anastomotic leakage. We also conducted a multifactor regression analysis. The results showed that the occurrence of anastomotic leakage was not related to whether a protective stoma was implemented. The occurrence rate of anastomotic leakage can be effectively reduced by correct and reasonable use of anastomotic techniques, ensuring that the anastomotic opening has a blood supply, minimizing the tension of the anastomotic opening, and controlling the amount of intestinal fluid and food intake to within a certain period of time. Studies have shown that a <5-cm distance between the anastomotic stoma and anal verge can significantly increase the incidence of anastomotic leakage.20,21 Our study also showed that diabetes and the anastomotic height significantly affect the incidence of anastomotic leakage. In addition, surgeons believe that a protective stoma can reduce or even prevent the occurrence of serious abdominal and pelvic infections, abscesses, and septic shock caused by anastomotic leakage after anterior resection of low and middle rectal cancer and that it can also avoid the need for a secondary surgery for anastomotic leakage.22,23 Therefore, a protective ileostomy is preferred for patients with an anastomotic stoma distance of <5 cm, regardless of whether they have diabetes, to avoid the above-mentioned serious adverse events caused by anastomotic leakage. However, a protective ileostomy is not recommended as a routine modality for all patients with rectal cancer.
Some surgeons consider that inflammatory edema and a lack of blood supply to the local microcirculation are present in the intestinal tract after neoadjuvant therapy, and the disappearance or serious adhesion of the anatomical space will lead to ischemia of anastomotic cells and disordered protein production, 24 thus increasing the incidence of anastomotic leakage. Therefore, a protective stoma is recommended. However, the main role of a protective stoma is to reduce the expansion effect of feces and fecal-associated infection at the anastomotic orifice. A protective stoma provides no substantial benefits in terms of improving the local blood supply or nutrition. Allison et al. 25 reported that in the mid to upper rectum, a good collateral blood supply is present between the small arteries within the mesorectum. In the lower rectum, however, there are only a few and quite variable intramural collaterals between the small arteries. Unrecognized destruction of arteriolar collateral branches during rectal resection may be related to anastomotic leakage. Our study shows that neoadjuvant therapy has no significant relationship with the occurrence of anastomotic leakage, and a protective ileostomy thus has little benefit for these patients. Therefore, to lower the incidence of anastomotic leakage, it is important to improve the nutritional status of patients with rectal cancer in the perioperative period after neoadjuvant therapy, reduce the damage to mesenteric vessels by careful performance of the operation, retain the blood supply and vitality of the remnant intestinal tract under the premise of radical cure, and reduce the risk of an unprotected ileostomy.
The routine recovery time after creation of a protective stoma is 3 to 6 months postoperatively. In the present study, the recovery time was about 7 months. Further analysis showed that more than 60% of patients in this study were diagnosed with advanced rectal cancer and received adjuvant chemotherapy after the operation. Most of these patients chose to return to the ileotomy after chemotherapy had been completed. To a certain extent, this increases the expenses related to daily care and stoma maintenance. The incidence of diarrhea in patients with ostomies increases during postoperative adjuvant therapy, and this directly affects the progress of adjuvant therapy. 26 Additionally, six patients in the treatment group of the present study were unable to return or had to convert to a permanent stoma for various reasons. However, there was no significant difference from the control group, implying that a protective stoma does not reduce the incidence of a postoperative permanent stoma. Lim et al. 27 reported that stage IV cancer, anastomosis-related complication, the colostomy type, systemic metastasis, and local recurrence were independent risk factors for a permanent stoma after anterior resection for rectal cancer. Waterland et al. 28 reported that nearly one-quarter of patients were unable to financially afford payment for a stoma, and the procedures in half of the patients were postponed for 6 months even if they completed the payment. The main reason for the delay was not only the need for follow-up adjuvant therapy but also complications, especially anastomotic leakage. Research has shown that anastomotic complications are one of the most important factors for the failure to return.29,30 The results of our study are basically consistent with this finding. Our study also indicates that candidates for preventive ileostomy should be carefully selected.
In conclusion, our clinical practice has shown that the occurrence of anastomotic leakage can be reduced by ensuring a preoperative evaluation, clear anatomical identification, correct and reasonable use of anastomotic techniques, control of postoperative intestinal fluid production and food intake within a certain period of time, and adequate nutritional support and fluid management. However, protective ileostomy is still recognized and accepted by some surgeons. Therefore, more systematic analyses of multicenter trials are expected to provide additional solid clinical evidence. We suggest that candidates for preventive ileostomy should be carefully selected according to their actual clinical condition, and such patients should be assessed especially carefully during surgical exploration. We do not recommend preventive ileostomy as a routine modality in patients undergoing anterior resection of rectal cancer.
