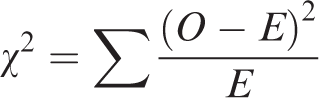Abstract
Anastomotic leakage is a major complication following laparoscopic low anterior resection in patients with low rectal cancer. This study investigated the preventive effects of three intraoperative techniques, including the preservation of the left colic artery, reinforcement suturing, and transanal decompression tube placement. A total of 88 patients undergoing laparoscopic rectal cancer surgery were retrospectively analyzed. Univariate and multivariate logistic regression analyses were performed to assess the associations between these interventions and the occurrence of anastomotic leakage. The overall anastomotic leakage rate was 7.95%. Left colic artery preservation, reinforcement suturing, and transanal decompression tube placement each showed a lower anastomotic leakage incidence (5.3%, 6.3%, and 7.4%, respectively) compared with nonapplication. The combined use of all three measures resulted in zero anastomotic leakage. Multivariate analysis confirmed a consistent protective trend, with left colic artery preservation demonstrating the strongest effect. Preserving the left colic artery, reinforcing the anastomosis, and reducing intraluminal pressure act synergistically to reduce the risk of anastomotic leakage. Their integrated use may offer an effective strategy to enhance anastomotic safety in rectal surgery.
Keywords
Introduction
With global transition in dietary patterns and the progressive aging of populations, the incidence of colorectal cancer (CRC) has been increasing steadily worldwide, posing a growing public health burden.1,2 Furthermore, emerging epidemiological and epigenetic evidence associate ambient air pollution with CRC pathogenesis, expanding the spectrum of recognized risk factors for this malignancy. 3 Concurrently, pooled analyses have identified microplastic exposure as another underrecognized environmental risk factor contributing to CRC development, highlighting the need to consider multiple environmental determinants in disease prevention. 4 Among CRC cases, low rectal cancer represents a particular clinical challenge because of its complex anatomy and high risk of postoperative complications.5,6 According to the most recent national epidemiological data from China, CRC ranks third in incidence and fifth in mortality among all malignancies, with rectal cancer accounting for 30%–50% of CRC cases. 7 Notably, low rectal cancer, defined as tumors with a distal margin ≤5 cm from the dentate line, comprises approximately 60%–75% of rectal cancer cases. 8 Its deep pelvic location, intimate relationship with the anal sphincter complex, and the substantial risk of anastomotic leakage (AL) following resection collectively make its management one of the most demanding areas in colorectal surgery.
Radical resection remains the cornerstone of curative treatment for rectal cancer, and the evolution of surgical techniques has profoundly shaped patient outcomes.9–11 Although conventional open surgery achieves oncological clearance, it is associated with significant surgical trauma, prolonged recovery, and postoperative immune suppression. The advent of minimally invasive surgery, particularly laparoscopic techniques, has revolutionized rectal cancer management by offering reduced surgical trauma, faster recovery, and superior cosmetic results. 12 Moreover, minimally invasive approaches have demonstrated advantages in preserving pelvic autonomic nerves and reducing intraoperative blood loss. 13 However, their potential to reduce the incidence of AL, a critical determinant of postoperative morbidity and survival, remains unclear.
Low anterior resection (LAR) is considered the gold standard sphincter-preserving procedure for patients with low rectal cancer.14,15 Nonetheless, postoperative AL continues to be a major concern for colorectal surgeons.16,17 Reported incidence rates range from 3% to 19% and remain above 10% even in modern laparoscopic series. 18 AL not only increases the risk of reoperation (odds ratio (OR) = 3.12, 95% confidence interval (CI): 1.98–4.91), hospital costs (an average increase of approximately US$15,000), and mortality (hazard ratio = 1.67) but is also strongly associated with higher local recurrence rates (relative risk (RR) = 2.05) and a 12.8% reduction in 5-year survival.19,20 Recent metabolomic studies have further demonstrated that perioperative metabolic alterations can predict postoperative complications and hospital stay in patients with CRC, thereby offering potential biomarkers for personalized risk stratification prior to surgery. 21 Consequently, effective prevention of AL is crucial for improving both short- and long-term outcomes.
Current strategies for AL prevention primarily include protective stoma creation, left colic artery (LCA) preservation technique (P technique), anastomotic suture reinforcement technique (S technique), and transanal tube placement technique (T technique). 22 However, these interventions are largely based on single-factor measures and remain controversial. For example, although a protective stoma can mitigate the severity of AL, stoma-related complications occur in up to 28% of cases. Similarly, evidence regarding the efficacy of the p technique varies considerably across studies (OR = 0.32–0.89). 23 This wide range of effect sizes indicates significant heterogeneity in study outcomes, supported by a high I2 statistic in recent systematic reviews,24,25 reflecting inconsistent conclusions about the protective effect of LCA preservation against AL. In addition to these surgical interventions, hematological parameters such as red blood cell distribution width and hematocrit have been validated as predictive markers for short-term outcomes and prognosis in patients with CRC undergoing radical surgery, which can aid in refining risk assessment models for AL prevention. 26
Against this background, Jianmin Xu and colleagues at Zhongshan Hospital, Fudan University, pioneered a combined preventive approach by integrating the simultaneous implementation of P, S, and T techniques (PST). 27 Data presented at the 2019 Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) annual meeting demonstrated that this technique reduced the preventive stoma rate to 0.3% and the AL incidence to 5.0%, markedly outperforming those reported in the Comparison of Laparoscopic versus Open Resection for Rectal Cancer (COLOR) II trial (stoma 36.3%, AL 13.9%) and the Randomized Comparison of Laparoscopic versus Robotic Resection for Rectal (ROLARR) cancer trial (stoma 85.1%, AL 11.1%). These results introduced a new paradigm for the prevention of AL following rectal cancer surgery.
Nevertheless, studies investigating the application of the PST technique in minimally invasive surgery for rectal cancer remain limited. Given the plausible complementary roles of LCA preservation, anastomotic reinforcement, and transanal drainage in preventing AL, further high-quality evidence is warranted.
Materials and methods
In this study, we conducted a single-center prospective randomized controlled trial involving 88 patients undergoing laparoscopic resection for low rectal cancer. Patients were randomly assigned to one of the following five groups to compare the efficacy of combined PST preventive measures versus single-factor interventions: (a) Group 1 (Control). No PST interventions (conventional laparoscopic LAR); (b) Group 2 (P-only). LCA preservation alone; (c) Group 3 (S-only). Anastomotic suture reinforcement alone; (d) Group 4 (T-only). Transanal decompression tube placement alone; (e) Group 5 (PST combined). Simultaneous application of P, S, and T techniques
The primary objectives were as follows: (a) delineate the independent contribution of each PST component to AL prevention in minimally invasive surgery and (b) elucidate the synergistic mechanisms among these techniques. The findings aimed to provide robust, high-level evidence to optimize laparoscopic rectal cancer surgery and to promote a paradigm shift in AL prevention from empirical, single-modality interventions toward a precise, multimodal, and mechanism-informed strategy.
Study population
This study prospectively enrolled patients admitted to the Department of Colorectal and Abdominal Tumor Surgery II, Jilin Provincial Cancer Hospital, between September 2020 and September 2023. All participants were initially diagnosed with rectal cancer using colonoscopy, with rectal cancer defined as tumors located ≤15 cm from the anal verge (as measured by withdrawal of the colonoscope and digital rectal examination). Eligible patients had no evidence of distant metastasis and were staged according to the eighth edition of the American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) tumor–node–metastasis (TNM) classification system. Staging criteria were defined as follows: clinically staged tumor (T) 1–3, clinically staged node (N) 0–1 (cT1–3N0–1) for patients with mid/low rectal cancer and cT4aN0–2, pathological tumor stage after neoadjuvant therapy (ycT) (ycT1–3)Nx for patients with upper rectal cancer (pathologically downstaged to T1–T3 after neoadjuvant chemoradiotherapy (nCRT), nodal status unspecified) for patients who received neoadjuvant treatment. All patients were considered suitable for laparoscopic surgery. In cases of conflicting staging findings among computed tomography (CT), endorectal ultrasonography (ERUS), and pelvic magnetic resonance imaging (MRI), a multidisciplinary team (MDT) discussion involving colorectal surgeons, diagnostic radiologists, and medical oncologists was conducted to reach a consensus. Pelvic MRI was prioritized as the gold standard for assessing local tumor depth and nodal involvement, whereas CT was used to rule out distant metastases, and ERUS served as a supplementary tool for early-stage tumors (cT1–T2). The reporting of this study conforms to Case Report (CARE) guidelines. 28
Inclusion criteria
The inclusion criteria were as follows: (a) age between 18 and 80 years; (b) histopathological confirmation of rectal adenocarcinoma via colonoscopic biopsy; (c) tumor located ≤15 cm from the anal verge; (d) no evidence of distant metastases (including pelvic, peritoneal, hepatic, pulmonary, cerebral, osseous, or distant lymph node metastases) as determined via ultrasonography, CT, or positron emission tomography–CT (PET-CT); (e) preoperative pelvic MRI and/or ERUS indicating clinical staging of cT1–3N0–1, upper rectal cancer with cT4aN0–2, or ycT1–3 after nCRT; (f) no history of other malignancies (except adequately treated basal cell carcinoma of the skin or carcinoma in situ of the cervix); (g) considered suitable for laparoscopic radical surgery; (h) provision of written informed consent.
Exclusion criteria
The exclusion criteria were as follows: (a) cT1N0 lesions suitable for local excision; (b) emergency surgery due to acute intestinal obstruction, hemorrhage, or perforation; (c) multiple primary colorectal malignancies; (d) hereditary or inflammatory bowel diseases, including familial adenomatous polyposis, Lynch syndrome, or inflammatory bowel disease; (e) conditions requiring simultaneous colectomy; (f) American Society of Anesthesiologists physical status classification >III; (g) pregnancy or lactation; (h) inability of the patient or the patient’s family to comprehend the study objectives or criteria; (i) receipt of preoperative treatment other than standard nCRT; (j) undergoing open or robotic surgery instead of laparoscopic resection.
Withdrawal criteria
The withdrawal criteria were as follows: (a) intraoperative exploration or postoperative pathology confirming distant metastasis; (b) requirement for combined organ resection during surgery; (c) intraoperative construction of a protective or permanent stoma; (d) postoperative pathological diagnosis other than rectal adenocarcinoma; (e) receipt of treatments in addition to the study protocol during the study period (except for subsequent treatment after recurrence or metastasis); (f) voluntary withdrawal from the study cohort after enrollment due to any reason; (g) inability to continue treatment due to newly developed nononcological comorbidities after enrollment; (h) incomplete participation or failure to complete the planned study protocol for any reason.
Surgical procedures
Standard laparoscopic procedure for rectal cancer
All procedures were performed with the patient in the modified lithotomy position. A 1.0-cm vertical infraumbilical incision was made, through which a 10-mm trocar was inserted to establish pneumoperitoneum at 13 mmHg. A 30° laparoscope was introduced. Two additional trocars (12 mm and 5 mm) were placed at two separate anatomical landmarks: the intersection of the lateral border of the right rectus abdominis muscle and the umbilical horizontal line and the other at the intersection of the lateral border of the right rectus abdominis muscle and the line connecting the bilateral anterior superior iliac spines. These served as the main and auxiliary working ports, respectively. Two 5-mm trocars were symmetrically positioned on the left side as assistant ports.
Using an ultrasonic scalpel, the peritoneum was incised longitudinally along the white line of Toldt and extended cranially to the root of the inferior mesenteric artery (IMA) at the anterior surface of the aorta. Dissection proceeded within the Toldt fascia plane cranially until the IMA root was exposed. The IMA and vein were clipped and divided using hemostatic clips. The sigmoid mesocolon was divided, and dissection was continued cranially along the left avascular plane of the sigmoid mesentery to connect the descending and sigmoid colon planes up to the splenic flexure. Caudally, the rectum was mobilized posteriorly using the ultrasonic scalpel down to the level of the rectosacral ligament. The mesenteric vascular pedicle was divided accordingly.
The bowel was skeletonized 5 cm distal to the tumor margin and transected using an endoscopic linear stapler. The umbilical port site was then extended to approximately 5 cm to exteriorize the proximal colon. A purse-string suture was placed at a point 10 cm proximal to the tumor margin using a purse-string clamp, and the bowel was divided for specimen extraction. The anvil head of a circular stapler was inserted into the proximal bowel and secured by tightening the purse-string. The circular stapler body was introduced transanally to perform end-to-end colorectal anastomosis under laparoscopic guidance (Figure 1).

Laparoscopic Preservation of the Left Colic Artery (P technique). (a) Conventional ligation strategy; (b) schematic illustration of the P technique and (c) intraoperative photograph.
After completion of the colorectal anastomosis with a circular stapler, the anastomotic site and residual stump were reinforced using interrupted “figure-of-eight” sutures with 3-0 absorbable material (Figure 2). This reinforcement technique enhances the mechanical stability of the anastomosis and may reduce the risk of postoperative AL.

Suture reinforcement of the anastomosis (S technique) during laparoscopic rectal cancer surgery. (a) Schematic illustration of the S technique and (b) intraoperative photograph.
Following completion of the colorectal anastomosis, a soft silicone drainage tube (1.2 cm in diameter) was inserted transanally into the rectum. The tube was advanced proximally so that its upper tip was positioned approximately 10 cm above the anastomotic site within the bowel lumen (Figure 3). The transanal tube facilitates decompression of the distal bowel and may reduce anastomotic tension and the risk of AL.
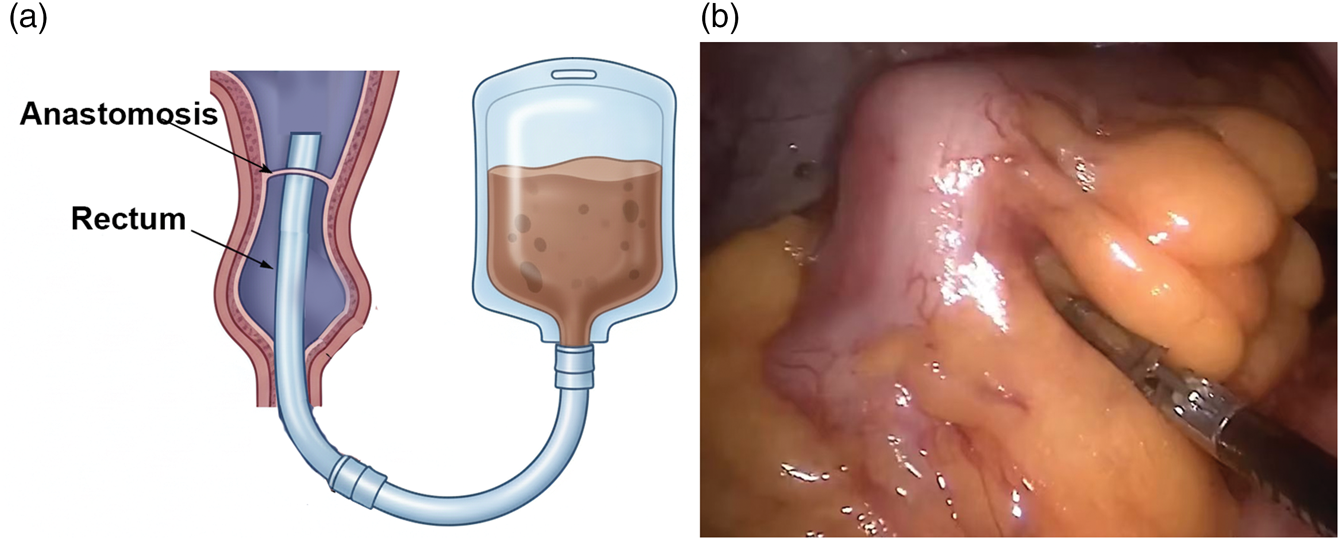
Transanal tube placement (T technique) during laparoscopic rectal cancer surgery. (a) Schematic illustration of the T technique and (b) intraoperative photograph.
Observation indicators
Diagnostic criteria for AL
The diagnostic criteria for AL were based on the definition proposed by the International Study Group of Rectal Cancer (ISREC). AL is defined as a defect in colorectal or coloanal anastomotic integrity, resulting in communication between the intraluminal and extraluminal compartments. According to this definition, a pelvic abscess adjacent to the anastomotic site is also considered an anastomotic leak. 29
At present, no universally accepted single diagnostic standard exists for AL following rectal cancer surgery. Therefore, diagnosis requires a comprehensive assessment of clinical manifestations, laboratory parameters, imaging findings, and physical examination results.
30
The major diagnostic features are summarized as follows:
Paroxysmal or persistent pain in the lower abdomen, perineum, or presacral region or a sensation of anal distension; Localized peritoneal irritation, which may progress to diffuse peritonitis, systemic inflammatory response, or septic shock; Intermittent or persistent fever accompanied by leukocytosis, neutrophilia, and elevated C-reactive protein (CRP) levels, after excluding other sources of infection such as wound, respiratory, or urinary tract infections; Presence of feculent or turbid drainage fluid from intra-abdominal or pelvic drains; Digital rectal examination revealing a palpable defect or fistulous opening at the anastomotic site; Radiologic evidence on contrast-enhanced CT demonstrating extraluminal air, fluid–gas levels, or discontinuity at the anastomotic site.
31
Classification of AL
AL was graded according to the ISREC classification system based on its clinical severity: (a) Grade A. Radiologic or minor clinical leakage that does not require active therapeutic intervention and does not affect postoperative recovery; (b) Grade B. Leakage with localized or mild peritonitis requiring active medical therapy, such as antibiotics and percutaneous or transanal drainage, but not reoperation; (c) Grade C. Leakage accompanied by overt peritoneal irritation, intra-abdominal sepsis, or fecal peritonitis necessitating emergency surgical intervention.
This standardized classification provides a reproducible framework for assessing the clinical impact of AL and facilitates objective comparison across studies. 32
Observational parameters
The following perioperative and postoperative parameters were recorded for all patients: operative duration (min), intraoperative blood loss (mL), postoperative hospital stay (days), incidence of AL (overall and stratified by ISREC grades A/B/C), and incidence and spectrum of postoperative complications (including infection, bleeding, ileus, and urinary dysfunction).
All postoperative complications (including AL and other adverse events) were prospectively documented. Overall surgical morbidity was graded according to the Clavien–Dindo classification to ensure standardized evaluation of surgical morbidity. 33 Anastomotic integrity was routinely assessed through clinical monitoring, drain output analysis, and postoperative imaging when clinically indicated. 31
Statistical analysis
All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) 26.0 software. Baseline characteristics between groups were compared using chi-square tests for categorical variables and independent samples t-tests or one-way analysis of variance (ANOVA) for continuous variables. The primary endpoint (AL rate) was analyzed using binary logistic regression to adjust for potential confounding factors. A two-sided p-value <0.05 was considered statistically significant.
Univariate analysis was performed to evaluate the association between individual factors, including preservation of the LCA, placement of a transanal drainage tube, and reinforcement of the anastomotic triangle, and the incidence of AL in patients with rectal cancer. The chi-square test (χ² test) and Fisher’s exact test were used, as appropriate. The chi-square statistic was calculated as follows:
The RR and risk difference were further calculated to quantify the association between exposure factors and AL occurrence using the following equations:
Multivariate logistic regression analysis was subsequently performed to determine the independent effects of each factor while adjusting for potential confounders. The dependent variable was binary (AL occurrence: yes = 1, no = 0), and the independent variables consisted of three dichotomous predictors: preservation of the LCA (P: yes = 1, no = 0), reinforcement of the anastomotic triangle (S: yes = 1, no = 0), and placement of a transanal drainage tube (T: yes = 1, no = 0). The regression model was defined as follows:
Continuous variables were compared using the t-test when normally distributed, whereas categorical variables were analyzed using the χ² test or Fisher’s exact test, as appropriate. A two-sided p-value <0.05 was considered statistically significant.
Results
Baseline characteristics of patients
A total of 88 patients were included in the final analysis, with 17–18 patients across the five groups (Table 1). Baseline demographic, clinical, and pathological characteristics were well-balanced across all groups. There were no significant differences between groups in age, sex, body mass index (BMI), tumor location, clinical stage, or nCRT status. These findings confirm the comparability of the study cohorts and validate the randomization process.
Baseline characteristics of patients.

BMI: body mass index; P-only: left colic artery preservation alone; S-only: anastomotic suture reinforcement alone; T-only: transanal decompression tube placement alone; PST combined: simultaneous application of P, S, and T techniques; ANOVA: analysis of variance; CRT: chemoradiotherapy.
Incidence of AL
A total of 88 patients who underwent laparoscopic colorectal surgery were included in this study. The objective was to evaluate the preventive effects of the three operative techniques, namely P, S, and T techniques, on the incidence of AL in patients with rectal cancer. Grouping and outcomes are summarized in Table 2.
Incidence of anastomotic leakage.

LCA: left colic artery; AL: anastomotic leakage.
Univariate analysis
In the univariate analysis, the incidence of AL was 5.3% (1/19) in patients undergoing LCA preservation (P-only group) and 8.7% (6/69) in those without preservation. The RR was 0.61 (95% CI: 0.08–4.72), suggesting a trend toward reduced AL risk with LCA preservation; however, the difference did not reach statistical significance (Fisher’s exact test, p = 0.53).
For patients with a transanal drainage tube (T-only group, n = 27), the AL rate was 7.4% (2/27), compared with 8.2% (5/61) in patients without drainage (RR = 0.90, 95% CI: 0.18–4.45; p = 1.00). Similarly, reinforcement of the anastomotic triangle (S-only group, n = 32) was associated with an AL rate of 6.3% (2/32), compared with 8.9% (5/56) in patients without reinforcement (RR = 0.70, 95% CI: 0.14–3.51; p = 0.71). None of these differences were statistically significant.
Multivariate and interaction analysis
Multivariate logistic regression analysis further demonstrated that LCA preservation was associated with a lower, although not statistically significant, risk of AL (OR = 0.55, 95% CI: 0.06–4.78). The effects of triangle reinforcement (OR = 0.65, 95% CI: 0.12–3.56) and transanal tube placement (OR = 0.85, 95% CI: 0.15–4.89) were likewise not significant.
Notably, among patients with LCA preservation, the incidence of AL remained consistently lower than in those without preservation, regardless of whether adjunctive techniques were employed (maximum 12.5% vs. minimum 6.7%). The LCA P-only group demonstrated the highest AL rate (12.5%); however, this subgroup included only eight patients. Therefore, the result should be interpreted with caution because of limited statistical power. This finding suggests that maintaining the integrity of the LCA may preserve local perfusion and mitigate the deleterious effects of other operative variables. The only subgroup with zero AL incidence comprised patients with the combined triple intervention (PST combined, n = 6), indicating a potential additive protective effect of multimodal strategies despite the limited sample size.
Although statistical significance was not achieved, the overall trend consistently supported LCA preservation as a potentially pivotal factor in reducing the risk of AL in patients with rectal cancer. Its protective effect may exceed that of ancillary techniques such as anastomotic reinforcement or transanal drainage. Larger prospective studies are warranted to validate these findings, clarify the independent and synergistic effects of the three interventions, and elucidate the physiological mechanisms through which LCA preservation enhances anastomotic healing.
Discussion
Mechanisms and efficacy of the P technique in preventing AL
Clinical observations in this study indicated that preservation of the LCA may be the most effective surgical maneuver for reducing the risk of AL following colorectal resection. 34 Detailed analysis suggests that its protective effects are likely multifactorial, including hemodynamic, structural, and biological mechanisms.
Maintenance of adequate arterial perfusion
Anatomically, LCA represents a major branch of the IMA, supplying the descending and sigmoid colon. High ligation of the IMA, as performed in conventional rectal cancer surgery, may compromise distal perfusion, particularly when collateral circulation via the marginal artery of Drummond is insufficient. 35 Because anastomotic healing critically depends on microcirculatory integrity, preserving the LCA ensures continuous and physiologically direct perfusion to the distal bowel segment, thereby mitigating ischemia-induced hypoxia and tissue necrosis at the anastomotic site.
Protection of neural and lymphatic integrity
The mesenteric nerve plexus and perivascular lymphatics course alongside the LCA. Preserving the vessel minimizes inadvertent nerve injury, thereby maintaining normal bowel motility, peristaltic coordination, and secretory activity. Similarly, preservation of lymphatic drainage may prevent local inflammatory accumulation and cytokine-mediated tissue damage around the anastomotic site, promoting a more favorable microenvironment for healing. 36
Reduction of operative trauma and inflammatory stress
LCA preservation limits unnecessary dissection and tissue manipulation, thereby reducing local ischemia–reperfusion injury and oxidative stress. 37 Attenuation of this iatrogenic inflammatory response further facilitates mucosal regeneration and collagen remodeling, both of which are essential for secure anastomotic healing.
Collectively, these mechanisms suggest that LCA preservation confers not only a vascular advantage but also broader physiological benefits, reinforcing its role as a cornerstone strategy in the prevention of AL.
Mechanisms and efficacy of the S technique in preventing AL
The S technique contributes meaningfully to reducing the incidence of AL, primarily through biomechanical and barrier-forming effects.25,38
Mechanical reinforcement
Supplementary suturing using interrupted or seromuscular inverting stitches enhances the mechanical strength of the anastomosis and stabilizes tissue approximation. This reinforcement counteracts elevated intraluminal pressure caused by postoperative bowel distension or early peristalsis, thereby preventing suture-line dehiscence. It also compensates for minor imperfections inherent to stapling, such as uneven tissue compression or incomplete staple closure, which may otherwise result in microleaks.
Local antileakage barrier
Even minute defects such as pinholes or staple gaps can serve as early sources of leakage. Reinforcement sutures may provide a secondary serosal seal, creating an additional barrier that may limit bacterial translocation and intra-abdominal contamination during the critical early postoperative phase.
Technical and psychological assurance
From the surgeon’s perspective, reinforcement suturing offers a “double safeguard,” particularly valuable in high-risk anastomoses, such as those in patients with low rectal tumors or marginally perfused sites, where mechanical stability and security are paramount.
Overall, although reinforcement suturing cannot fully substitute for adequate perfusion, it serves as an effective adjunctive measure that enhances mechanical integrity and early anastomotic stability.
Mechanisms and efficacy of the T technique in preventing AL
The T technique provides an auxiliary benefit in preventing AL, primarily by decompressing the anastomotic region. However, its efficacy is generally inferior to that of LCA preservation or anastomotic reinforcement for several physiological reasons.39,40
Limited to mechanical decompression
A transanal tube primarily reduces intraluminal pressure; however, it does not address the fundamental biological determinants of anastomotic integrity, including ischemia or microvascular insufficiency. In patients with sacrificed LCA and compromised distal perfusion, decompression alone cannot prevent ischemic necrosis or microdehiscence. Similarly, it does not actively promote tissue repair, serving only as a passive drain for leaked contents.
Temporal limitation
The tube is typically retained for only several postoperative days, whereas complete anastomotic healing requires weeks. Once the tube is removed, intraluminal pressure gradually returns to baseline, and any preexisting marginal ischemia or suture tension may lead to delayed leakage.
Potential adverse effects
Prolonged placement may cause anal discomfort, sphincter irritation, or transient inhibition of the defecation reflex. Furthermore, reliance on a drainage tube may create a false sense of security, potentially leading to underappreciation of more critical preventive factors such as ensuring adequate perfusion and reinforcement.
In summary, although transanal drainage can modestly reduce early mechanical stress on the anastomosis, it should be viewed as an adjunct rather than a substitute for vascular preservation and precise anastomotic technique. Optimal outcomes are achieved through a combination of adequate perfusion, stable mechanical closure, and selective decompression.
Core role of LCA preservation and the synergistic effects of combined PST techniques
Primary role of LCA preservation
Among the 19 patients with preservation of the LCA, only one case of AL occurred (5.3%), whereas no leakage was observed in any of the combined-intervention subgroups, irrespective of the addition of T technique or S technique. These findings underscore LCA preservation as the central protective factor in maintaining anastomotic integrity. 34
Interestingly, in patients undergoing the P technique without concurrent S or T technique (n = 8), the AL rate increased to 12.5%. This suggests that LCA preservation alone, although pivotal, may be insufficient under adverse intraoperative conditions such as excessive anastomotic tension, local contamination, or subtle microvascular compromise. Therefore, maintaining arterial perfusion forms the physiological foundation of protection; however, adjunctive measures may be necessary to optimize this advantage in high-risk contexts.
From a mechanistic perspective, this observation is consistent with those of previous vascular imaging and fluorescence angiography studies demonstrating that adequate perfusion alone does not guarantee anastomotic healing unless local mechanical stability and pressure equilibrium are simultaneously maintained. 41 Therefore, LCA preservation can be viewed as a necessary but not sufficient condition for AL prevention, with its efficacy modulated by tension, suture integrity, and postoperative intraluminal dynamics.
Potential adjunctive effect of S technique
In patients without preservation of the LCA, the incidence of AL was 6.7% in those undergoing the S technique, compared with 8.3% in those without reinforcement, a nonsignificant but directionally favorable difference (p = 0.71). This trend suggests that sutural reinforcement may partially compensate for reduced perfusion by enhancing mechanical resilience and sealing microscopic defects.
Biomechanically, anastomotic reinforcement stabilizes tissue approximation, distributes local tension more evenly, and seals potential microgaps resulting from stapling irregularities. Under ischemic conditions, such reinforcement may prevent early leakage during the critical inflammatory phase of healing, when tissue strength is lowest. 42 Although statistical power was limited, the consistent downward trend suggests that reinforcement suturing serves as an effective adjunct, particularly in patients with rectal cancer for whom vascular preservation is not feasible because of oncologic constraints.
Localized protective role of the T technique
In the high-risk subgroup without LCA preservation or in the subgroup undergoing the T technique (n = 36), patients in the latter subgroup demonstrated a slightly higher AL rate (11.1%, 1/9) than those without placement (8.3%, 3/36), likely reflecting sampling bias given the small sample size. However, when considered in the context of patients with LCA preservation, the T technique appears to exert its benefit primarily when adequate blood supply is maintained.
Theoretically, the T technique mitigates intraluminal pressure at the anastomotic site, thereby promoting decompression and facilitating mucosal healing. However, under ischemic or mechanically unstable conditions, decompression alone cannot counteract necrotic progression or microdehiscence. Therefore, its efficacy depends on the baseline physiological stability of the anastomosis, particularly perfusion integrity, reinforcing the notion that the T technique should function as a complementary rather than a primary intervention.
Synergistic and interactive effects among P, S, and T techniques
1. P technique + S technique synergy. When the LCA was preserved, even without placement of the transanal tube, none of the patients (n = 2) who underwent the S technique developed AL, whereas in the non-LCA group, the S technique reduced the AL rate from 8.3% to 6.7%. This pattern indicates a potential synergistic amplification effect. Adequate perfusion appears to enhance the mechanical reinforcement benefit, likely by improving fibroblast activity, collagen cross-linking, and mucosal regeneration at the sutured interface. 2. P technique + T technique combination. Similarly, in all nine patients who underwent both LCA preservation and the placement of transanal tube (irrespective of S technique), no AL was observed. Conversely, in 18 patients without LCA preservation but with the placement of the transanal tube, the AL rate reached 11.1% (2/18). These findings suggest that the decompressive benefit of the T technique is conditional on sufficient perfusion. Adequate arterial inflow ensures tissue viability, allowing pressure reduction to effectively lower mechanical stress on a healing anastomosis. 3.Triple-combination effect. The only subgroup with zero AL incidence comprised patients who underwent all three interventions (P technique + T technique + S technique, n = 6). Although the small sample size limits definitive statistical inference, this pattern strongly suggests an additive or synergistic interaction in which preserved perfusion, mechanical reinforcement, and pressure relief collectively address the major pathogenic pathways of AL, including ischemia, mechanical disruption, and excessive luminal stress.
This observation aligns with emerging evidence in colorectal surgery emphasizing multimodal, individualized prevention frameworks rather than single-factor interventions. 43 It underscores the potential of a “physiologic-mechanical-integrative” paradigm for anastomotic protection.
Statistical findings and study limitations
Univariate analyses revealed RR estimates of 0.61 (95% CI: 0.08–4.72) for P technique, 0.70 (95% CI: 0.14–3.51) for S technique, and 0.90 (95% CI: 0.18–4.45) for T technique. Although none reached statistical significance, likely because of the limited sample size, the consistent direction of effect supports the protective potential of all three interventions.
Multivariate logistic regression yielded adjusted OR of 0.55 for P technique, 0.65 for S technique, and 0.85 for T technique, further indicating a trend toward risk reduction. The nonsignificant results should not obscure the biologically plausible and clinically consistent pattern. Importantly, the current model did not incorporate interaction terms, which may underestimate the synergistic effects among these variables. 44 Future analyses with larger, multicenter datasets and advanced interaction modeling are warranted to elucidate these combined effects more robustly.
Clinical implications
Taken together, the data suggest that preservation of the LCA represents the cornerstone of AL prevention, providing a vascular foundation upon which mechanical and decompressive strategies can build. A key limitation of our study is the relatively small sample size of the groups, which may have introduced random bias and limited the generalizability of these findings. Future prospective studies with larger cohorts are needed to validate the efficacy of isolated LCA preservation for AL prevention. An integrated PST approach, simultaneously optimizing blood supply, anastomotic strength, and pressure control, appears to offer the most comprehensive protection. 45
Although the present findings require validation in larger prospective cohorts, they highlight an emerging paradigm: AL prevention should not rely on a single technical maneuver but rather on the synergistic orchestration of vascular, mechanical, and physiological principles tailored to each patient’s intraoperative risk profile.
Conclusion
This study provides a comprehensive clinical and mechanistic analysis of three targeted intraoperative strategies, the P, T, and S techniques, for the prevention of AL following colorectal surgery. The findings demonstrate that LCA preservation plays a central role in preventing AL after colorectal surgery by maintaining adequate perfusion and microvascular stability at the anastomotic site. Although LCA preservation alone provides significant physiological protection, its effect may be further enhanced through reinforcement suturing and transanal decompression, which together improve mechanical stability and reduce intraluminal pressure.
Although statistical significance was not achieved because of the limited sample size, all analyses consistently indicated a protective trend across these interventions. Notably, the combined application of LCA preservation, suturing, and transanal decompression achieved zero AL incidence, highlighting their potential synergistic effect.
Therefore, an integrated strategy that preserves vascular supply, reinforces the anastomosis, and reduces intraluminal pressure may represent an effective approach to reducing the risk of AL in patients with colorectal cancer. Larger prospective studies are warranted to confirm these findings and further optimize surgical protocols.
Footnotes
Acknowledgments
We sincerely thank all patients and their families for their participation and trust in this study. We are grateful to the surgical team, nursing staff, and research coordinators at the Department of Colorectal and Abdominal Tumor Surgery II, Jilin Provincial Cancer Hospital, for their invaluable technical support and dedication throughout the study period.
Author contributions
Chunxiao Lie: Participated in surgery, conducted investigation, performed data curation, and drafted the original manuscript.
Zhaocheng Chi: Responsible for funding acquisition, performed surgery, and managed the project.
Shanshan Qian: Contributed to project management.
All authors have read and agreed to the published version of the manuscript.
Data availability statement
The datasets generated and analyzed during the current study are not publicly available due to privacy and ethical restrictions protecting patient confidentiality but are available from the corresponding author on reasonable request, subject to approval by the Institutional Review Board of Jilin Provincial Cancer Hospital. All relevant data supporting the conclusions of this article are included within the manuscript and its supplementary materials.
Disclosure of potential conflicts of interest
The authors declare that they have no financial or nonfinancial conflicts of interest that are directly or indirectly related to the work submitted for publication.
Ethics statements
All patients included in this study provided written informed consent for the surgical and perioperative treatments received, in accordance with the ethical standards of the institutional review board. All procedures were performed at Jilin Cancer Hospital, Changchun, China, between August 2021 and December 2023. Written informed consent for the publication of clinical details and study findings was obtained from all participating patients prior to data collection, ensuring compliance with patient privacy protection regulations. The study protocol was reviewed and approved by the Institutional Review Board (IRB) of Jilin Cancer Hospital (IRB Approval No. 202012-46-02) prior to patient recruitment.
Funding
This work was supported by the Pandeng Fund of the National Cancer Center (Grant No.: NCC201907B03).